Abstract
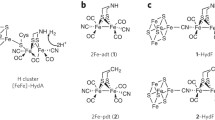
[FeFe]-hydrogenase catalyzes the reversible reduction of protons to H2 at a complex metallocofactor site, the H-cluster. Biosynthesis of this active-site H-cluster requires three maturation enzymes: the radical S-adenosylmethionine enzymes HydE and HydG synthesize the nonprotein ligands, while the GTPase HydF provides a scaffold for assembly of the 2Fe subcluster of the H-cluster ([2Fe]H) prior to its transfer to hydrogenase. To delineate the assembly and delivery steps for the 2Fe precursor cluster coordinated to HydF ([2Fe]F), we have heterologously expressed HydF in the presence of HydE alone (HydFE) or HydG alone (HydFG), and characterized the resulting purified HydFE and HydFG using UV–visible, EPR, and FTIR spectroscopies and biochemical assays. The iron–sulfur clusters on HydF are modified by co-expression with HydE or HydG, as evidenced by the changes in the visible, EPR, and FTIR spectral features. Further, biochemical assays show that HydFE is capable of activating HydAΔEFG to a limited extent (~ 1% of WT) even though the normal source of CO and CN– ligands of [2Fe]H (HydG) was absent. Activation assays performed with HydFG, in contrast, exhibit no ability to mature HydAΔEFG. It appears that in the case of HydFE, trace diatomics from the cellular environment are incorporated into a [2Fe]F-like precursor on HydF in the absence of HydG. We conclude that the product of HydE, presumably the dithiomethylamine ligand of [2Fe]H, is absolutely essential to the activation process, while the diatomic products of HydG can be provided from alternate sources.
Graphic abstract







Similar content being viewed by others
References
Shepard EM, Mus F, Betz J, Byer A, Duffus BR, Peters JW, Broderick JB (2014) [FeFe]-hydrogenase maturation. Biochemistry 53:4090–4104
Shepard EM, Byer AS, Broderick JB (2017) Iron-sulfur cluster states of the hydrogenase Maturase HydF. Biochemistry 56:4733–4734
Mulder DW, Shepard EM, Meuser JE, Joshi N, King PW, Posewitz MC, Broderick JB, Peters JW (2011) Insights into [FeFe]-hydrogenase structure, mechanism, and maturation. Structure 19:1038–1052
Peters JW, Schut GJ, Boyd ES, Mulder DW, Shepard EM, Broderick JB, King PW, Adams MWW (2015) [FeFe]- and [NiFe]-hydrogenase diversity, mechanism, and maturation. BBA Mol Cell Res 1853:1350–1369
Lubitz W, Ogata H, Rudiger O, Reijerse E (2014) Hydrogenases. Chem Rev 114:4081–4148
Mulder DW, Ortillo DO, Gardenghi DJ, Naumov A, Ruebush SS, Szilagyi RK, Huynh BH, Broderick JB, Peters JW (2009) Activation of HydAΔEFG requires a preformed [4Fe-4S] cluster. Biochemistry 48:6240–6248
Mulder DW, Boyd ES, Sarma R, Lange RK, Endrizzi JA, Broderick JB, Peters JW (2010) Stepwise [FeFe]-hydrogenase H-cluster assembly revealed in the structure of HydA(DeltaEFG). Nature 465:248–251
McGlynn SE, Ruebush SS, Naumov A, Nagy LE, Dubini A, King PW, Broderick JB, Posewitz MC, Peters JW (2007) In vitro activation of [FeFe] hydrogenase: new insights into hydrogenase maturation. J Biol Inorg Chem 12:443–447
Broderick JB, Byer AS, Duschene KS, Duffus BR, Betz JN, Shepard EM, Peters JW (2014) H-cluster assembly during the maturation of the [FeFe]-hydrogenase. J Biol Inorg Chem 19:747–757
Byer AS, Shepard EM, Peters JW, Broderick JB (2015) Radical S-adenosyl-l-methionine chemistry in the synthesis of hydrogenase and nitrogenase metal cofactors. J Biol Chem 290:3987–3994
