Abstract
Objectives
To analyze the effect of disease-modifying antirheumatic drugs (DMARDs) on the outcome of interstitial lung disease secondary to rheumatoid arthritis (RA-ILD).
Patients and methods
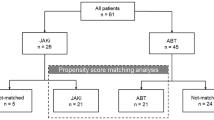
We performed a multicenter, prospective, observational study of patients with RA-ILD receiving DMARDs between 2015 and 2017. The patients were assessed using high-resolution computed tomography and pulmonary function tests at baseline and at 24 months. The radiological assessment was centralized. The main outcome measure at 24 months was changed in lung function (improvement, stabilization, worsening, or death). We recorded the 28-joint Disease Activity Score 28 (DAS28) and adverse events. A logistic regression analysis was performed to identify factors associated with worsening of ILD.
Results
After 24 months, lung disease was stabilized in 40 patients (57.1%), improved in 8 (11.4%), and worse in 21 (30.0%). One patient (1.4%) died. The factors associated with worsening of ILD in the multivariate analysis were treatment with abatacept, tocilizumab, or rituximab (OR, 0.102 [95%CI, 0.015–0.686]), DAS28 (OR, 1.969 [95%CI, 1.005–3.857]), and smoking (OR, 6.937 [95%CI, 1.378–4.900]). During follow-up, 30 patients (42.9%) experienced an adverse event, which was severe in 12 cases (17.1%).
Conclusions
Lung function is stable and inflammatory activity well controlled in most patients with RA-ILD receiving treatment with DMARDs. Non-anti-TNF DMARDs reduce the risk of worsening of lung disease in 90% of patients. The inflammatory activity of RA and smoking, on the other hand, are associated with worsening.
Key Points • We have performed prospectively evaluated lung and joint function in patients with RA-ILD receiving treatment with various DMARDs. • In our study, the lung function is stable and inflammatory activity well controlled in most patients with RA-ILD receiving treatment with DMARDs. • Neither csDMARDs nor anti-TNF agents were associated with a significant risk of worsening of lung disease, whereas non-anti-TNF bDMARDs could reduce the risk of worsening of lung disease. • Smoking and poor control of joint involvement were the main factors associated with worsening of lung disease. |

Similar content being viewed by others
References
Smolen JS, Aletaha D, McInnes IB (2016) Rheumatoid arthritis. Lancet. 388(10055):2023–2038
Hallowell RW, Horton MR (2014) Interstitial lung disease in patients with rheumatoid arthritis: spontaneous and drug induced. Drugs. 74(4):443–450
Gabbay E (2015) ea. Interstitial lung disease in recent onset rheumatoid arthritis.
Mori S (2015) ea. Different risk factors between interstitial lung disease and airway disease in rheumatoid arthritis.
Alexiou I, Germenis A, Koutroumpas A, Kontogianni A, Theodoridou K, Sakkas LI (2008) Anti-cyclic citrullinated peptide-2 (CCP2) autoantibodies and extra-articular manifestations in Greek patients with rheumatoid arthritis. Clin Rheumatol 27(4):511–513
Mena-Vazquez N, Perez Albaladejo L, Manrique-Arija S, Romero Barco CM, Gomez Cano C, Urena Garnica I et al (2019) Analysis of clinical-analytical characteristics in patients with rheumatoid arthritis and interstitial lung disease: case-control study. Reumatol Clin
Olson AL, Swigris JJ, Sprunger DB, Fischer A, Fernandez-Perez ER, Solomon J, Murphy J, Cohen M, Raghu G, Brown KK (2011) Rheumatoid arthritis-interstitial lung disease-associated mortality. Am J Respir Crit Care Med 183(3):372–378
Sihvonen S, Korpela M, Laippala P, Mustonen J, Pasternack A (2004) Death rates and causes of death in patients with rheumatoid arthritis: a population-based study. Scand J Rheumatol 33(4):221–227
Saketkoo LA, Espinoza LR (2008) Rheumatoid arthritis interstitial lung disease: mycophenolate mofetil as an antifibrotic and disease-modifying antirheumatic drug. Arch Intern Med 168. United States:1718–1719
Conway R, Low C, Coughlan RJ, O'Donnell MJ, Carey JJ (2014) Methotrexate and lung disease in rheumatoid arthritis: a meta-analysis of randomized controlled trials. Arthritis Rheum 66(4):803–812
Ramos-Casals M, Brito-Zeron P, Munoz S, Soria N, Galiana D, Bertolaccini L et al (2007) Autoimmune diseases induced by TNF-targeted therapies: analysis of 233 cases. Medicine (Baltimore) 86(4):242–251
Roubille C, Haraoui B (2014) Interstitial lung diseases induced or exacerbated by DMARDS and biologic agents in rheumatoid arthritis: a systematic literature review. Semin Arthritis Rheum 43(5):613–626
Conway R (2015) ea. Methotrexate and lung disease in rheumatoid arthritis: a meta-analysis of randomized controlled trials.
