Abstract
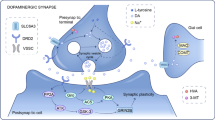
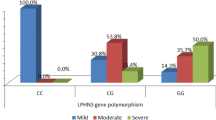
Polymorphisms in latrophilin 3 (LPHN3) were recently reported to be associated with attention-deficit/hyperactivity disorder (ADHD), and subsequently other researchers tried to replicate the findings in different populations. This study was aimed to confirm the role of the LPHN3 in ADHD and explore the potential interactions with environmental risk factors in Chinese Han population. We examined the association of LPHN3 with ADHD in a population of 473 ADHD children and 585 controls. As a supplement of ADHD diagnosis, Conners Parent Symptom Questionnaire (PSQ) was used to evaluate ADHD symptoms. Blood lead levels (BLLs) were measured by atomic absorption spectrophotometry and other potential environmental risk factors were determined via a questionnaire filled out by the parents. Finally, after validation in an independent sample (284 cases and 390 controls), we observed significant associations between LPHN3 variants rs1868790 and ADHD risk in combined stage within codominant model [TA/AA: OR (95% CI) = 1.636 (1.325–2.021)], dominant model [OR (95% CI) = 1.573 (1.288–1.922)], and additive model [OR (95% CI) = 1.535 (1.266–1.862)]. Furthermore, rs1868790 significantly interacted with BLLs and maternal stress to modify ADHD susceptibility (P < 0.05), and rs1868790 was found to be related with ADHD symptoms (P < 0.05). Expression quantitative trait loci analysis further indicated that rs1868790 took part in the regulation of LPHN3 gene expression. As the first study to comprehensively explore the role of LPHN3 in ADHD in Chinese children, our research suggests that LPHN3 gene has a significant effect on the ADHD in a Chinese population.


Similar content being viewed by others
References
Faraone SV, Biederman J, Mennin D, Gershon J, Tsuang MT (1996) A prospective four-year follow-up study of children at risk for ADHD: psychiatric, neuropsychological, and psychosocial outcome. J Am Acad Child Adolesc Psychiatry 35(11):1449–1459. https://doi.org/10.1097/00004583-199611000-00013
Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, Sklar P (2005) Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry 57(11):1313–1323. https://doi.org/10.1016/j.biopsych.2004.11.024
Neale BM, Medland SE, Ripke S, Asherson P, Franke B, Lesch K-P, Faraone SV, Nguyen TT, Schäfer H, Holmans P, Daly M, Steinhausen H-C, Freitag C, Reif A, Renner TJ, Romanos M, Romanos J, Walitza S, Warnke A, Meyer J, Palmason H, Buitelaar J, Vasquez AA, Lambregts-Rommelse N, Gill M, Anney RJL, Langely K, O’Donovan M, Williams N, Owen M, Thapar A, Kent L, Sergeant J, Roeyers H, Mick E, Biederman J, Doyle A, Smalley S, Loo S, Hakonarson H, Elia J, Todorov A, Miranda A, Mulas F, Ebstein RP, Rothenberger A, Banaschewski T, Oades RD, Sonuga-Barke E, McGough J, Nisenbaum L, Middleton F, Hu X, Nelson S (2010) Meta-analysis of genome-wide association studies of attention deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 49(9):884–897. https://doi.org/10.1016/j.jaac.2010.06.008
Li Z, S-h Chang, L-y Zhang, Gao L, Wang J (2014) Molecular genetic studies of ADHD and its candidate genes: a review. Psychiatry Res 219(1):10–24. https://doi.org/10.1016/j.psychres.2014.05.005