Kuchenreuther JM, Britt RD, Swartz JR (2012) New insights into [FeFe] hydrogenase activation and maturase function. PLoS One 7:e45850
Brazzolotto X, Rubach JK, Gaillard J, Gambarelli S, Atta M, Fontecave M (2006) The [Fe-Fe]-hydrogenase maturation protein HydF from Thermotoga maritima is a GTPase with an iron–sulfur cluster. J Biol Chem 281:769–774
McGlynn SE, Shepard EM, Winslow MA, Naumov AV, Duschene KS, Posewitz MC, Broderick WE, Broderick JB, Peters JW (2008) HydF as a scaffold protein in [FeFe] hydrogenase H-cluster biosynthesis. FEBS Lett 582:2183–2187
Czech I, Silakov A, Lubitz W, Happe T (2010) The [FeFe]-hydrogenase maturase HydF from Clostridium acetobutylicum contains a CO and CN-ligated cluster. FEBS Lett 584:638–642
Shepard EM, McGlynn SE, Bueling AL, Grady-Smith C, George SJ, Winslow MA, Cramer SP, Peters JW, Broderick JB (2010) Synthesis of the 2Fe-subcluster of the [FeFe]-hydrogenase H-cluster on the HydF scaffold. Proc Natl Acad Sci USA 107:10448–10453
Scott AG, Szilagyi RK, Mulder DW, Ratzloff MW, Byer AS, King PW, Broderick WE, Shepard EM, Broderick JB (2018) Compositional and structural insights into the nature of the H-cluster precursor on HydF. Dalton Trans 47:9521–9535
Németh B, Esmieu C, Redman HJ, Berggren G (2019) Monitoring H-cluster assembly using a synthetic HydF protein. Dalton Trans 48:5978–5986
Berggren G, Adamska A, Lambertz C, Simmons TR, Esselborn J, Atta M, Gambarelli S, Mouesca JM, Reijerse E, Lubitz W, Happe T, Artero V, Fontecave M (2013) Biomimetic assembly and activation of [FeFe]-hydrogenases. Nature 499:66–69
Posewitz MC, King PW, Smolinski SL, Zhang L, Seibert M, Ghirardi ML (2004) Discovery of two novel radical S-adenosylmethionine proteins required for the assembly of an active [Fe] hydrogenase. J Biol Chem 279:25711–25720
Pilet E, Nicolet Y, Mathevon C, Douki T, Fontecilla-Camps JC, Fontecave M (2009) The role of the maturase HydG in [FeFe]-hydrogenase active site synthesis and assembly. FEBS Lett 583:506–511
Shepard EM, Duffus BR, McGlynn SE, Challand MR, Swanson KD, Roach PL, Peters JW, Broderick JB (2010) [FeFe]-hydrogenase maturation: HydG-catalyzed synthesis of carbon monoxide. J Am Chem Soc 132:9247–9249
Driesener RC, Duffus BR, Shepard EM, Bruzas IR, Duschene KS, Coleman NJ-R, Marrison APG, Salvadori E, Kay CWM, Peters JW, Broderick JB, Roach PL (2013) Biochemical and kinetic characterization of radical S-adenosylmethionine enzyme HydG. Biochemistry 52:8696–8707
Duffus BR, Ghose S, Peters JW, Broderick JB (2014) Reversible H atom abstraction catalyzed by the radical S-adenosylmethionine enzyme HydG. J Am Chem Soc 136:13086–13089
Kuchenreuther JM, Myers WK, Suess DLM, Stich TA, Pelmenschikov V, Shiigi SA, Cramer SP, Swartz JR, Britt RD, George SJ (2014) The HydG enzyme generates an Fe(CO)2(CN) synthon in assembly of the FeFe hydrogenase H-cluster. Science 343:424–427
Pagnier A, Martin L, Zeppieri L, Nicolet Y, Fontecilla-Camps JC (2016) CO and CN-syntheses by [FeFe]-hydrogenase maturase HydG are catalytically differentiated events. Proc Natl Acad Sci USA 113:104–109
Driesener RC, Challand MR, McGlynn SE, Shepard EM, Boyd ES, Broderick JB, Peters JW, Roach PL (2010) [FeFe]-hydrogenase cyanide ligands derived from S-adenosylmethionine-dependent cleavage of tyrosine. Angew Chem Int Ed Engl 49:1687–1690
Kuchenreuther JM, George SJ, Grady-Smith CS, Cramer SP, Swartz JR (2011) Cell-free H-cluster synthesis and [FeFe] hydrogenase activation: all five CO and CN-ligands derive from tyrosine. PLoS One 6:e20346
