Abstract
Background
Antimicrobial peptides (AMPs), such as human beta-defensin-2 (hBD-2) and the CC-chemokine ligand 20 (CCL20), exhibit direct microbicidal effects and mediator-like activity. It was hypothesized that wounding induces the expression of AMPs and pro-inflammatory mediators and that endogenous mediators, such as insulin-like growth factor-1 (IGF-1) and transforming growth factor-alpha (TGF-alpha), modulate this induced expression.
Material and methods
Monolayers of gingival epithelial cells (GECs) and gingival fibroblast (HGFs) from three different donors were wounded using the scratch assay (in vitro wounding) in the presence (test group) or absence (control group) of IGF-1 and TGF-alpha. In vitro wound closure was monitored over time (0, 6, 24, 48, 72 h), and wound areas were microscopically analyzed (Axio-Vision® Software, Zeiss). Gene expression analysis of the GAPDH, hBD-2, CCL20, interleukin-1 beta (IL-1 beta), and interleukin-8 (IL-8) was performed by qPCR.
Results
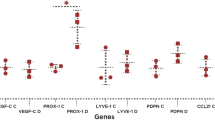
In comparison to control cells, IGF-1 and TGF-alpha significantly enhanced in vitro wound closure (P < 0.05). In GECs, IGF-1 induced the gene expression of IL-1 beta and IL-8 when compared to control cells (P < 0.05). In HGFs, wounding per se induced the messenger RNA of hBD-2, CCL20, and IL-1 beta, whereas IGF-1 and TGF-alpha reversed this effect (P < 0.05).
Conclusion
In gingival cells, the gene expression of AMPs was altered by injury, and endogenous growth factors further influenced the expression profiles, but with high interindividual differences.







Similar content being viewed by others
References
Stavropoulos A, Wikesjo UM (2012) Growth and differentiation factors for periodontal regeneration: a review on factors with clinical testing. J Periodontal Res 47:545–553. doi:10.1111/j.1600-0765.2012.01478.x
Raja S, Byakod G, Pudakalkatti P (2009) Growth factors in periodontal regeneration. Int J Dental Hyg 7:82–89. doi:10.1111/j.1601-5037.2009.00380.x
Yu F-SX, Yin J, Xu K, Huang J (2010) Growth factors and corneal epithelial wound healing. Brain Res Bull 81:229–235. doi:10.1016/j.brainresbull.2009.08.024
Annunziata M, Granata R, Ghigo E (2011) The IGF system. Acta Diabetol 48:1–9. doi:10.1007/s00592-010-0227-z
Han X, Amar S (2003) IGF-1 signaling enhances cell survival in periodontal ligament fibroblasts vs. gingival fibroblasts. J Dental Res 82:454–9
Bennett NT, Schultz GS (1993) Growth factors and wound healing: biochemical properties of growth factors and their receptors. Ame J Surg 165:728–737
Marikovsky M, Vogt P, Eriksson E, Rubin JS, Taylor WG, Joachim S, Klagsbrun M (1996) Wound fluid-derived heparin-binding EGF-like growth factor (HB-EGF) is synergistic with insulin-like growth factor-I for Balb/MK keratinocyte proliferation. J Invest Dermatol 106:616–621
Pierre EJ, Perez-Polo JR, Mitchell AT, Matin S, Foyt HL, Herndon DN (1997) Insulin-like growth factor-I liposomal gene transfer and systemic growth hormone stimulate wound healing. J Burn Care & Rehabil 18:287–291
Steenfos HH (1994) Growth factors and wound healing. Scand J Plast Reconstr Surg Hand Surg / Nordisk Plastikkirurgisk Forening [and] Nordisk klubb for Handkirurgi 28:95–105
Brown DL, Kane CD, Chernausek SD, Greenhalgh DG (1997) Differential expression and localization of insulin-like growth factors I and II in cutaneous wounds of diabetic and nondiabetic mice. Am J Pathol 151:715–724
Blakytny R, Jude EB, Martin Gibson J, Boulton AJ, Ferguson MW (2000) Lack of insulin-like growth factor 1 (IGF1) in the basal keratinocyte layer of diabetic skin and diabetic foot ulcers. J Pathol 190:589–594. doi:10.1002/(SICI)1096-9896(200004)190:5<589::AID-PATH553>3.0.CO;2-T
Hodak E, Gottlieb AB, Anzilotti M, Krueger JG (1996) The insulin-like growth factor 1 receptor is expressed by epithelial cells with proliferative potential in human epidermis and skin appendages: correlation of increased expression with epidermal hyperplasia. J Invest Dermatol 106:564–570