Arcos-Burgos M, Jain M, Acosta M, Shively S, Stanescu H, Wallis D, Domene S, Karkera J (2010) A common variant of the latrophilin 3 gene, LPHN3, confers susceptibility to ADHD and predicts effectiveness of stimulant medication. Mol Psychiatry 15(11):1053–1066. https://doi.org/10.1038/mp.2010.6
Hwang IW, Lim MH, Kwon HJ, Jin HJ (2015) Association of LPHN3 rs6551665 A/G polymorphism with attention deficit and hyperactivity disorder in Korean children. Gene 566(1):68–73. https://doi.org/10.1016/j.gene.2015.04.033
Gomez-Sanchez CI, Riveiro-Alvarez R, Soto-Insuga V, Rodrigo M, Tirado-Requero P, Mahillo-Fernandez I, Abad-Santos F, Carballo JJ, Dal-Ré R, Ayuso C (2015) Attention deficit hyperactivity disorder: genetic association study in a cohort of Spanish children. Behav Brain Funct BBF 12(1):2. https://doi.org/10.1186/s12993-015-0084-6
Ribases M, Ramos-Quiroga JA, Sanchez-Mora C, Bosch R, Richarte V, Palomar G, Gastaminza X, Bielsa A, Arcos-Burgos M, Muenke M, Castellanos FX, Cormand B, Bayes M, Casas M (2011) Contribution of LPHN3 to the genetic susceptibility to ADHD in adulthood: a replication study. Genes Brain Behav 10(2):149–157. https://doi.org/10.1111/j.1601-183X.2010.00649.x
Bruxel EM, Salatino-Oliveira A, Akutagava-Martins GC, Tovo-Rodrigues L, Genro JP, Zeni CP, Polanczyk GV, Chazan R, Schmitz M, Arcos-Burgos M, Rohde LA, Hutz MH (2015) LPHN3 and attention-deficit/hyperactivity disorder: a susceptibility and pharmacogenetic study. Genes Brain Behav 14(5):419–427. https://doi.org/10.1111/gbb.12224
Acosta MT, Swanson J, Stehli A, Molina BSG, The MTAT, Martinez AF, Arcos-Burgos M, Muenke M (2016) ADGRL3 (LPHN3) variants are associated with a refined phenotype of ADHD in the MTA study. Mol Genet Genom Med 4(5):540–547. https://doi.org/10.1002/mgg3.230
Martinez AF, Abe Y, Hong S, Molyneux K, Yarnell D, Löhr H, Driever W, Acosta MT, Arcos-Burgos M, Muenke M (2016) An ultraconserved brain-specific enhancer within ADGRL3 (LPHN3) underpins attention-deficit/hyperactivity disorder susceptibility. Biol Psychiatry 80(12):943–954. https://doi.org/10.1016/j.biopsych.2016.06.026
Choudhry Z, Sengupta SM, Grizenko N, Fortier ME, Thakur GA, Bellingham J, Joober R (2012) LPHN3 and attention-deficit/hyperactivity disorder: interaction with maternal stress during pregnancy. J Child Psychol Psychiatry 53(8):892–902. https://doi.org/10.1111/j.1469-7610.2012.02551.x
Labbe A, Liu A, Atherton J, Gizenko N, Fortier ME, Sengupta SM, Ridha J (2012) Refining psychiatric phenotypes for response to treatment: contribution of LPHN3 in ADHD. Am J Med Genet Part B Neuropsychiatr Genet 159B(7):776–785. https://doi.org/10.1002/ajmg.b.32083
Fallgatter AJ, Ehlis A-C, Dresler T, Reif A, Jacob CP, Arcos-Burgos M, Muenke M, Lesch K-P (2013) Influence of a Latrophilin 3 (LPHN3) risk haplotype on event-related potential measures of cognitive response control in attention-deficit hyperactivity disorder (ADHD). Eur Neuropsychopharmacol 23(6):458–468. https://doi.org/10.1016/j.euroneuro.2012.11.001
Jain M, Velez J, Acosta M, Palacio L, Balog J, Roessler E, Pineda D, Londono A, Palacio J, Arbelaez A (2012) A cooperative interaction between LPHN3 and 11q doubles the risk for ADHD. Mol Psychiatry 17(7):741–747. https://doi.org/10.1038/mp.2011.59
Acosta M, Velez JI, Bustamante M, Balog J, Arcos-Burgos M, Muenke M (2011) A two-locus genetic interaction between LPHN3 and 11q predicts ADHD severity and long-term outcome. Transl Psychiatry. https://doi.org/10.1038/tp.2011.14
