Abstract
Objectives
To assess the clinical and radiographic outcomes of the regenerative treatment of peri-implantitis using a vancomycin and tobramycin impregnated allograft (VTA) after a 12-month period.
Material and methods
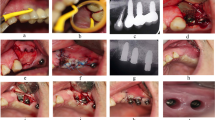
Thirteen consecutive patients who required a regenerative treatment of peri-implantitis were recruited. For the 17 implant sites, a flap was raised, and after mechanical and chemical implant decontamination, a vancomycin and tobramycin impregnated allograft was placed in the defect and then covered with a collagen membrane. Soft tissues were sutured allowing a non-submerged healing. Clinical and radiographic variables were evaluated at baseline and at 12 months after treatment.
Results
No signs of continuous bone loss were observed and no implant was lost, yielding a 100% survival rate. All patient’s clinical examination at 12 months revealed peri-implant health showing absence of suppuration and a statistically significant reduction in terms of bleeding on probing scores (70.6%, P = 0.001). Initial probing pocket depth (7.88 ± 1.22 mm) was significantly reduced at 12 months healing, a mean reduction of 4.23 ± 1.47 mm (P = 0.001) was achieved. The mean radiological infrabony defect at baseline reached 4.33 ± 1.62 mm, and was significantly reduced up to 0.56 ± 0.88 mm, which represents an 86.99 ± 18.2% bone fill from the original infrabony defect.
Conclusions
Within the limits of the study, the application of VTA with a collagen membrane yielded positive outcomes in terms of radiographic bone fill, pocket depth reduction, and attachment gain after a 12-month period. Thus, VTA plus a collagen membrane seem to be suitable for the regenerative treatment of peri-implantitis.
Clinical relevance
The use of locally delivered antibiotic together with the bone graft may reduce the undesirable effects related to the systemic administration and the risk of resistances. In the light of the results obtained, these grafting materials might offer new treatment strategies in the surgical regenerative treatment of peri-implantitis.

Similar content being viewed by others
References
Lindhe J, Meyle J and Group DoEWoP (2008) Peri-implant diseases: consensus report of the sixth European workshop on periodontology. J Clin Periodontol 35(8 Suppl):282–285. https://doi.org/10.1111/j.1600-051X.2008.01283.x
Serino G, Turri A, Lang NP (2013) Probing at implants with peri-implantitis and its relation to clinical peri-implant bone loss. Clin Oral Implants Res 24(1):91–95. https://doi.org/10.1111/j.1600-0501.2012.02470.x
Derks J, Tomasi C (2015) Peri-implant health and disease. A systematic review of current epidemiology. J Clin Periodontol 42 Suppl 16:S158–S171. https://doi.org/10.1111/jcpe.12334
Marotti J, Tortamano P, Cai S, Ribeiro MS, Franco JE, de Campos TT (2013) Decontamination of dental implant surfaces by means of photodynamic therapy. Lasers Med Sci 28(1):303–309. https://doi.org/10.1007/s10103-012-1148-6
Heitz-Mayfield LJ, Salvi GE, Mombelli A, Faddy M, Lang NP, Implant Complication Research G (2012) Anti-infective surgical therapy of peri-implantitis. A 12-month prospective clinical study. Clin Oral Implants Res 23(2):205–210. https://doi.org/10.1111/j.1600-0501.2011.02276.x
Alhag M, Renvert S, Polyzois I, Claffey N (2008) Re-osseointegration on rough implant surfaces previously coated with bacterial biofilm: an experimental study in the dog. Clin Oral Implants Res 19(2):182–187. https://doi.org/10.1111/j.1600-0501.2007.01429.x
Schou S, Holmstrup P, Jorgensen T, Stoltze K, Hjorting-Hansen E, Wenzel A (2003) Autogenous bone graft and ePTFE membrane in the treatment of peri-implantitis. I. Clinical and radiographic observations in cynomolgus monkeys. Clin Oral Implants Res 14(4):391–403. https://doi.org/10.1034/j.1600-0501.2003.120909.x
Machado MA, Stefani CM, Sallum EA, Sallum AW, Tramontina VA, Nogueira-Filho GR, Nociti Junior FH (2000) Treatment of ligature-induced peri-implantitis defects by regenerative procedures. Part II: a histometric study in dogs. J Oral Sci 42(3):163–168. https://doi.org/10.2334/josnusd.42.163
Albouy JP, Abrahamsson I, Persson LG, Berglundh T (2011) Implant surface characteristics influence the outcome of treatment of peri-implantitis: an experimental study in dogs. J Clin Periodontol 38(1):58–64. https://doi.org/10.1111/j.1600-051X.2010.01631.x
