Abstract
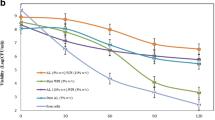
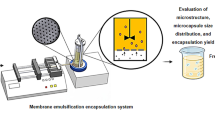
Pea protein-alginate microcapsules with or without a chitosan coating and containing Lactobacillus rhamnosus R0011 and L. helveticus R0052 were produced by extrusion and tested for survivability during storage and in an in vitro gastrointestinal environment. Both microcapsule formulations provided significant protection for cells incubated in synthetic stomach juice at 37°C for 2 h, followed by 3 h in simulated intestinal fluid, relative to non-encapsulated bacteria. However, evaluation of cell viability during 9 weeks of storage at room temperature revealed that chitosan coating significantly improved microcapsule performance compared to non-coated microcapsules. Refrigerated storage had no negative impact on the microcapsule protection ability of both types of microcapsules. Notably, chitosan-containing microcapsules showed much higher bacterial survival counts during challenge tests even after storage. Moreover, the addition of chitosan to the microcapsule formulation did not increase the microcapsule size.
Similar content being viewed by others
References
FAO/WHO. Report of a Joint FAO/WHO expert consultation on evaluation of health and nutritional properties of probiotics in food including powder milk with live lactic acid bacteria. Food and Agriculture Organization of the United Nations, World Health Organization, Geneva, Switzerland (2001)
Varankovich NV, Nickerson MT, Korber DR. Probiotic-based strategies for therapeutic and prophylactic use against multiple gastrointestinal diseases. Front. Microbiol. 685: 1–14 (2015)
Chandramouli V, Kailasapathy K, Peiris P, Jones M. An improved method of microencapsulation and its evaluation to protect Lactobacillus spp. in simulated gastric conditions. J. Microbiol. Meth. 56: 27–35 (2004)
Jiménez-Pranteda ML, Aguilera M, McCartney AL, Hoyles L, Jiménez-Valera M, Náder-Macías ME, Ramos-Cormenzana A, Monteoliva-Sánchez M. Investigation of the impact of feeding Lactobacillus plantarum CRL 1815 encapsulated in microbially derived polymers on the rat fecal microbiota. J. Appl. Microbiol. 113: 399–410 (2012)
Khan NH, Korber DR, Low NH, Nickerson MT. Development of extrusion-based legume protein isolate–alginate capsules for the protection and delivery of the acid sensitive probiotic, Bifidobacterium adolescentis. Food Res. Int. 54: 730–737 (2013)
Varankovich NV, Khan NH, Nickerson MT, Kalmokoff M, Korber DR. Evaluation of pea protein–polysaccharide matrices for encapsulation of acid-sensitive bacteria. Food Res. Int. 70: 118–124 (2015)
Annan NT, Borza AD, Hansen LT. Encapsulation in alginate-coated gelatin microspheres improves survival of the probiotic Bifidobacterium adolescentis 15703T during exposure to simulated gastro-intestinal conditions. Food Res. Int. 41: 184–193 (2008)
Holzapfel WH, Schillinger U. Introduction to pre-and probiotics. Food Res. Int. 35: 109–116 (2002)
Voo WP, Ravindra P, Tey BT, Chan ES. Comparison of alginate and pectin based beads for production of poultry probiotic cells. J. Biosci. Bioeng. 111: 294–299 (2011)
Stone AK, Karalash A, Tyler RT, Warkentin TD, Nickerson MT. Functional attributes of pea protein isolates prepared using different extraction methods and cultivars. Food Res. Int. 76: 31–38 (2015)
Smith A, Perelman M, Hinchcliffe M, Limited AD, Centre AE, Park NS, Nottingham B. Chitosan: A promising safe and immune-enhancing adjuvant for intranasal vaccines. Hum. Vaccin. Immunother. 10: 797–807 (2014)
Hirano S. Chitin biotechnology applications. Biotechnol. Annu. Rev. 2: 237–58 (1996)
