Abstract
Objectives
Hydrophilic implant surfaces promote faster osseointegration of dental implants with a higher bone-implant contact (BIC) rate. Animal and in vitro studies proved that ultraviolet (UV) irradiation of titanium implants regains hydrophilicity. Clinical impact is still unclear. The objective of this RCT was to assess the removal torque (RT) required to unfix a surface-treated implant (test group) versus the original surface implant (control group) performed at various points in time. The null hypothesis stated that test and control implants will show the same deliberation force at specific time points.
Material and methods
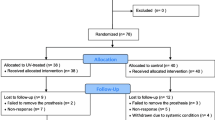
One hundred eighty partially edentulous patients were randomly assigned to six groups. In single-stage surgery, each patient received one test and one control implant. In total, 180 test and 180 control implants were placed epicrestally. Test implants received a surface treatment with UV irradiation prior to insertion, in order to reduce carbon and enhance hydrophilicity and thus wettability. Maximum RT values for test and control implants were recorded with a torque measuring device at implant placement (T1), after 1 (group 1), 2 (group 2), 3 (group 3), 4 (group 4), 6 (group 5) (T2), and 8 weeks (group 6) of healing. Subsequently, implants were returned to their original position for the continuation of the healing process.
Results
No implant was lost. Age, gender, smoking, implant position, and bone quality could be excluded as confounding factors because of the lack of statistical significance. At T2, RT values were higher for test implants compared with those for control implants, being statistically significant in groups 2, 3, 4, and 6 (p < 0.05).
Conclusions
Our data support rejection of the null hypothesis.
Clinical relevance
Photo-activation of the surface of titanium implants leads to higher resistance to RT forces compared with that of non-treated implants, indicating improved healing and implant stability especially in the early healing phase.




Similar content being viewed by others
References
Aita H, Hori N, Takeuchi M, Suzuki T, Yamada M, Anpo M, Ogawa T (2009) The effect of ultraviolet functionalization of titanium on integration with bone. Biomaterials 30(6):1015–1025
Albrektsson T, Branemark PI, Hansson HA et al (1981) Osseointegrated titanium implants. Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop Scand 52(2):155–170
Albrektsson T (2008) Hard tissue implant interface. Aust Dent J 53(Suppl 1):S34–S38
Albrektsson T, Buser D, Chen ST, Cochran D, DeBruyn H, Jemt T, Koka S, Nevins M, Sennerby L, Simion M, Taylor TD, Wennerberg A (2012) Statements from the Estepona consensus meeting on peri-implantitis, February 2-4, 2012. Clin Implant Dent Relat Res 14(6):781–782
Albrektsson T, Dahlin C, Jemt T, Sennerby L, Turri A, Wennerberg A (2014) Is marginal bone loss around oral implants the result of a provoked foreign body reaction? Clin Implant Dent Relat Res 16(2):155–165
Albrektsson T, Canullo L, Cochran D, de Bruyn H (2016) “Peri-implantitis”: a complication of a foreign body or a man-made “disease”. Facts and fiction. Clin Implant Dent Relat Res 18(4):840–849
Atsumi M, Park S-H, Wang H-L (2007) Methods used to assess implant stability: current status. Int J Oral Maxillofac Implants 22(5):743–754
Att W, Hori N, Takeuchi M, Ouyang J, Yang Y, Anpo M, Ogawa T (2009) Time-dependent degradation of titanium osteoconductivity: an implication of biological aging of implant materials. Biomaterials 30(29):5352–5363