Buchter A, Meyer U, Kruse-Losler B, Joos U, Kleinheinz J (2004) Sustained release of doxycycline for the treatment of peri-implantitis: randomised controlled trial. Br J Oral Maxillofac Surg 42(5):439–444. https://doi.org/10.1016/j.bjoms.2004.06.005
Rams TE, Degener JE, van Winkelhoff AJ (2014) Antibiotic resistance in human peri-implantitis microbiota. Clin Oral Implants Res 25(1):82–90. https://doi.org/10.1111/clr.12160
Mombelli A, Decaillet F (2011) The characteristics of biofilms in peri-implant disease. J Clin Periodontol 38(Suppl 11):203–213. https://doi.org/10.1111/j.1600-051X.2010.01666.x
Fraimow HS (2009) Systemic antimicrobial therapy in osteomyelitis. Semin Plast Surg 23(02):90–99. https://doi.org/10.1055/s-0029-1214161
Campoccia D, Montanaro L, Speziale P, Arciola CR (2010) Antibiotic-loaded biomaterials and the risks for the spread of antibiotic resistance following their prophylactic and therapeutic clinical use. Biomaterials 31(25):6363–6377. https://doi.org/10.1016/j.biomaterials.2010.05.005
Buttaro MA, Gimenez MI, Greco G, Barcan L, Piccaluga F (2005) High active local levels of vancomycin without nephrotoxicity released from impacted bone allografts in 20 revision hip arthroplasties. Acta Orthop 76(3):336–340
Winkler H, Stoiber A, Kaudela K, Winter F, Menschik F (2008) One stage uncemented revision of infected total hip replacement using cancellous allograft bone impregnated with antibiotics. J Bone Joint Surg Br 90(12):1580–1584. https://doi.org/10.1302/0301-620X.90B12.20742
Appelbaum PC (2007) Reduced glycopeptide susceptibility in methicillin-resistant Staphylococcus aureus (MRSA). Int J Antimicrob Agents 30(5):398–408. https://doi.org/10.1016/j.ijantimicag.2007.07.011
Whelton A (1984) The aminoglycosides. Clin Orthop Relat Res:66–74
Winkler H, Janata O, Berger C, Wein W, Georgopoulos A (2000) In vitro release of vancomycin and tobramycin from impregnated human and bovine bone grafts. J Antimicrob Chemother 46(3):423–428. https://doi.org/10.1093/jac/46.3.423
Armitage GC (1999) Development of a classification system for periodontal diseases and conditions. Ann Periodontol 4(1):1–6. https://doi.org/10.1902/annals.1999.4.1.1
Ramel CF, Lussi A, Ozcan M, Jung RE, Hammerle CH, Thoma DS (2016) Surface roughness of dental implants and treatment time using six different implantoplasty procedures. Clin Oral Implants Res 27(7):776–781. https://doi.org/10.1111/clr.12682
O’Leary TJ, Drake RB, Naylor JE (1972) The plaque control record. J Periodontol 43(1):38. https://doi.org/10.1902/jop.1972.43.1.38
Serino G, Turri A, Lang NP (2015) Maintenance therapy in patients following the surgical treatment of peri-implantitis: a 5-year follow-up study. Clin Oral Implants Res 26(8):950–956. https://doi.org/10.1111/clr.12418
Renvert S, Polyzois I, Claffey N (2012) Surgical therapy for the control of peri-implantitis. Clin Oral Implants Res 23 Suppl 6:84–94. https://doi.org/10.1111/j.1600-0501.2012.02554.x
Charalampakis G, Rabe P, Leonhardt A, Dahlen G (2011) A follow-up study of peri-implantitis cases after treatment. J Clin Periodontol 38(9):864–871. https://doi.org/10.1111/j.1600-051X.2011.01759.x
Khoshkam V, Chan HL, Lin GH, MacEachern MP, Monje A, Suarez F, Giannobile WV, Wang HL (2013) Reconstructive procedures for treating peri-implantitis: a systematic review. J Dent Res 92(12_suppl):131S–138S. https://doi.org/10.1177/0022034513509279
Carcuac O, Derks J, Charalampakis G, Abrahamsson I, Wennstrom J, Berglundh T (2016) Adjunctive systemic and local antimicrobial therapy in the surgical treatment of peri-implantitis: a randomized controlled clinical trial. J Dent Res 95(1):50–57. https://doi.org/10.1177/0022034515601961
Jepsen K, Jepsen S, Laine ML, Anssari Moin D, Pilloni A, Zeza B, Sanz M, Ortiz-Vigon A, Roos-Jansaker AM, Renvert S (2016) Reconstruction of peri-implant osseous defects: a multicenter randomized trial. J Dent Res 95(1):58–66. https://doi.org/10.1177/0022034515610056
Roos-Jansaker AM (2007) Long time follow up of implant therapy and treatment of peri-implantitis. Swed Dent J Suppl:7–66
Roccuzzo M, Bonino F, Bonino L, Dalmasso P (2011) Surgical therapy of peri-implantitis lesions by means of a bovine-derived xenograft: comparative results of a prospective study on two different implant surfaces. J Clin Periodontol 38(8):738–745. https://doi.org/10.1111/j.1600-051X.2011.01742.x
Sahrmann P, Attin T, Schmidlin PR (2011) Regenerative treatment of peri-implantitis using bone substitutes and membrane: a systematic review. Clin Implant Dent Relat Res 13(1):46–57. https://doi.org/10.1111/j.1708-8208.2009.00183.x