Türkoðlu T, Taþcýoðlu S. Novel strategy for the ionotropic crosslinking of chitosan-alginate polyelectrolyte complexes. J. Appl. Polym. Sci. 131: 1–7 (2014)
Wang J, Lian Z, Wang H, Jin X, Liu Y. Synthesis and antimicrobial activity of schiff base of chitosan and acylated chitosan. J. Appl. Polym. Sci. 123: 3242–3247 (2012)
Zheng H, Gao M, Ren Y, Lou R, Xie H, Yu W, Liu X, Ma X. Controlling gel structure to modulate cell adhesion and spreading on the surface of microcapsules. ACS Appl. Mater. Interfaces 8: 19333–19342 (2016)
Chávarri M, Marañón I, Ares R, Ibáñez FC, Marzo F, Villarán C. Microencapsulation of a probiotic and prebiotic in alginate-chitosan capsules improves survival in simulated gastro-intestinal conditions. Int. J. Food Microbiol. 142: 185–189 (2010)
Gareau MG, Wine E, Reardon C, Sherman PM. Probiotics prevent death caused by Citrobacter rodentium infection in neonatal mice. J. Infect. Dis. 201: 81–91 (2010)
Gareau MG, Wine E, Rodrigues DM, Cho JH, Whary MT, Philpott DJ, MacQueen G, Sherman PM. Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60: 307–317 (2011)
Rodrigues DM, Sousa AJ, Johnson-Henry KC, Sherman PM, Gareau MG. Probiotics are effective for the prevention and treatment of Citrobacter rodentium–induced colitis in mice. J. Infect. Dis. 206: 99–109 (2012)
Klemmer KJ, Korber DR, Low NH, Nickerson MT. Pea protein-based capsules for probiotic and prebiotic delivery. Int. J. Food Sci. Tech. 46: 2248–2256 (2011)
Krasaekoopt W, Bhandari B, Deeth H. Evaluation of encapsulation techniques of probiotics for yoghurt. Int. Dairy J. 13: 3–13 (2003)
Pedersen C, Jonsson H, Lindberg JE, Roos S. Microbiological characterization of wet wheat distillers' grain, with focus on isolation of lactobacilli with potential as probiotics. Appl. Environ. Microb. 70: 1522–1527 (2004)
Foerst P, Kulozik U, Schmitt M, Bauer S, Santivarangkna C. Storage stability of vacuum-dried probiotic bacterium Lactobacillus paracasei F19. Food Bioprod. Process. 90: 295–300 (2012)
Champagne C, Mondou F. Effect of polymers and storage temperature on the stability of freeze-dried lactic acid bacteria. Food Res. Int. 29: 555–562 (1996)
Obradovic N, Krunic T. Influence of chitosan coating on mechanical stability of biopolymer carriers with probiotic starter culture in fermented whey beverages. Int. J. Polym. Sci. 2015: 1–8 (2015)
Zanjani MAK, Tarzi BG, Sharifan A, Mohammadi N. Microencapsulation of probiotics by calcium alginate-gelatinized starch with chitosan coating and evaluation of survival in simulated human gastro-intestinal condition. Iran. J. Pharm. Res. 13: 843–852 (2013)
Krasaekoopt W, Bhandari B, Deeth H. The influence of coating materials on some properties of alginate beads and survivability of microencapsulated probiotic bacteria. Int. Dairy J. 14: 737–743 (2004)
Ouwerx C, Velings N, Mestdagh MM, Axelos MA V. Physico-chemical properties and rheology of alginate gel beads formed with various divalent cations. Polym. Gels Netw. 6: 393–408 (1998)
Valenzuela C, Hernández V, Morales MS, Pizarro F. Heme iron release from alginate beads at in vitro simulated gastrointestinal conditions. Biol. Trace Elem. Res. 172: 251–257 (2016)
Li Y, Hu M, Du Y, Xiao H, McClements DJ. Control of lipase digestibility of emulsified lipids by encapsulation within calcium alginate beads. Food Hydrocolloid. 25: 122–130 (2011)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varankovich, N., Martinez, M.F., Nickerson, M.T. et al. Survival of probiotics in pea protein-alginate microcapsules with or without chitosan coating during storage and in a simulated gastrointestinal environment. Food Sci Biotechnol 26, 189–194 (2017). https://doi.org/10.1007/s10068-017-0025-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-017-0025-2