Riva V, Marino C, Giorda R, Molteni M, Nobile M (2015) The role of DCDC2 genetic variants and low socioeconomic status in vulnerability to attention problems. Eur Child Adolesc Psychiatry 24(3):309–318. https://doi.org/10.1007/s00787-014-0580-5
Riva V, Battaglia M, Nobile M, Cattaneo F, Lazazzera C, Mascheretti S, Giorda R, Mérette C, Émond C, Maziade M, Marino C (2015) GRIN2B predicts attention problems among disadvantaged children. Eur Child Adolesc Psychiatry 24(7):827–836. https://doi.org/10.1007/s00787-014-0627-7
van Baar AL, Vermaas J, Knots E, de Kleine MJK, Soons P (2009) Functioning at school age of moderately preterm children born at 32 to 36 weeks’ gestational age. Pediatrics 124(1):251–257. https://doi.org/10.1542/peds.2008-2315
Froehlich TE, Anixt JS, Loe IM, Chirdkiatgumchai V, Kuan L, Gilman RC (2011) Update on environmental risk factors for attention-deficit/hyperactivity disorder. Curr Psychiatry Rep 13(5):333. https://doi.org/10.1007/s11920-011-0221-3
Silva D, Colvin L, Hagemann E, Bower C (2014) Environmental risk factors by gender associated with attention-deficit/hyperactivity disorder. Pediatrics 133(1):e14–e22. https://doi.org/10.1542/peds.2013-1434
Eichler A, Hudler L, Grunitz J, Grimm J, Raabe E, Goecke TW, Fasching PA, Beckmann MW, Kratz O, Moll GH, Kornhuber J, Heinrich H (2018) Effects of prenatal alcohol consumption on cognitive development and ADHD-related behaviour in primary-school age: a multilevel study based on meconium ethyl glucuronide. J Child Psychol Psychiatry 59(2):110–118. https://doi.org/10.1111/jcpp.12794
Huang L, Wang Y, Zhang L, Zheng Z, Zhu T, Qu Y, Mu D (2018) Maternal smoking and attention-deficit/hyperactivity disorder in offspring: a meta-analysis. Pediatrics. https://doi.org/10.1542/peds.2017-2465
Van den Bergh BRH, van den Heuvel MI, Lahti M, Braeken M, de Rooij SR, Entringer S, Hoyer D, Roseboom T, Räikkönen K, King S, Schwab M (2017) Prenatal developmental origins of behavior and mental health: the influence of maternal stress in pregnancy. Neurosci Biobehav Rev. https://doi.org/10.1016/j.neubiorev.2017.07.003
Byun Y-H, Ha M, Kwon H-J, Hong Y-C, Leem J-H, Sakong J, Kim SY, Lee CG, Kang D, Choi H-D, Kim N (2013) Mobile phone use, blood lead levels, and attention deficit hyperactivity symptoms in children: a longitudinal study. PLoS One 8(3):e59742. https://doi.org/10.1371/journal.pone.0059742
Huang S, Hu H, Sánchez BN, Peterson KE, Ettinger AS, Lamadrid-Figueroa H, Schnaas L, Mercado-García A, Wright RO, Basu N, Cantonwine DE, Hernández-Avila M, Téllez-Rojo MM (2016) Childhood blood lead levels and symptoms of attention deficit hyperactivity disorder (ADHD): a cross-sectional study of mexican children. Environ Health Perspect 124(6):868–874. https://doi.org/10.1289/ehp.1510067
Gu X, Yuan F-F, Huang X, Hou Y, Wang M, Lin J, Wu J (2018) Association of PIK3CG gene polymorphisms with attention-deficit/hyperactivity disorder: a case–control study. Prog Neuro Psychopharmacol Biol Psychiatry 81(Supplement C):169–177. https://doi.org/10.1016/j.pnpbp.2017.10.020
Dadds MR, Schollar-Root O, Lenroot R, Moul C, Hawes DJ (2016) Epigenetic regulation of the DRD4 gene and dimensions of attention-deficit/hyperactivity disorder in children. Eur Child Adolesc Psychiatry 25(10):1081–1089. https://doi.org/10.1007/s00787-016-0828-3