Chan HL, Lin GH, Suarez F, MacEachern M, Wang HL (2014) Surgical management of peri-implantitis: a systematic review and meta-analysis of treatment outcomes. J Periodontol 85(8):1027–1041. https://doi.org/10.1902/jop.2013.130563
Matarasso S, Iorio Siciliano V, Aglietta M, Andreuccetti G, Salvi GE (2014) Clinical and radiographic outcomes of a combined resective and regenerative approach in the treatment of peri-implantitis: a prospective case series. Clin Oral Implants Res 25(7):761–767. https://doi.org/10.1111/clr.12183
Heitz-Mayfield LJ and Mombelli A (2014) The therapy of peri-implantitis: a systematic review. Int J Oral Maxillofac Implants 29 Suppl:325–45. doi: https://doi.org/10.11607/jomi.2014suppl.g5.3
Etter TH, Hakanson I, Lang NP, Trejo PM, Caffesse RG (2002) Healing after standardized clinical probing of the perlimplant soft tissue seal: a histomorphometric study in dogs. Clin Oral Implants Res 13(6):571–580. https://doi.org/10.1034/j.1600-0501.2002.130601.x
Claffey N, Clarke E, Polyzois I, Renvert S (2008) Surgical treatment of peri-implantitis. J Clin Periodontol 35(8 Suppl):316–332. https://doi.org/10.1111/j.1600-051X.2008.01277.x
Mombelli A, Feloutzis A, Bragger U, Lang NP (2001) Treatment of peri-implantitis by local delivery of tetracycline. Clinical, microbiological and radiological results. Clin Oral Implants Res 12(4):287–294. https://doi.org/10.1034/j.1600-0501.2001.012004287.x
Renvert S, Lessem J, Lindahl C, Svensson M (2004) Treatment of incipient peri-implant infections using topical minocycline microspheres versus topical chlorhexidine gel as an adjunct to mechanical debridement. J Int Acad Periodontol 6(4 Suppl):154–159
Renvert S, Lessem J, Dahlen G, Lindahl C, Svensson M (2006) Topical minocycline microspheres versus topical chlorhexidine gel as an adjunct to mechanical debridement of incipient peri-implant infections: a randomized clinical trial. J Clin Periodontol 33(5):362–369. https://doi.org/10.1111/j.1600-051X.2006.00919.x
Schar D, Ramseier CA, Eick S, Arweiler NB, Sculean A, Salvi GE (2013) Anti-infective therapy of peri-implantitis with adjunctive local drug delivery or photodynamic therapy: six-month outcomes of a prospective randomized clinical trial. Clin Oral Implants Res 24(1):104–110. https://doi.org/10.1111/j.1600-0501.2012.02494.x
Renvert S, Lessem J, Dahlen G, Renvert H, Lindahl C (2008) Mechanical and repeated antimicrobial therapy using a local drug delivery system in the treatment of peri-implantitis: a randomized clinical trial. J Periodontol 79(5):836–844. https://doi.org/10.1902/jop.2008.070347
Bassetti M, Schar D, Wicki B, Eick S, Ramseier CA, Arweiler NB, Sculean A, Salvi GE (2014) Anti-infective therapy of peri-implantitis with adjunctive local drug delivery or photodynamic therapy: 12-month outcomes of a randomized controlled clinical trial. Clin Oral Implants Res 25(3):279–287. https://doi.org/10.1111/clr.12155
Renvert S, Polyzois I, Maguire R (2009) Re-osseointegration on previously contaminated surfaces: a systematic review. Clin Oral Implants Res 20(Suppl 4):216–227. https://doi.org/10.1111/j.1600-0501.2009.01786.x
Bert F, Leflon-Guibout V, Le Grand J, Bourdon N, Nicolas-Chanoine MH (2009) Emergence of vancomycin-dependent enterococci following glycopeptide therapy: case report and review. Pathol Biol (Paris) 57(1):56–60. https://doi.org/10.1016/j.patbio.2008.07.017
Edin ML, Miclau T, Lester GE, Lindsey RW, Dahners LE (1996) Effect of cefazolin and vancomycin on osteoblasts in vitro. Clin Orthop Relat Res:245–251
van Winkelhoff AJ, Herrera D, Oteo A, Sanz M (2005) Antimicrobial profiles of periodontal pathogens isolated from periodontitis patients in the Netherlands and Spain. J Clin Periodontol 32(8):893–898. https://doi.org/10.1111/j.1600-051X.2005.00782.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in this study involving human participants, were in accordance with ethical standards of the institutional research committee and with the principles stated in the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Nart, J., de Tapia, B., Pujol, À. et al. Vancomycin and tobramycin impregnated mineralized allograft for the surgical regenerative treatment of peri-implantitis: a 1-year follow-up case series. Clin Oral Invest 22, 2199–2207 (2018). https://doi.org/10.1007/s00784-017-2310-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-017-2310-0