Ju JH, Kim SI, Lee JH, Lee SI, Yoo WH, Choe JY, Chung SH, Lee J, Lee YH, Lee SS, Yoon HJ, Yoon CH, Kim HY, Park SH (2007) Risk of interstitial lung disease associated with leflunomide treatment in Korean patients with rheumatoid arthritis. Arthritis Rheum 56(6):2094–2096
Suissa S, Hudson M, Ernst P (2006) Leflunomide use and the risk of interstitial lung disease in rheumatoid arthritis. Arthritis Rheum 54(5):1435–1439
Rojas-Serrano J (2015) ea. Methotrexate and lung disease in rheumatoid arthritis: comment on the article by Conway et al.
Conway R, Low C, Coughlan RJ, O'Donnell MJ, Carey JJ (2016) Leflunomide use and risk of lung disease in rheumatoid arthritis: a systematic literature review and metaanalysis of randomized controlled trials. J Rheumatol 43(5):855–860
Detorakis EE, Magkanas E, Lasithiotaki I, Sidiropoulos P, Boumpas DT, Gourtsoyiannis N et al (2017) Evolution of imaging findings, laboratory and functional parameters in rheumatoid arthritis patients after one year of treatment with anti-TNF-alpha agents. Clin Exp Rheumatol 35(1):43–52
Nakashita T, Ando K, Kaneko N, Takahashi K, Motojima S (2014) Potential risk of TNF inhibitors on the progression of interstitial lung disease in patients with rheumatoid arthritis. BMJ Open 4(8):e005615
Fernandez-Diaz C, Loricera J, Castaneda S, Lopez-Mejias R, Ojeda-Garcia C, Olive A et al (2018) Abatacept in patients with rheumatoid arthritis and interstitial lung disease: a national multicenter study of 63 patients. Semin Arthritis Rheum 48:22–27
Matteson EL, Bongartz T, Ryu JH, Crowson CS, Hartman TE, Dellaripa PF (2012) Open-label, pilot study of the safety and clinical effects of rituximab in patients with rheumatoid arthritis-associated interstitial pneumonia. Open J Rheumatol Autoimmune Dis 02(03):53–58
Andreina M, Giulia C, Federica F, Elisa G, Vincenzo V, Fabiola A et al (2019) Tocilizumab therapy in rheumatoid arthritis with interstitial lung disease: a multicenter retrospective study. Intern Med J
Hadjinicolaou AV, Nisar MK, Parfrey H, Chilvers ER, Ostor AJ (2012) Non-infectious pulmonary toxicity of rituximab: a systematic review. Rheumatology (Oxford) 51(4):653–662
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO 3rd et al (2010) 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League against rheumatism collaborative initiative. Arthritis Rheum 62(9):2569–2581
Travis WD, Costabel U, Hansell DM, King TE Jr, Lynch DA, Nicholson AG et al (2013) An official American Thoracic Society/European Respiratory Society statement: update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 188(6):733–748
Gaujoux-Viala C, Mouterde G, Baillet A, Claudepierre P, Fautrel B, Le Loet X et al (2012) Evaluating disease activity in rheumatoid arthritis: which composite index is best? A systematic literature analysis of studies comparing the psychometric properties of the DAS, DAS28, SDAI and CDAI. Joint Bone Spine 79(2):149–155
Maska L, Anderson J, Michaud K (2011) Measures of functional status and quality of life in rheumatoid arthritis: Health Assessment Questionnaire Disability Index (HAQ), Modified Health Assessment Questionnaire (MHAQ), Multidimensional Health Assessment Questionnaire (MDHAQ), Health Assessment Questionnaire II (HAQ-II), Improved Health Assessment Questionnaire (Improved HAQ), and Rheumatoid Arthritis Quality of Life (RAQoL). Arthritis Care Res 63(Suppl 11):S4–S13
Perez-Alvarez R, Perez-de-Lis M, Diaz-Lagares C, Pego-Reigosa JM, Retamozo S, Bove A, Brito-Zeron P, Bosch X, Ramos-Casals M (2011) Interstitial lung disease induced or exacerbated by TNF-targeted therapies: analysis of 122 cases. Semin Arthritis Rheum 41(2):256–264
Mera-Varela A, Perez-Pampin E (2014) Abatacept therapy in rheumatoid arthritis with interstitial lung disease. J Clin Rheumatol 20(8):445–446