Branemark PI, Zarb GA, Albrektsson T (1985) Tissue-integrated prostheses: osseointegration in clinical. Quintessence, Chicago [u. a]
Buser D, Nydegger T, Hirt HP, Cochran DL, Nolte LP (1998) Removal torque values of titanium implants in the maxilla of miniature pigs. Int J Oral Maxillofac Implants 13(5):611–619
Buser D, Nydegger T, Oxland T, Cochran DL, Schenk RK, Hirt HP, Snétivy D, Nolte LP (1999) Interface shear strength of titanium implants with a sandblasted and acid-etched surface: a biomechanical study in the maxilla of miniature pigs. J Biomed Mater Res 45(2):75–83
Buser D, Broggini N, Wieland M, Schenk RK, Denzer AJ, Cochran DL, Hoffmann B, Lussi A, Steinemann SG (2004) Enhanced bone apposition to a chemically modified SLA titanium surface. J Dent Res 83(7):529–533
Chowdhary R, Chowdhary N, Mishra SK (2011) Re-osseointegration of loosened implant in a splinted fixed prosthesis. Niger J Clin Pract 14(1):102–105
Donath K, Laass M, Gunzl HJ (1992) The histopathology of different foreign-body reactions in oral soft tissue and bone tissue. Virchows Arch A Pathol Anat Histopathol 420(2):131–137
Esposito M, Ardebili Y, Worthington H (2014) Interventions for replacing missing teeth: different types of dental implants. Cochrane Database Syst Rev 22(7):CD003815. https://doi.org/10.1002/14651858.CD003815.pub4
Faeda RS, Tavares HS, Sartori R, Guastaldi AC, Marcantonio E Jr (2009) Biological performance of chemical hydroxyapatite coating associated with implant surface modification by laser beam: biomechanical study in rabbit tibias. J Oral Maxillofac Surg 67(8):1706–1715
Ferguson SJ, Broggini N, Wieland M et al (2006) Biomechanical evaluation of the interfacial strength of a chemically modified sandblasted and acid-etched titanium surface. J Biomed Mater Res A 78(2):291–297
Franchi M, Fini M, Martini D, Orsini E, Leonardi L, Ruggeri A, Giavaresi G, Ottani V (2005) Biological fixation of endosseous implants. Micron 36(7–8):665–671
Funato A, Yamada M, Ogawa T (2013) Success rate, healing time, and implant stability of photofunctionalized dental implants. Int J Oral Maxillofac Implants 28(5):1261–1271
Gottlow J, Dard M, Kjellson F, Obrecht M, Sennerby L (2012) Evaluation of a new titanium-zirconium dental implant: a biomechanical and histological comparative study in the mini pig. Clin Implant Dent Relat Res 14(4):538–545
He F, Yang G, Wang X et al (2009) Effect of electrochemically deposited nanohydroxyapatite on bone bonding of sandblasted/dual acid-etched titanium implant. Int J Oral Maxillofac Implants 24(5):790–799
Hyzy SL, Cheng A, Cohen DJ, Yatzkaier G, Whitehead AJ, Clohessy RM, Gittens RA, Boyan BD, Schwartz Z (2016) Novel hydrophilic nanostructured microtexture on direct metal laser sintered Ti-6Al-4V surfaces enhances osteoblast response in vitro and osseointegration in a rabbit model. J Biomed Mater Res A 104(8):2086–2098
Ivanoff CJ, Sennerby L, Lekholm U (1997) Reintegration of mobilized titanium implants. An experimental study in rabbit tibia. Int J Oral Maxillofac Surg 26(4):310–315
Jividen G Jr, Misch CE (2000) Reverse torque testing and early loading failures: help or hindrance? J Oral Implantol 26(2):82–90
Krafft T, Graef F, Karl M (2015) Osstell resonance frequency measurement values as a prognostic factor in implant dentistry. J Oral Implantol 41(4):e133–e137
Lang NP, Salvi GE, Huynh-Ba G, Ivanovski S, Donos N, Bosshardt DD (2011) Early osseointegration to hydrophilic and hydrophobic implant surfaces in humans. Clin Oral Implants Res 22(4):349–356