Betz JN, Boswell NW, Fugate CJ, Holliday GL, Akiva E, Scott AG, Babbitt PC, Peters JW, Shepard EM, Broderick JB (2015) [FeFe]-hydrogenase maturation: insights into the role HydE plays in dithiomethylamine biosynthesis. Biochemistry 54:1807–1818
Rohac R, Amara P, Benjdia A, Martin L, Ruffié P, Favier A, Berteau O, Mouesca J-M, Fontecilla-Camps JC, Nicolet Y (2016) Carbon–sulfur bond-forming reaction catalysed by the radical SAM enzyme HydE. Nat Chem 8:491–500
Czech I, Stripp S, Sanganas O, Leidel N, Happe T, Haumann M (2011) The [FeFe]-hydrogenase maturation protein HydF contains a H-cluster like [4Fe4S]–2Fe site. FEBS Lett 585:225–230
Shepard EM, Byer AS, Betz JN, Peters JW, Broderick JB (2016) A redox active [2Fe-2S] cluster on the hydrogenase maturase HydF. Biochemistry 55:3514–3527
Shepard EM, Byer AS, Aggarwal P, Betz JN, Scott AG, Shisler KA, Usselman RJ, Eaton GR, Eaton SS, Broderick JB (2017) Electron spin relaxation and biochemical characterization of the hydrogenase maturase HydF: insights into [2Fe-2S] and [4Fe-4S] cluster communication and hydrogenase activation. Biochemistry 56:3234–3247
Stoll S, Schweiger A (2006) EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J Magn Res 178:42–55
Rupp H, Rao KK, Hall DO, Cammack R (1978) Electron spin relaxation of iron–sulphur proteins studied by microwave power saturation. Biochim Biophys Acta 537:255–269
Lemon BJ, Peters JW (1999) Binding of exogenously added carbon monoxide at the active site of the iron-only hydrogenase (CpI) from Clostridium pasteurianum. Biochemistry 38:12969–12973
Quillin ML, Arduini RM, Olson JS, Phillips GNJ (1993) High-resolution crystal structures of distal histidine mutants of sperm whale myoglobin. J Mol Biol 234:140–155
Maines MD (1997) The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol 37:517–554
Galazzo L, Maso L, De Rosa E, Bortolus M, Doni D, Acquasaliente L, De Filippis V, Costantini P, Carbonera D (2017) Identifying conformational changes with site-directed spin labeling reveals that the GTPase domain of HydF is a molecular switch. Sci Rep 7:1714
Vallese F, Berto P, Ruzzene M, Cendron L, Sarno S, De Rosa E, Giacometti GM, Costantini P (2012) Biochemical analysis of the interactions between the proteins involved in the [FeFe]-hydrogenase maturation process. J Biol Chem 287:36544–36555
Bortolus M, Costantini P, Doni D, Carbonera D (2018) Overview of the maturation machinery of the H-cluster of [FeFe]-hydrogenases with a focus on HydF. Int J Mol Sci 19:3118
Peters JW, Szilagyi RK, Naumov A, Douglas T (2006) A radical solution for the biosynthesis of the H-cluster of hydrogenase. FEBS Lett 580:363–367
Acknowledgements
The authors gratefully acknowledge the U.S. Department of Energy, Office of Basic Energy Sciences (DE-SC0005404 to J.B.B. and E.M.S.), for supporting all experimental work other than the FTIR spectroscopy. FTIR spectroscopy was supported by the U.S. Department of Energy, Office of Basic Energy Sciences, Chemical Sciences, Geosciences and Biosciences Division, and under U.S. Department of Energy Contract No. DE-AC36-08-GO28308 with the National Renewable Energy Lab.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Byer, A.S., Shepard, E.M., Ratzloff, M.W. et al. H-cluster assembly intermediates built on HydF by the radical SAM enzymes HydE and HydG. J Biol Inorg Chem 24, 783–792 (2019). https://doi.org/10.1007/s00775-019-01709-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-019-01709-7