Abstract
Objectives
To assess the association between oral mucosa hyperpigmentation in patients with leukemia and imatinib mesylate use. Additionally, we compared our data to those obtained from a systematic review.
Materials and methods
A cross-sectional study was conducted with 74 patients undergoing treatment with imatinib mesylate. Sociodemographic characteristics, oral mucosa alterations, and medical history were evaluated. Oral hyperpigmentation was scored. The use of imatinib mesylate and hydroxyurea was evaluated. Association between oral hyperpigmentation and imatinib mesylate was assessed. A systematic review was also conducted to retrieve case reports or case series of patients with oral hyperpigmentation associated with imatinib mesylate.
Results
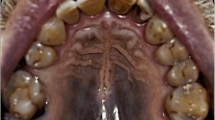
Among the 74 participants, 41 were male (55.4%) and 33 were female (44.6%). Participants’ mean age was 49.3 years. Sixty-six (89.2%) patients developed hyperpigmented lesions in the hard palate mucosa. In multivariate analysis, patients who had used imatinib mesylate for > 72 months had a hyperpigmentation score 1.62 times higher than those who had used this medication during a shorter period. Patients who had used hydroxyurea for > 30 days had a hyperpigmentation score 1.43 times higher than those who had used this medication during a shorter period. The systematic review retrieved 20 clinical cases of patients undergoing imatinib mesylate treatment and exhibiting oral hyperpigmentation.
Conclusions
The development of oral hyperpigmentation is associated with imatinib mesylate use. Hydroxyurea seems to increment such an association.
Clinical relevance
To assist providers in the differential diagnosis of hyperpigmented lesions associated with imatinib mesylate, as well as in the clinical management of such lesions.



Similar content being viewed by others
References
Xie Y, Davies SM, Xiang Y, Robison LL, Ross JA (2003) Trends in leukemia incidence and survival in the United States (1973–1998). Cancer 97:2229–2235
Jemal A, Tiwari RC, Murray T, Ghafoor A, Samuels A, Ward E, Feuer EJ, Thun MJ (2004) American Cancer Society, Cancer statistics. CA Cancer J Clin 54:8–29
Nowell PC, Hungerford DA (1960) Chromosome studies on normal and leukemic human leukocytes. J Natl Cancer Inst 25:85–109
Rowley JD (1973) A new consistent chromosomal abnormality in chronic myelogenous leukaemia identified by quinacrine fluorescence and Giemsa staining. Nature 243:290–293
Shtivelman E, Lifshitz B, Gale RP, Canaani E (1985) Fused transcript of abl and bcr genes in chronic myelogenous leukaemia. Nature 315:550–554
Abramson S, Miller RG, Phillips RA (1977) The identification in adult bone marrow of pluripotent and restricted stem cells of the myeloid and lymphoid systems. J Exp Med 145:1567–1579
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 66:271–289
Faderl S, Kantarjian HM, Talpaz M, Estrov Z (1998) Clinical significance of cytogenetic abnormalities in adult acute lymphoblastic leukemia. Blood 91:3995–4019
Schlieben S, Borkhardt A, Reinisch I, Ritterbach J, Janssen JW, Ratei R, Schrappe M, Repp R, Zimmermann M, Kabisch H, Janka-Schaub G, Bartram CR, Ludwig WD, Riehm H, Lampert F, Harbott J (1996) Incidence and clinical outcome of children with BCR/ABL-positive acute lymphoblastic leukemia (ALL). A prospective RT-PCR study based on 673 patients enrolled in the German pediatric multicenter therapy trials ALL-BFM-90 and CoALL-05-92. Leukemia 10:957–963