Andresen JL, Ehlers N (1998) Chemotaxis of human keratocytes is increased by platelet-derived growth factor-BB, epidermal growth factor, transforming growth factor-alpha, acidic fibroblast growth factor, insulin-like growth factor-I, and transforming growth factor-beta. Curr Eye Res 17:79–87
Li Y, Fan J, Chen M, Li W, Woodley DT (2006) Transforming growth factor-alpha: a major human serum factor that promotes human keratinocyte migration. J Invest Dermatol 126:2096–2105. doi:10.1038/sj.jid.5700350
Kim I, Mogford JE, Chao JD, Mustoe TA (2001) Wound epithelialization deficits in the transforming growth factor-alpha knockout mouse. Wound Repair Regen 9:386–390
Kheradmand F, Folkesson HG, Shum L, Derynk R, Pytela R, Matthay MA (1994) Transforming growth factor-alpha enhances alveolar epithelial cell repair in a new in vitro model. Am J Physiol 267:L728–738
Dale BA, Kimball JR, Krisanaprakornkit S, Roberts F, Robinovitch M, O’Neal R, Valore EV, Ganz T, Anderson GM, Weinberg A (2001) Localized antimicrobial peptide expression in human gingiva. J Periodontal Res 36:285–294
Dale BA (2002) Periodontal epithelium: a newly recognized role in health and disease. Periodontol 2000(30):70–78
Dale BA (2003) Fascination with epithelia: architecture, proteins, and functions. J Dent Res 82:866–869
Dale BA, Fredericks LP (2005) Antimicrobial peptides in the oral environment: expression and function in health and disease. Curr Issues Mol Biol 7:119–133
Lehrer RI (2004) Primate defensins. Nat Rev Microbiol 2:727–738
Chung WO, Dommisch H, Yin L, Dale BA (2007) Expression of defensins in gingiva and their role in periodontal health and disease. Curr Pharm Des 13:3073–3083
Yang D, Biragyn A, Hoover DM, Lubkowski J, Oppenheim JJ (2004) Multiple roles of antimicrobial defensins, cathelicidins, and eosinophil-derived neurotoxin in host defense. Annu Rev Immunol 22:181–215
Dunsche A, Acil Y, Dommisch H, Siebert R, Schroder JM, Jepsen S (2002) The novel human beta-defensin-3 is widely expressed in oral tissues. Eur J Oral Sci 110:121–124
Dommisch H, Acil Y, Dunsche A, Winter J, Jepsen S (2005) Differential gene expression of human beta-defensins (hBD-1, -2, -3) in inflammatory gingival diseases. Oral Microbiol Immunol 20:186–190
Rizzo A, Paolillo R, Buommino E, Lanza AG, Guida L, Annunziata M, Carratelli CR (2008) Modulation of cytokine and beta-defensin 2 expressions in human gingival fibroblasts infected with Chlamydia pneumoniae. Int Immunopharmacol 8:1239–1247. doi:10.1016/j.intimp.2008.04.015
Dommisch H, Reinartz M, Backhaus T, Deschner J, Chung W, Jepsen S (2012) Antimicrobial responses of primary gingival cells to Porphyromonas gingivalis. J Clin Periodontol 39:913–922. doi:10.1111/j.1600-051X.2012.01933.x
Hoover DM, Boulegue C, Yang D, Oppenheim JJ, Tucker K, Lu W, Lubkowski J (2002) The structure of human macrophage inflammatory protein-3alpha/CCL20. Linking antimicrobial and CC chemokine receptor-6-binding activities with human beta-defensins. J Biol Chem 277:37647–37654
Yang D, Chertov O, Bykovskaia SN, Chen Q, Buffo MJ, Shogan J, Anderson M, Schroder JM, Wang JM, Howard OM, Oppenheim JJ (1999) Beta-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6. Science 286:525–528
Schutyser E, Struyf S, Van Damme J (2003) The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev 14:409–426
Yang D, Chen Q, Chertov O, Oppenheim JJ (2000) Human neutrophil defensins selectively chemoattract naive T and immature dendritic cells. J Leukoc Biol 68:9–14
Hasturk H, Kantarci A, Goguet-Surmenian E, Blackwood A, Andry C, Serhan CN, Van Dyke TE (2007) Resolvin E1 regulates inflammation at the cellular and tissue level and restores tissue homeostasis in vivo. J Immunol 179:7021–7029
Weber A, Wasiliew P, Kracht M (2010) Interleukin-1beta (IL-1beta) processing pathway. Sci Signal 3:cm2. doi:10.1126/scisignal.3105cm2
Schroder JM, Christophers E (1992) The biology of NAP-1/IL-8, a neutrophil-activating cytokine. Immunol Ser 57:387–416