Gong Y, Cai T (1993) Chinese-Wechsler intelligence scale for children Hunan. Map Press, Beijing
Furtado EF, Roriz STdS (2016) Inattention and impulsivity associated with prenatal alcohol exposure in a prospective cohort study with 11-years-old Brazilian children. Eur Child Adolesc Psychiatry 25(12):1327–1335. https://doi.org/10.1007/s00787-016-0857-y
Yuan F-F, Gu X, Huang X, Hou Y-W, Zhong Y, Lin J, Wu J (2017) Attention-deficit/hyperactivity disorder associated with KChIP1 rs1541665 in Kv channels accessory proteins. PLoS One 12(11):e0188678. https://doi.org/10.1371/journal.pone.0188678
Skoglund C, Chen Q, D´Onofrio BM, Lichtenstein P, Larsson H (2014) Familial confounding of the association between maternal smoking during pregnancy and ADHD in offspring. J Child Psychol Psychiatry 55(1):61–68. https://doi.org/10.1111/jcpp.12124
Browne HA, Modabbernia A, Buxbaum JD, Hansen SN, Schendel DE, Parner ET, Reichenberg A, Grice DE (2016) Prenatal maternal smoking and increased risk for Tourette syndrome and chronic tic disorders. J Am Acad Child Adolesc Psychiatry 55(9):784–791. https://doi.org/10.1016/j.jaac.2016.06.010
Langley K, Heron J, Smith GD, Thapar A (2012) Maternal and paternal smoking during pregnancy and risk of ADHD symptoms in offspring: testing for intrauterine effects. Am J Epidemiol 176(3):261–268. https://doi.org/10.1093/aje/kwr510
Li Y, Zeng Y, Zhu W, Cui Y, Li J (2016) Path model of antenatal stress and depressive symptoms among Chinese primipara in late pregnancy. BMC Pregnancy Childbirth 16(1):180–186. https://doi.org/10.1186/s12884-016-0972-2
Fan J, Du YS, Wang LW (2005) The norm and reliability of the Conners Parent Symptom Questionnaire in Chinese urban children. Shanghai Arch Psychiatry 17(6):321–323
Young J, Rugino T, Dammerman R, Lyne A, Newcorn JH (2014) Efficacy of guanfacine extended release assessed during the morning, afternoon, and evening using a modified Conners’ Parent Rating Scale–revised: short form. J Child Adolesc Psychopharmacol 24(8):435–441. https://doi.org/10.1089/cap.2013.0134
Lee PH, Shatkay H (2008) F-SNP: computationally predicted functional SNPs for disease association studies. Nucleic Acids Res 36(Database issue):D820–D824. https://doi.org/10.1093/nar/gkm904
Ward LD, Kellis M (2012) HaploReg: a resource for exploring chromatin states, conservation, and regulatory motif alterations within sets of genetically linked variants. Nucleic Acids Res 40(D1):D930–D934. https://doi.org/10.1093/nar/gkr917
Ramasamy A, Trabzuni D, Guelfi S, Varghese V, Smith C, Walker R, De T, Consortium UKBE, North American Brain Expression C, Coin L, de Silva R, Cookson MR, Singleton AB, Hardy J, Ryten M, Weale ME (2014) Genetic variability in the regulation of gene expression in ten regions of the human brain. Nat Neurosci 17(10):1418–1428. https://doi.org/10.1038/nn.3801
Andersson T, Alfredsson L, Källberg H, Zdravkovic S, Ahlbom A (2005) Calculating measures of biological interaction. Eur J Epidemiol 20(7):575–579. https://doi.org/10.1007/s10654-005-7835-x
Hahn LW, Ritchie MD, Moore JH (2003) Multifactor dimensionality reduction software for detecting gene–gene and gene–environment interactions. Bioinformatics 19(3):376–382. https://doi.org/10.1093/bioinformatics/btf869