Curtis JR, Sarsour K, Napalkov P, Costa LA, Schulman KL (2015) Incidence and complications of interstitial lung disease in users of tocilizumab, rituximab, abatacept and anti-tumor necrosis factor alpha agents, a retrospective cohort study. Arthritis Res Ther 17:319
Nakashita T, Ando K, Takahashi K, Motojima S (2016) Possible effect of abatacept on the progression of interstitial lung disease in rheumatoid arthritis patients. Respir Investig 54(5):376–379
Jimenez-Alvarez L, Arreola JL, Ramirez-Martinez G, Ortiz-Quintero B, Gaxiola M, Reynoso-Robles R et al (2011) The effect of CTLA-4Ig, a CD28/B7 antagonist, on the lung inflammation and T cell subset profile during murine hypersensitivity pneumonitis. Exp Mol Pathol 91(3):718–722
Md Yusof MY, Kabia A, Darby M, Lettieri G, Beirne P, Vital EM, Dass S, Emery P (2017) Effect of rituximab on the progression of rheumatoid arthritis-related interstitial lung disease: 10 years' experience at a single Centre. Rheumatology (Oxford) 56:1348–1357
Kawashiri SY, Kawakami A, Sakamoto N, Ishimatsu Y, Eguchi K (2012) A fatal case of acute exacerbation of interstitial lung disease in a patient with rheumatoid arthritis during treatment with tocilizumab. Rheumatol Int 32(12):4023–4026
Wendling D, Vidon C, Godfrin-Valnet M, Rival G, Guillot X, Prati C (2013) Exacerbation of combined pulmonary fibrosis and emphysema syndrome during tocilizumab therapy for rheumatoid arthritis. Joint Bone Spine 80(6):670–671
Zhang Y, Li H, Wu N, Dong X, Zheng Y (2017) Retrospective study of the clinical characteristics and risk factors of rheumatoid arthritis-associated interstitial lung disease. Clin Rheumatol 36(4):817–823
Deane KD, Nicolls MR (2013) Developing better biomarkers for connective tissue disease-associated interstitial lung disease: citrullinated hsp90 autoantibodies in rheumatoid arthritis. Arthritis Rheum 65(4):864–868
Fabre A, Treacy A, Lavelle LP, Narski M, Faheem N, Healy D, Dodd JD, Keane MP, Egan JJ, Jebrak G, Mal H, Butler MW (2017) Smoking-related interstitial fibrosis: evidence of radiologic regression with advancing age and smoking cessation. Copd. 14(6):603–609
Sparks JA, He X, Huang J, Fletcher EA, Zaccardelli A, Friedlander HM, Gill RR, Hatabu H, Nishino M, Murphy DJ, Iannaccone CK, Mahmoud TG, Frits ML, Lu B, Rosas IO, Dellaripa PF, Weinblatt ME, Karlson EW, Shadick NA, Doyle TJ (2019) Rheumatoid arthritis disease activity predicting incident clinically apparent rheumatoid arthritis-associated interstitial lung disease: a prospective cohort study. Arthritis Rheum 71(9):1472–1482
Acknowledgments
Grant for medical researchers of the “Fundación Española de Reumatología”. To the Spanish Rheumatology Society (SER) for the translation of the manuscript.
Funding
Grant for Medical Researchers of the “Fundación Española de Reumatología” 2019.
Author information
Authors and Affiliations
Contributions
NMV participated in the design of the study, carried out patient recruitment and statistical analysis, and drafted the manuscript. FJGN and SMA were a contributor in including patients. They were a major contributor in writing the manuscript and they were a contributor in analyzing and interpreting the patient data. MCAH and MIPM collected radiology data. IUG, MCRB, FGJN, IAO, LPA, and CGC were a major contributor in including patients. AFN: a contributor in writing the manuscript. He was a contributor in analyzing and interpreting the patient data. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
All subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Málaga (“Comité de Ética de la Investigación de Málaga”) (Project identification code 4/2015, P15).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Mena-Vázquez, N., Godoy-Navarrete, F.J., Manrique-Arija, S. et al. Non-anti-TNF biologic agents are associated with slower worsening of interstitial lung disease secondary to rheumatoid arthritis. Clin Rheumatol 40, 133–142 (2021). https://doi.org/10.1007/s10067-020-05227-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05227-9