Lee J-T, Cho S-A (2016) Biomechanical evaluation of laser-etched Ti implant surfaces vs. chemically modified SLA Ti implant surfaces: removal torque and resonance frequency analysis in rabbit tibias. J Mech Behav Biomed Mater 61:299–307
Lucente J, Galante J, Trisi P, Kenealy JN (2006) Reintegration success of osseotite implants after intentional countertorque liberation in the endentulous human mandible. Implant Dent 15(2):178–185
Manresa C, Bosch M, Echeverria JJ (2014) The comparison between implant stability quotient and bone-implant contact revisited: an experiment in Beagle dog. Clin Oral Implants Res 25(11):1213–1221
Meredith N (1998) Assessment of implant stability as a prognostic determinant. Int J Prosthodont 11(5):491–501
Ogawa T (2014) Ultraviolet photofunctionalization of titanium implants. Int J Oral Maxillofac Implants 29(1): e95–102
Raghavendra S, Wood MC, Taylor TD (2005) Early wound healing around endosseous implants: a review of the literature. Int J Oral Maxillofac Implants 20(3):425–431
Sartoretto S C, Alves A T N N, Zarranz L et al (2016) Hydrophilic surface of Ti6Al4V-ELI alloy improves the early bone apposition of sheep tibia. Clin Oral Implants Res
Schlee M, van der Schoor WP, van der Schoor ARM (2015) Immediate loading of trabecular metal-enhanced titanium dental implants: interim results from an international proof-of-principle study. Clin Implant Dent Relat Res 17(Suppl 1):e308–e320
Schwartz Z, Boyan BD (1994) Underlying mechanisms at the bone-biomaterial interface. J Cell Biochem 56(3):340–347
Schwarz F, Wieland M, Schwartz Z et al (2009) Potential of chemically modified hydrophilic surface characteristics to support tissue integration of titanium dental implants. J Biomed Mater Res B Appl Biomater 88(2):544–557
Simeone SG, Rios M, Simonpietri J (2016) Reverse torque of 30 Ncm applied to dental implants as test for osseointegration-a human observational study. Int J Implant Dent 2(1):26
Smeets R, Stadlinger B, Schwarz F et al (2016) Impact of dental implant surface modifications on osseointegration. Biomed Res Int 2016:6285620
Sullivan DY, Sherwood RL, Collins TA et al (1996) The reverse-torque test: a clinical report. Int J Oral Maxillofac Implants 11(2):179–185
Suzuki T, Hori N, Att W, Kubo K, Iwasa F, Ueno T, Maeda H, Ogawa T (2009) Ultraviolet treatment overcomes time-related degrading bioactivity of titanium. Tissue Eng A 15(12):3679–3688
Tjellstrom A, Jacobsson M, Albrektsson T (1988) Removal torque of osseointegrated craniofacial implants: a clinical study. Int J Oral Maxillofac Implants 3(4):287–289
Verborgt O, Gibson GJ, Schaffler MB (2000) Loss of osteocyte integrity in association with microdamage and bone remodeling after fatigue in vivo. J Bone Miner Res 15(1):60–67
Wang R, Hashimoto K, Fujishima A, Chikuni M, Kojima E, Kitamura A, Shimohigoshi M, Watanabe T (1997) Light-induced amphiphilic surfaces. Nature 388(6641):431–432
Acknowledgments
The authors thank Dipl. Math. Ulrike von Hehn for her statistical support.
Funding
The study was supported by a grant from Ushio. The implants were provided by BioHorizons. W+H provided the calibrated drilling device.
Author information
Authors and Affiliations
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Algirdas Puisys and Markus Schlee shared first authorship.
Rights and permissions
About this article
Cite this article
Puisys, A., Schlee, M., Linkevicius, T. et al. Photo-activated implants: a triple-blinded, split-mouth, randomized controlled clinical trial on the resistance to removal torque at various healing intervals. Clin Oral Invest 24, 1789–1799 (2020). https://doi.org/10.1007/s00784-019-03041-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-03041-5