Abstract
Objectives
To evaluate the correlation between dental plaque formation and gingival health in subjects performing high oral hygiene standards over short or extended intervals.
Materials and methods
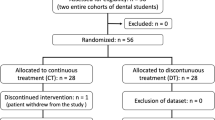
Fifty-two non-dental students volunteered for this study. The subjects, trained to perform high oral hygiene standards, were randomized to perform oral hygiene at 12-, 24-, 48-, or 72-h interval over 30 days. The plaque index (PlI) and the gingival index (GI) were evaluated at baseline, 15, and 30 days. For the statistical analysis, oral hygiene intervals were collapsed into daily (12 and 24 h; G12/24) and extended (48 and 72 h; G48/72) intervals. Summary statistics (mean ± SD) and Spearman correlations between the PlI and the GI at baseline, 15, and 30 days were estimated.
Results
At baseline, correlation coefficients between PlI and GI were positive for both groups (r = 0.29 and r = 0.25). At day 15 and 30, correlation was maintained with similar baseline values for the G48/72 group. GI levels did not increase despite an increase in PlI for the G12/24 group, and the correlation was lower than that observed at baseline (r = 0.13 vs. r = 0.29).
Conclusions
In subjects with high oral hygiene standards, the oral hygiene frequency governs the correlation between dental plaque formation and gingival health. Subjects performing high oral hygiene standards at daily intervals will maintain gingival health in difference to subjects using extended hygiene intervals.
Clinical relevance
Subjects performing high oral hygiene standards at daily intervals will maintain gingival health in difference to subjects using extended hygiene intervals.

Similar content being viewed by others
References
Mariotti A (1999) Dental plaque-induced gingival diseases. Ann Periodontol 4(1):7–19. https://doi.org/10.1902/annals.1999.4.1.7
Albandar JM (2002) Periodontal diseases in North America. Periodontol 29(1):31–69. https://doi.org/10.1034/j.1600-0757.2002.290103.x
Ababneh KT, Abu Hwaij ZM, Khader YS (2012) Prevalence and risk indicators of gingivitis and periodontitis in a multi-centre study in North Jordan: a cross sectional study. BMC Oral Health 12(1):1–8. https://doi.org/10.1186/1472-6831-12-1
Chiapinotto FA, Vargas-Ferreira F, Demarco FF, Correa FO, Masotti AS (2013) Risk factors for gingivitis in a group of Brazilian schoolchildren. J Public Health Dent 73(1):9–17. https://doi.org/10.1111/jphd.12001
Idrees MM, Azzeghaiby SN, Hammad MM, Kujan OB (2014) Prevalence and severity of plaque-induced gingivitis in a Saudi adult population. Saudi Med J 35(11):1373–1377
Oppermann RV, Haas AN, Rosing CK, Susin C (2015) Epidemiology of periodontal diseases in adults from Latin America. Periodontol 67(1):13–33. https://doi.org/10.1111/prd.12061
Schätzle M, Löe H, Burgin W, Anerud A, Boysen H, Lang NP (2003) Clinical course of chronic periodontitis. I. Role of gingivitis. J Clin Periodontol 30(10):887–901. https://doi.org/10.1034/j.1600-051X.2003.00414.x
Pham TA, Ueno M, Shinada K, Kawaguchi Y (2012) Factors affecting oral malodor in periodontitis and gingivitis patients. J Investig Clin Dent 3(4):284–290. https://doi.org/10.1111/j.2041-1626.2012.00155.x
Tomazoni F, Zanatta FB, Tuchtenhagen S, da Rosa GN, del Fabro JP, Ardenghi TM (2014) Association of gingivitis with child oral health-related quality of life. J Periodontol 85(11):1557–1565. https://doi.org/10.1902/jop.2014.140026
Löe H, Theilade E, Jensen SB (1965) Experimental gingivitis in man. J Periodontol 36(3):177–187. https://doi.org/10.1902/jop.1965.36.3.177
Breuer MM, Cosgrove RS (1989) The relationship between gingivitis and plaque levels. J Periodontol 60(4):172–175. https://doi.org/10.1902/jop.1989.60.4.172
