Abstract
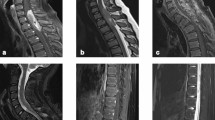
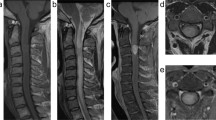
The CIC-DUX4 translocation is the most common genetic alteration of small round cell sarcomas without EWSR1 rearrangement. These “Ewing-like sarcomas” usually occur in peripheral soft tissues, and rare primary central nervous system (CNS) tumors have been described. We report a rare case of primary spinal intramedullary Ewing-like sarcoma harboring CIC-DUX4 translocation. A 23-year-old man presented with weakness in the extremities. Magnetic resonance imaging revealed a large intramedullary tumor spanning C3–C5 with heterogeneous enhancement following gadolinium administration. Histologically, most of the tumor displayed dense myeloid proliferation composed of medium- to slightly small-sized primitive cells. Postoperatively, he received local adjuvant radiation therapy without tumor progression for 10 months. Target RNA sequencing analysis revealed the CIC-DUX4 fusion gene. Methylation array analysis resulted in a diagnosis of “methylation class CNS Ewing sarcoma family tumor with CIC alteration”. Although this tumor lacked characteristic histological features such as lobular structures in association with desmoplastic stroma, relatively uniform nuclei with prominent nucleoli and eosinophilic cytoplasm, which are often found in CIC-rearranged sarcomas of soft tissue, were identified. Recently, many CNS and soft tissue tumors require genetic analysis for precise diagnosis. To consider certain molecular testing, careful histological examination is essential.



Similar content being viewed by others
References
Kawamura-Saito M, Yamazaki Y, Kaneko K et al (2006) Fusion between CIC and DUX4 up-regulates PEA3 family genes in Ewing-like sarcomas with t(4;19)(q35;q13) translocation. Hum Mol Genet 15:2125–2137
Graham C, Chilton-MacNeill S, Zielenska M et al (2012) The CIC-DUX4 fusion transcript is present in a subgroup of pediatric primitive round cell sarcomas. Hum Pathol 43:180–189
Italiano A, Sung YS, Zhang L et al (2012) High prevalence of CIC fusion with double-homeobox (DUX4) transcription factors in EWSR1-negative undifferentiated small blue round cell sarcomas. Genes Chromosomes Cancer 51:207–218
Choi EY, Thomas DG, McHugh JB et al (2013) Undifferentiated small round cell sarcoma with t(4;19)(q35;q13.1) CIC-DUX4 fusion: a novel highly aggressive soft tissue tumor with distinctive histopathology. Am J Surg Pathol 37:1379–1386
Fletcher CDM, Bridge JA, Hogendoorn PCW et al (2013) WHO classification of tumours of soft tissue and bone, 4th edn. IARC, Lyon
Specht K, Sung YS, Zhang L et al (2014) Distinct transcriptional signature and immunoprofile of CIC- DUX4 positive round cell tumors compared to EWSR1-rearranged Ewing sarcomas—further evidence towards distinct pathologic entities. Genes Chromosomes Cancer 53:622–633
Smith SC, Buehler D, Choi EYM et al (2015) CIC-DUX sarcomas demonstrate frequent MYC amplification and ETS-family transcription factor expression. Mod Pathol 28:57–68
Gambarotti M, Benini S, Gamberi G et al (2016) CIC-DUX4 fusion-positive round-cell sarcomas of soft tissue and bone: a single-institution morphological and molecular analysis of seven cases. Histopathol 69:624–634
Yoshida A, Goto K, Kodaira M et al (2016) CIC-rearranged sarcomas: a study of 20 cases and comparisons with Ewing sarcomas. Am J Surg Pathol 40:313–323
Antonescu CR, Owosho AA, Zhang L et al (2017) Sarcomas with CIC-rearrangements are a distinct pathologic entity with aggressive outcome: a clinicopathologic and molecular study of 115 cases. Am J Surg Pathol 41:941–949
Jiménez G, Shvartsman SY, Paroush ZJ (2012) The Capicua repressor–a general sensor of RTK signaling in development and disease. Cell Sci 125:1383–1391
Bielle F, Zanello M, Guillemot D et al (2014) Unusual primary cerebral localization of a CIC-DUX4 translocation tumor of the Ewing sarcoma family. Acta Neuropathol 128:309–311
Ito M, Ishikawa M, Kitajima M et al (2016) A case report of CIC-rearranged undifferentiated small round cell sarcoma in the cerebrum. Diagn Cytopathol 44:828–832