Lubin JH, Gail MH (1990) On power and sample size for studying features of the relative odds of disease. Am J Epidemiol 131(3):552–566. https://doi.org/10.1093/oxfordjournals.aje.a115530
Sánchez-Mora C, Richarte V, Garcia-Martínez I, Pagerols M, Corrales M, Bosch R, Vidal R, Viladevall L, Casas M, Cormand B, Ramos-Quiroga JA, Ribasés M (2015) Dopamine receptor DRD4 gene and stressful life events in persistent attention deficit hyperactivity disorder. Am J Med Genet Part B Neuropsychiatr Genet 168(6):480–491. https://doi.org/10.1002/ajmg.b.32340
Konrad K, Eickhoff SB (2010) Is the ADHD brain wired differently? A review on structural and functional connectivity in attention deficit hyperactivity disorder. Hum Brain Mapp 31(6):904–916. https://doi.org/10.1002/hbm.21058
Lange M, Norton W, Coolen M, Chaminade M, Merker S, Proft F, Schmitt A, Vernier P, Lesch KP, Bally-cuif L (2012) The ADHD-susceptibility gene lphn3.1 modulates dopaminergic neuron formation and locomotor activity during zebrafish development. Mol Psychiatry 17(9):946–954. https://doi.org/10.1038/mp.2012.29
Reuter I, Knaup S, Romanos M, Lesch K-P, Drepper C, Lillesaar C (2016) Developmental exposure to acetaminophen does not induce hyperactivity in zebrafish larvae. J Neural Transm 123(8):841–848. https://doi.org/10.1007/s00702-016-1556-z
Lange M, Froc C, Grunwald H, Norton WHJ, Bally-Cuif L (2018) Pharmacological analysis of zebrafish lphn3.1 morphant larvae suggests that saturated dopaminergic signaling could underlie the ADHD-like locomotor hyperactivity. Prog Neuro Psychopharmacol Biol Psychiatry 84(Pt A):181–189. https://doi.org/10.1016/j.pnpbp.2018.02.010
Orsini CA, Setlow B, DeJesus M, Galaviz S, Loesch K, Ioerger T, Wallis D (2016) Behavioral and transcriptomic profiling of mice null for Lphn3, a gene implicated in ADHD and addiction. Mol Genet Genom Med 4(3):322–343. https://doi.org/10.1002/mgg3.207
Wallis D, Hill DS, Mendez IA, Abbott LC, Finnell RH, Wellman PJ, Setlow B (2012) Initial characterization of mice null for Lphn3, a gene implicated in ADHD and addiction. Brain Res 1463:85–92. https://doi.org/10.1016/j.brainres.2012.04.053
Hawi Z, Cummins TDR, Tong J, Johnson B, Lau R, Samarrai W, Bellgrove MA (2015) The molecular genetic architecture of attention deficit hyperactivity disorder. Mol Psychiatry 20(3):289–297. https://doi.org/10.1038/mp.2014.183
Wu J, Xiao H, Sun H, Zou L, Zhu L-Q (2012) Role of dopamine receptors in ADHD: a systematic meta-analysis. Mol Neurobiol 45(3):605–620. https://doi.org/10.1007/s12035-012-8278-5
Wermter A-K, Laucht M, Schimmelmann BG, Banaschweski T, Sonuga-Barke EJS, Rietschel M, Becker K (2010) From nature versus nurture, via nature and nurture, to gene × environment interaction in mental disorders. Eur Child Adolesc Psychiatry 19(3):199–210. https://doi.org/10.1007/s00787-009-0082-z
Seckl JR, Holmes MC (2007) Mechanisms of disease: glucocorticoids, their placental metabolism and fetal “programming” of adult pathophysiology. Nat Clin Pract Endocrinol Metab 3(6):479–488. https://doi.org/10.1038/ncpendmet0515
Eichel K, von Zastrow M (2018) Subcellular organization of GPCR signaling. Trends Pharmacol Sci 39(2):200–208. https://doi.org/10.1016/j.tips.2017.11.009
Gustavsson N, Wu B, Han W (2012) Calcium sensing in exocytosis. In: Islam MS (ed) Calcium signaling. Springer, Dordrecht, pp 731-757. https://doi.org/10.1007/978-94-007-2888-2_32