Lie MA, Timmerman MF, van der Velden U, van der Weijden GA (1998) Evaluation of 2 methods to assess gingival bleeding in smokers and non-smokers in natural and experimental gingivitis. J Clin Periodontol 25(9):695–700. https://doi.org/10.1111/j.1600-051X.1998.tb02509.x
Oliveira SC, Slot DE, Celeste RK, Abegg C, Keijser BJ, van der Weijden FA (2015) Correlations between two different methods to score bleeding and the relationship with plaque in systemically healthy young adults. J Clin Periodontol 42(10):908–913. https://doi.org/10.1111/jcpe.12435
Kistler JO, Booth V, Bradshaw DJ, Wade WG (2013) Bacterial community development in experimental gingivitis. PLoS One 8(8):e71227. https://doi.org/10.1371/journal.pone.0071227
Claydon NC (2008) Current concepts in toothbrushing and interdental cleaning. Periodontol 48(1):10–22. https://doi.org/10.1111/j.1600-0757.2008.00273.x
Pinto TM, de Freitas GC, Dutra DA, Kantorski KZ, Moreira CH (2013) Frequency of mechanical removal of plaque as it relates to gingival inflammation: a randomized clinical trial. J Clin Periodontol 40(10):948–954. https://doi.org/10.1111/jcpe.12135
de Freitas GC, Pinto TM, Grellmann AP, Dutra DA, Susin C, Kantorski KZ, Moreira CH (2016) Effect of self-performed mechanical plaque control frequency on gingival inflammation revisited: a randomized clinical trial. J Clin Periodontol 43(4):354–358. https://doi.org/10.1111/jcpe.12520
Theilade E, Wright WH, Jensen SB, Löe H (1966) Experimental gingivitis in man. II. A longitudinal clinical and bacteriological investigation. J Periodontal Res 1(1):1–13. https://doi.org/10.1111/j.1600-0765.1966.tb01842.x
Löe H (1967) The gingival index, the plaque index and the retention index systems. J Periodontol 38(Suppl):610–616. https://doi.org/10.1902/jop.1967.38.6_part2.610
Silness J, Löe H (1964) Periodontal disease in pregnancy. II. Correlation between oral hygiene and periodontal condition. Acta Odontol Scand 22(1):121–135. https://doi.org/10.3109/00016356408993968
Haffajee AD, Teles RP, Patel MR, Song X, Veiga N, Socransky SS (2009) Factors affecting human supragingival biofilm composition. I. Plaque mass. J Periodontal Res 44(4):511–519. https://doi.org/10.1111/j.1600-0765.2008.01154.x
Heller D, Helmerhorst EJ, Gower AC, Siqueira WL, Paster BJ, Oppenheim FG (2016) Microbial diversity in the early in vivo-formed dental biofilm. Appl Environ Microbiol 82(6):1881–1888. https://doi.org/10.1128/AEM.03984-15
Teles FR, Teles RP, Uzel NG, Song XQ, Torresyap G, Socransky SS, Haffajee AD (2012) Early microbial succession in redeveloping dental biofilms in periodontal health and disease. J Periodontal Res 47(1):95–104. https://doi.org/10.1111/j.1600-0765.2011.01409.x
Acknowledgments
The authors would like to thank Alessandra Pascotini Grellmann, MSc, and Danilo Antônio Milbrat Dutra, MSc, for collaborating in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest.
Ethical approval
This study was performed in accordance with the Declaration of Helsinki and was approved by the Ethics Committee in Research of Federal University of Santa Maria. (CAAE: 0186.0.243.00-10).
Informed consent
After an explanation of the purpose of study and any questions answered by one of the investigators, subjects agreeing to participate in this study signed an informed consent.
Rights and permissions
About this article
Cite this article
De David, S.C., Mário, T.G., De Freitas, G.C. et al. Correlation between plaque control and gingival health using short and extended oral hygiene intervals. Clin Oral Invest 22, 2593–2597 (2018). https://doi.org/10.1007/s00784-018-2358-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-018-2358-5