Donahue JE, Yakirevich E, Zhong S et al (2018) Primary spinal epidural CIC-DUX4 undifferentiated sarcoma in a child. Pediatr Dev Pathol 21:411–417
Sturm D, Orr BA, Toprak UH et al (2016) New brain tumor entities emerge from molecular classification of CNS-PNETs. Cell 164:1060–1072
Smith R, Owen LA, Trem DJ et al (2006) Expression profiling of EWS/FLI identifies NKX2.2 as a critical target gene in Ewing’s sarcoma. Cancer Cell 9:405–416
Yoshida A, Sekine S, Tsuta K et al (2012) NKX2.2 is a useful immunohistochemical marker for Ewing sarcoma. Am J Surg Pathol 36:993–999
Shibuya R, Matsuyama A, Nakamoto M et al (2014) The combination of CD99 and NKX2.2, a transcriptional target of EWSR1-FLI1, is highly specific for the diagnosis of Ewing sarcoma. Virchows Arch 465:599–605
Louis DN, Ohgaki H, Wiestler OD et al (2016) WHO classification of tumours of the central nervous system, revised, 4th edn. IARC, Lyon
Perry A (2016) WHO’s arrived in 2016! An updated weather forecast for integrated brain tumor diagnosis. Brain Tumor Pathol 33:157–160
Komori T (2017) Updated 2016 WHO classification of tumors of the CNS: turning the corner where molecule meets pathology. Brain Tumor Pathol 34:139–140
Komori T (2020) Updating the grading criteria for adult diffuse gliomas: beyond the WHO2016CNS classification. Brain Tumor Pathol 37:1–4
Yokoo H, Kinjo S, Hirato J et al (2006) Fluorescence in situ hybridization targeted for chromosome 1p of oligodendrogliomas (in Japanese). Rinsho Kensa 50:761–766
Sugita S, Arai Y, Tonooka A et al (2014) A novel CIC-FOXO4 gene fusion in undifferentiated small round cell sarcoma: a genetically distinct variant of Ewing-like sarcoma. Am J Surg Pathol 38:1571–1576
Yoshida A, Arai Y, Kobayashi E et al (2017) CIC break-apart fluorescence in situ hybridization misses a subset of CIC-DUX4 sarcomas: a clinicopathological and molecular study. Histopathology 71:461–469
Ikota H, Nobusawa S, Arai H et al (2015) Evaluation of IDH1 status in diffusely infiltrating gliomas by immunohistochemistry using anti-mutant and wild type IDH1 antibodies. Brain Tumor Pathol 32:237–244
Kao YC, Sung YS, Zhang L et al (2016) Recurrent BCOR internal tandem duplication and YWHAE-NUTM2B fusions in soft tissue undifferentiated round cell sarcoma of infancy: overlapping genetic features with clear cell sarcoma of kidney. Am J Surg Pathol 40:1009–1020
Yoshida Y, Nobusawa S, Nakata S et al (2018) CNS high-grade neuroepithelial tumor with BCOR internal tandem duplication: a comparison with its counterparts in the kidney and soft tissue. Brain Pathol 28:710–720
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25:1754–1760
Capper D, Stichel D, Sahm F et al (2018) DNA methylation-based classification of central nervous system tumours. Nature 555:469–474
Lazo de la Vega L, Hovelson DH, Cani AK et al (2016) Targeted next generation sequencing of CIC-DUX4 soft tissue sarcomas demonstrates low mutational burden and recurrent chromosome 1p loss. Hum Pathol 58:161–170
Lee JC, Villanueva-Meyer JE, Ferris SP et al (2019) Clinicopathologic and molecular features of intracranial desmoplastic small round cell tumors. Brain Pathol 30:213–225
Ordóñez NG (1998) Desmoplastic small round cell tumor: I: a histopathologic study of 39 cases with emphasis on unusual histological patterns. Am J Surg Pathol 22:1303–1313
Zhang PJ, Goldblum JR, Pawel BR et al (2003) Immunophenotype of desmoplastic small round cell tumors as detected in cases with EWS-WT1 gene fusion product. Mod Pathol 16:229–235
Al-Ibraheemi A, Broehm C, Tanas MR et al (2019) Desmoplastic small round cell tumors with atypical presentations: a report of 34 cases. Int J Surg Pathol 27:236–243
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yamada, S., Muto, J., De Leon, J.C.A. et al. Primary spinal intramedullary Ewing-like sarcoma harboring CIC-DUX4 translocation: a similar cytological appearance as its soft tissue counterpart but no lobulation in association with desmoplastic stroma. Brain Tumor Pathol 37, 111–117 (2020). https://doi.org/10.1007/s10014-020-00366-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10014-020-00366-y