Hehlmann R, Berger U, Pfirrmann M et al (2003) Randomized comparison of interferon α and hydroxyurea with hydroxyurea monotherapy in chronic myeloid leukemia (CML-study II): prolongation of survival by the combination of interferon α and hydroxyurea. Leukemia 17:1529–1537
Henkes M, van der Kuip H, Aulitzky WE (2008) Therapeutic options for chronic myeloid leukemia: focus on imatinib (Glivec®, Gleevec™). Ther Clin Risk Manag 4:163
Kumar B, Saraswat A, Kaur I (2002) Mucocutaneous adverse effects of hydroxyurea: a prospective study of 30 psoriasis patients. Clin Exp Dermatol 27:8–13
Savage DG, Antman KH (2002) Imatinib mesylate—a new oral targeted therapy. New Engl J Med 346:683–693
Payandeh M, Sadeghi M, Sadeghi E (2015) Treatment and survival in patients with chronic myeloid leukemia in a chronic phase in West Iran. Asian Pac J Cancer Prev 16:7555–7559
Thomas DA, Faderl S, Cortes J (2004) Treatment of Philadelphia chromosome–positive acute lymphocytic leukemia with hyper-CVAD and imatinib mesylate. Blood 103:4396–4407
Piccaluga PP, Paolini S, Martinelli G (2007) Tyrosine kinase inhibitors for the treatment of Philadelphia chromosome-positive adult acute lymphoblastic leukemia. Cancer 110:1178–1186
Cohen MH, Williams G, Johnson JR, Duan J, Gobburu J, Rahman A, Benson K, Leighton J, Kim SK, Wood R, Rothmann M, Chen G, U K, Staten AM, Pazdur R (2002) Approval summary for imatinib mesylate capsules in the treatment of chronic myelogenous leukemia. Clin Cancer Res 8:935–942
Deininger MW, O’brien SG, Ford JM, Druker BJ (2003) Practical management of patients with chronic myeloid leukemia receiving imatinib. J Clin Oncol 21:1637–1647
Lim DS, Muir J (2002) Oral lichenoid reaction to imatinib (STI 571, Gleevec). Dermatology 205:169–171
Ena P, Chiarolini F, Siddi GM, Cossu A (2004) Oral lichenoid eruption secondary to imatinib (Glivec®). J Dermatol Treat 15:253–255
Pascual JC, Matarredona J, Miralles J, Conesa V, Borras-Blasco J (2006) Oral and cutaneous lichenoid reaction secondary to imatinib: report of two cases. Int J Dermatol 45:1471–1473
Mattsson U, Halbritter S, Serikoff EM, Christerson L, Warfvinge G (2011) Oral pigmentation in the hard palate associated with imatinib mesylate therapy: a report of three cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 111:12–16
Wong M, Sade S, Gilbert M, Klieb HB (2011) Oral melanosis after tyrosine kinase inhibition with imatinib for chronic myelogenous leukemia: report of a case and review of the literature. Dermatol Online J 17:4
Li CC, Malik SM, Blaeser BF, Dehni WJ, Kabani SP, Boyle N, Toner M, Woo SB (2012) Mucosal pigmentation caused by imatinib: report of three cases. Head Neck Pathol 6:290–295
Khoo TL, Catalano A, Supple S, Chong L, Yeoh SC, Yeung S, Iland H (2013) Hyperpigmentation of the hard palate associated with imatinib therapy for chronic myeloid leukemia with a genetic variation in the proto-oncogene c-KIT. Leuk Lymphoma 54:186–188
Roeker LE, Wolanskyj AP (2014) Imatinib-associated melanosis of the palate. Am J Hematol 89:564–564
Tosios KI, Kalogirou EM, Sklavounou A (2018) Drug-associated hyperpigmentation of the oral mucosa: report of 4 cases. Oral Surg Oral Med Oral Pathol Oral Radiol 125:54–66
Singh N, Bakhshi S (2007) Imatinib-induced dental hyperpigmentation in childhood chronic myeloid leukemia. J Pediatr Hematol Oncol 29:208–209
Mcpherson T, Sherman V, Turner R (2009) Imatinib-associated hyperpigmentation, a side effect that should be recognized. J Eur Acad Dermatol Venereol 23:82–83