Van Den Bergh BRH (2011) Developmental programming of early brain and behaviour development and mental health: a conceptual framework. Dev Med Child Neurol 53:19–23. https://doi.org/10.1111/j.1469-8749.2011.04057.x
Joo H, Lim M-H, Ha M, Kwon H-J, Yoo SJ, Choi K-H, Paik K-C (2017) Secondhand smoke exposure and low blood lead levels in association with attention-deficit hyperactivity disorder and its symptom domain in children: a community-based case–control study. Nicotine Tob Res 19(1):94–101. https://doi.org/10.1093/ntr/ntw152
Eubig PA, Aguiar A, Schantz SL (2010) Lead and PCBs as risk factors for attention deficit/hyperactivity disorder. Environ Health Perspect 118(12):1654–1667. https://doi.org/10.1289/ehp.0901852
Arbuckle TE, Davis K, Boylan K, Fisher M, Fu J (2016) Bisphenol A, phthalates and lead and learning and behavioral problems in Canadian children 6–11 years of age: CHMS 2007–2009. NeuroToxicology 54(Supplement C):89–98. https://doi.org/10.1016/j.neuro.2016.03.014
Goodlad JK, Marcus DK, Fulton JJ (2013) Lead and attention-deficit/hyperactivity disorder (ADHD) symptoms: a meta-analysis. Clin Psychol Rev 33(3):417–425. https://doi.org/10.1016/j.cpr.2013.01.009
Aguiar A, Eubig PA, Schantz SL (2010) Attention deficit/hyperactivity disorder: a focused overview for children’s environmental health researchers. Environ Health Perspect 118(12):1646–1653. https://doi.org/10.1289/ehp.1002326
Costa LG, Aschner M, Vitalone A, Syversen T, Soldin OP (2004) Developmental neuropathology of environmental agents. Annu Rev Pharmacol Toxicol 44:87–110. https://doi.org/10.1146/annurev.pharmtox.44.101802.121424
Cory-Slechta DA (1995) Relationships between lead-induced learning impairments and changes in dopaminergic, cholinergic, and glutamatergic neurotransmitter system functions. Annu Rev Pharmacol Toxicol 35(1):391–415. https://doi.org/10.1146/annurev.pa.35.040195.002135
Godwin HA (2001) The biological chemistry of lead. Curr Opin Chem Biol 5(2):223–227. https://doi.org/10.1016/S1367-5931(00)00194-0
Flora GJ, Seth PK (2000) Alterations in some membrane properties in rat brain following exposure to lead. Cytobios 103(403):103–109
Luo M, Xu Y, Cai R, Tang Y, Ge M-M, Liu Z-H, Xu L, Hu F, Ruan D-Y, Wang H-L (2014) Epigenetic histone modification regulates developmental lead exposure induced hyperactivity in rats. Toxicol Lett 225(1):78–85. https://doi.org/10.1016/j.toxlet.2013.11.025
Acknowledgements
We are sincerely grateful to the participating families for their cooperation. We thank Dr. Jun Lin from Wuhan Medical and Health Center for Women and Children, and Dr. Yan Zhong from Children’s Hospital of Hunan province for their widespread support for our study and sharing their knowledge. This work was supported partially by the National Natural Science Foundation of China (81773456), the Fundamental Research Funds for the Central Universities, HUST (2016 YXMS218) supported Dr. Jing Wu.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments. Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, X., Zhang, Q., Gu, X. et al. LPHN3 gene variations and susceptibility to ADHD in Chinese Han population: a two-stage case–control association study and gene–environment interactions. Eur Child Adolesc Psychiatry 28, 861–873 (2019). https://doi.org/10.1007/s00787-018-1251-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-018-1251-8