Dommisch H, Chung WO, Rohani MG, Williams D, Rangarajan M, Curtis MA, Dale BA (2007) Protease-activated receptor 2 mediates human beta-defensin 2 and CC chemokine ligand 20 mRNA expression in response to proteases secreted by Porphyromonas gingivalis. Infect Immun 75:4326–4333
Tokumaru S, Sayama K, Shirakata Y, Komatsuzawa H, Ouhara K, Hanakawa Y, Yahata Y, Dai X, Tohyama M, Nagai H, Yang L, Higashiyama S, Yoshimura A, Sugai M, Hashimoto K (2005) Induction of keratinocyte migration via transactivation of the epidermal growth factor receptor by the antimicrobial peptide LL-37. J Immunol 175:4662–4668
Dommisch H, Chung WO, Jepsen S, Hacker BM, Dale BA (2010) Phospholipase C, p38/MAPK, and NF-kappaB-mediated induction of MIP-3alpha/CCL20 by Porphyromonas gingivalis. Innate Immun 16:226–234. doi:10.1177/1753425909339237
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Harishkumar M, Masatoshi Y, Hiroshi S, Tsuyomu I, Masugi M (2013) Revealing the mechanism of in vitro wound healing properties of Citrus tamurana extract. BioMed Res Int 2013:963457. doi:10.1155/2013/963457
Mustafa M, Zarrough A, Bolstad AI, Lygre H, Mustafa K, Hasturk H, Serhan CN, Kantarci A, Van Dyke TE (2013) Resolvin D1 protects periodontal ligament. Am J Physiol Cell Physiol. doi:10.1152/ajpcell.00242.2012
Tang XP, Tang GD, Fang CY, Liang ZH, Zhang LY (2013) Effects of ginsenoside Rh2 on growth and migration of pancreatic cancer cells. World J Gastroenterol : WJG 19:1582–1592. doi:10.3748/wjg.v19.i10.1582
Elijah IE, Branski LK, Finnerty CC, Herndon DN (2011) The GH/IGF-1 system in critical illness. Best Practice & Research Clinical Endocrinology &. Metabolism 25:759–767. doi:10.1016/j.beem.2011.06.002
Oberringer M, Meins C, Bubel M, Pohlemann T (2008) In vitro wounding: effects of hypoxia and transforming growth factor beta1 on proliferation, migration and myofibroblastic differentiation in an endothelial cell-fibroblast co-culture model. J Mol Histol 39:37–47. doi:10.1007/s10735-007-9124-3
Wang L, Ko CY, Meyers EE, Pedroja BS, Pelaez N, Bernstein AM (2011) Concentration-dependent effects of transforming growth factor beta1 on corneal wound healing. Mol Vision 17:2835–2846
Tall EG, Bernstein AM, Oliver N, Gray JL, Masur SK (2010) TGF-beta-stimulated CTGF production enhanced by collagen and associated with biogenesis of a novel 31-kDa CTGF form in human corneal fibroblasts. Invest Ophthalmol Vis Sci 51:5002–5011. doi:10.1167/iovs.09-5110
Garlick JA, Parks WC, Welgus HG, Taichman LB (1996) Re-epithelialization of human oral keratinocytes in vitro. J Dental Res 75:912–918
Niyonsaba F, Ushio H, Nakano N, Ng W, Sayama K, Hashimoto K, Nagaoka I, Okumura K, Ogawa H (2007) Antimicrobial peptides human beta-defensins stimulate epidermal keratinocyte migration, proliferation and production of proinflammatory cytokines and chemokines. J Invest Dermatol 127:594–604
Niyonsaba F, Ogawa H, Nagaoka I (2004) Human beta-defensin-2 functions as a chemotactic agent for tumour necrosis factor-alpha-treated human neutrophils. Immunology 111:273–281
Joly S, Organ CC, Johnson GK, McCray PB Jr, Guthmiller JM (2005) Correlation between beta-defensin expression and induction profiles in gingival keratinocytes. Mol Immunol 42:1073–1084. doi:10.1016/j.molimm.2004.11.001
Acknowledgments
The authors kindly thank Mrs. J. Eich and Ms. D. Lalaouni for their excellent technical assistance. This study was financially supported by the German Society of Periodontology (Deutsche Gesellschaft für Parodontologie, DGP; authors: H.D., S.J.) and the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG, Clinical Research Unit 208; authors: TP2, H.D., S.J.; TP7, W.G.; TP4, J.D.; TP8, A.J.; TP9, L.H.; TP10, J.W.).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 56 kb)
Rights and permissions
About this article
Cite this article
Dommisch, H., Winter, J., Götz, W. et al. Effect of growth factors on antimicrobial peptides and pro-inflammatory mediators during wound healing. Clin Oral Invest 19, 209–220 (2015). https://doi.org/10.1007/s00784-014-1239-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-014-1239-9