Resende RG, Teixeira RGL, de Oliveira Vasconcelos F, de Souza Silva ME, Abreu MHG, Gomez RS (2012) Imatinib-associated hyperpigmentation of the palate in post-HSCT patient. J Cranio-Maxillofac Surg 40:140–143
Steele JC, Triantafyllou A, Rajlawat BP, Field EA (2012) Oral mucosal hyperpigmentation and horizontal melanonychia caused by imatinib. Clin Exp Dermatol 37:432–433
López-Labady J, Villarroel-Dorrego M, Bascones Martínez A (2013) Pigmentación inusual del paladar asociada a imatinib: reporte de caso clínico. Av Odontoestomatol 29:309–314
Song HS, Kang HY (2014) Imatinib mesylate-induced hyperpigmentation of the nose and palate. Ann Dermatol 26:532–533
Lyne A, Creedon A, Bailey BM (2015) Mucosal pigmentation of the hard palate in a patient taking imatinib. BMJ Case Rep pii: bcr2015209335
Romeo U, Palaia G, Fantozzi PJ, Tenore G, Bosco D (2015) A rare case of melanosis of the hard palate mucosa in a patient with chronic myeloid leukemia. Case Rep Dent 817094
Bombeccari GP, Garagiola U, Pallotti F, Rossi M, Porrini M, Giannì AB, Spadari F (2017) Hyperpigmentation of the hard palate mucosa in a patient with chronic myeloid leukaemia taking imatinib. Maxillofac Plast Reconstr Surg 39:37
Di Tullio F, Mandel VD, Scotti R, Padalino C, Pellacani G (2018) Imatinib-induced diffuse hyperpigmentation of the oral mucosa, the skin, and the nails in a patient affected by chronic myeloid leukemia: report of a case and review of the literature. Int J Dermatol 57: 784–790
Torres-Pereira CC (2011) Are we ready to blame imatinib for palatal pigmentation? Oral Surg Oral Med Oral Pathol Oral Radiol Endod 112:287
American Cancer Society (2016) Cancer treatment and survivorship facts & figures 2016–2017. American Cancer Society, Atlanta
Redaelli A, Bell C, Casagrande J, Stephens J, Botteman M, Laskin B, Pashos C (2004) Clinical and epidemiologic burden of chronic myelogenous leukemia. Expert Rev Anticancer Ther 4:85–96
Rix U, Hantschel O, Dürnberger G et al (2007) Chemical proteomic profiles of the BCR-ABL inhibitors imatinib, nilotinib, and dasatinib reveal novel kinase and nonkinase targets. Blood 110:4055–4063
Goldman JM, Melo JV (2001) Targeting the BCR-ABL tyrosine kinase in chronic myeloid leukemia. New Engl J Med 344:1084–1086
Gambacorti-Passerini C, Antolini L, Mahon FX, Guilhot F, Deininger M, Fava C, Nagler A, Della Casa CM, Morra E, Abruzzese E, D'Emilio A, Stagno F, le Coutre P, Hurtado-Monroy R, Santini V, Martino B, Pane F, Piccin A, Giraldo P, Assouline S, Durosinmi MA, Leeksma O, Pogliani EM, Puttini M, Jang E, Reiffers J, Piazza R, Valsecchi MG, Kim DW (2011) Multicenter independent assessment of outcomes in chronic myeloid leukemia patients treated with imatinib. J Natl Cancer Inst 103:553–561
Jain P, Das VNR, Ranjan A, Chaudhary R, Pandey K (2013) Comparative study for the efficacy, safety and quality of life in patients of chronic myeloid leukemia treated with Imatinib or hydroxyurea. J Res Pharm Pract 2:156–161
Airiau K, Turcq B, Mahon FX, Belloc F (2017) A new mechanism of resistance to ABL1 tyrosine kinase inhibitors in a BCR-ABL1-positive cell line. Leuk Res 61:44–52
Miettinen M, Lasota J (2005) KIT (CD117): a review on expression in normal and neoplastic tissues, and mutations and their clinicopathologic correlation. Appl Immunohistochem Mol Morphol 13:205–220
Jeon S, Kim NH, Kim JY, Lee AY (2009) Stem cell factor induces ERM proteins phosphorylation through PI3K activation to mediate melanocyte proliferation and migration. Pigment Cell Melanoma Res 22:77–85
Wehrle-Haller B (2003) The role of Kit-ligand in melanocyte development and epidermal homeostasis. Pigment Cell Melanoma Res 16:287–296
Cario-André M, Ardilouze L, Pain C, Gauthier Y, Mahon FX, Taieb A (2006) Imatinib mesilate inhibits melanogenesis in vitro. Br J Dermatol 155:493–494
Buchdunger E, Cioffi CL, Law N, Stover D, Ohno-Jones S, Druker BJ, Lydon NB (2000) Abl protein-tyrosine kinase inhibitor STI571 inhibits in vitro signal transduction mediated by c-kit and platelet-derived growth factor receptors. J Pharmacol Exp Ther 295:139–145
Arora B, Kumar L, Sharma A, Wadhwa J, Kochupillai V (2004) Pigmentary changes in chronic myeloid leukemia patients treated with imatinib mesylate. Ann Oncol 15:358–359
Robson ME, Glogowski E, Sommer G, Antonescu CR, Nafa K, Maki RG, Ellis N, Besmer P, Brennan M, Offit K (2004) Pleomorphic characteristics of a germ-line KIT mutation in a large kindred with gastrointestinal stromal tumors, hyperpigmentation, and dysphagia. Clin Cancer Res 10:1250–1254
Alexandrescu DT, Dasanu CA, Farzanmehr H, Kauffman CL (2008) Persistent cutaneous hyperpigmentation after tyrosine kinase inhibition with imatinib for GIST. Dermatol Online J 14:7
Lerman MA, Karimbux N, Guze KA, Woo SB (2009) Pigmentation of the hard palate. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 107:8–12
de Andrade BAB, Fonseca FP, Pires FR, Mesquita ATM, Falci SGM, dos Santos Silva AR, Vargas PA, Jorge J, de Almeida OP (2013) Hard palate hyperpigmentation secondary to chronic chloroquine therapy: report of five cases. J Cutan Pathol 40:833–838
Meleti M, Vescovi P, Mooi WJ, van der Waal I (2008) Pigmented lesions of the oral mucosa and perioral tissues: a flow-chart for the diagnosis and some recommendations for the management. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 105:606–616
Gondak RO, da Silva-Jorge R, Jorge J, Lopes MA, Vargas PA (2012) Oral pigmented lesions: clinicopathologic features and review of the literature. Med Oral Patol Oral Cir Bucal 17:919
Eisen D (2000) Disorders of pigmentation in the oral cavity. Clin Dermatol 18:579–587
Kauzman ABDS, Pavone M, Blanas N, Bradley G (2004) Pigmented lesions of the oral cavity: review, differential diagnosis, and case presentations. J Can Dent Assoc 70:682–683
Axeix T, HEDIN CA (1982) Epidemiologic study of excessive oral melanin pigmentation with special reference to the influence of tobacco habits. Eur J Oral Sci 90:434–442
Lenane P, Powell FC (2000) Oral pigmentation. J Eur Acad Dermatol Venereol 14:448–465
Kleinegger CL, Hammond HL, Finkelstein MW (2000) Oral mucosal hyperpigmentation secondary to antimalarial drug therapy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 90:189–194
Kim HW (1988) Generalized oral and cutaneous hyperpigmentation in Addison's disease. Odontostomatol Trop= Trop Dent J 11:87–90
Machaczka M, Gossart M (2013) Multiple skin lesions caused by imatinib mesylate treatment of chronic myeloid leukemia. Pol Arch Med Wewn 123:251–252
Dervis E, Ayer M, Belli AA, Barut SG (2016) Cutaneous adverse reactions of imatinib therapy in patients with chronic myeloid leukemia: a six-year follow up. Eur J Dermatol 26:133–137
Funding
This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES-Finance code 001). RAM and TAS are research fellows of CNPq.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oliveira, S.R., de Azevedo Branco, L.G., Rocha, A.L. et al. Association of oral mucosa hyperpigmentation with imatinib mesylate use: a cross-sectional study and a systematic literature review. Clin Oral Invest 23, 4371–4382 (2019). https://doi.org/10.1007/s00784-019-02886-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-02886-0