Abstract
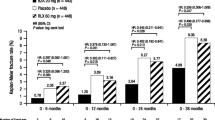
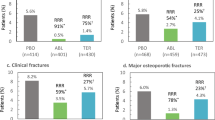
This study examined whether the global clinical data for bazedoxifene could be extrapolated to a Japanese population by evaluating the results of a phase 2 study in postmenopausal Japanese women with osteoporosis as compared to those of a pivotal, phase 3 study. The efficacy of bazedoxifene 20 and 40 mg versus placebo on lumbar spine bone mineral density (BMD), bone turnover markers, lipid profile, incidence of fractures, and safety parameters was compared between the Japanese phase 2 study (N = 429) and the global phase 3 study (N = 7,492) during a 2-year period. In the primary population for assessment of bridging, differences in the mean percent change from baseline in lumbar spine BMD at 2 years relative to placebo were greater for women treated with bazedoxifene 20 and 40 mg in the phase 2 study than in the phase 3 study. BMD changes in the bazedoxifene groups were confirmed to be similar between the phase 2 study population and a subset of the phase 3 study population with similar baseline characteristics. The effects of bazedoxifene on incidence of fractures, bone turnover markers, and lipid metabolism were similar between studies. There were no major differences in safety parameters between studies. The greater improvement in lumbar spine BMD and similar results in bone turnover markers, fracture incidence, and safety profile observed with bazedoxifene in the phase 2 study compared with the phase 3 study confirmed the feasibility of extrapolating the global clinical data to a Japanese population.


Similar content being viewed by others
References
Johnell O, Kanis JA (2006) An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int 17:1726–1733
National Osteoporosis Foundation (2010) Fast Facts on Osteoporosis. http://www.nof.org/node/40. Accessed 17 Nov 2011
Finkelstein JS, Brockwell SE, Mehta V, Greendale GA, Sowers MR, Ettinger B, Lo JC, Johnston JM, Cauley JA, Danielson ME, Neer RM (2008) Bone mineral density changes during the menopause transition in a multiethnic cohort of women. J Clin Endocrinol Metab 93:861–868
Lau EM (2009) The epidemiology of osteoporosis in Asia. IBMS BoneKEy 6:190–193
International Osteoporosis Foundation (2009) The Asian Audit. Epidemiology, costs and burden of osteoporosis in Asia 2009. http://www.iofbonehealth.org/publications/asian-audit-2009.html. Accessed 25 Oct 2010
Hagino H, Furukawa K, Fujiwara S, Okano T, Katagiri H, Yamamoto K, Teshima R (2009) Recent trends in the incidence and lifetime risk of hip fracture in Tottori, Japan. Osteoporos Int 20:543–548
North American Menopause Society (2010) Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society. Menopause 17:25–54
Papaioannou A, Kennedy CC, Dolovich L, Lau E, Adachi JD (2007) Patient adherence to osteoporosis medications: problems, consequences and management strategies. Drugs Aging 24:37–55
Miller PD, Chines AA, Christiansen C, Hoeck HC, Kendler DL, Lewiecki EM, Woodson G, Levine AB, Constantine G, Delmas PD (2008) Effects of bazedoxifene on BMD and bone turnover in postmenopausal women: 2-yr results of a randomized, double-blind, placebo-, and active-controlled study. J Bone Miner Res 23:525–535
Silverman SL, Christiansen C, Genant HK, Vukicevic S, Zanchetta JR, de Villiers TJ, Constantine GD, Chines AA (2008) Efficacy of bazedoxifene in reducing new vertebral fracture risk in postmenopausal women with osteoporosis: results from a 3-year, randomized, placebo-, and active-controlled clinical trial. J Bone Miner Res 23:1923–1934
Xu L, Tsai KS, Kim GS, Wu Y, Vincendon P, Chines AA, Constantine GD (2011) Efficacy and safety of bazedoxifene in postmenopausal Asian women. Osteoporos Int 22:559–565
Itabashi A, Yoh K, Chines AA, Miki T, Takada M, Sato H, Gorai I, Sugimoto T, Mizunuma H, Ochi H, Constantine GD, Ohta H (2011) Effects of bazedoxifene on bone mineral density, bone turnover, and safety in postmenopausal Japanese women with osteoporosis. J Bone Miner Res 26:519–529
Orimo H, Sugioka Y, Fukinaga M, Muto Y, Hotokebuchi T, Gorai I, Nakamura T, Kushida K, Tanaka H, Ikai T, Oh-Hashi Y (1998) Diagnostic criteria of primary osteoporosis. J Bone Miner Metab 16:139–150
D’Agostino RB Jr (1998) Propensity score methods for bias reduction in the comparison of a treatment to a non-randomized control group. Stat Med 17:2265–2281
Sarkar S, Watts S, Ohashi Y, Carroll RJ, Uesaka H, Mason TM, Rivera C (2002) Bridging data between two ethnic populations. A new application of matched case-control methodology. Drug Inf J 36:349–356
Mizuno S, Yamaguchi T, Fukushima A, Matsuyama Y, Ohashi Y (2005) Overlap coefficient for assessing the similarity of pharmacokinetic data between ethnically different populations. Clin Trials 2:174–181
National Osteoporosis Foundation (2010) Clinician’s guide to prevention and treatment of osteoporosis. http://www.nof.org/professionals/pdfs/NOF_ClinicianGuide2009_v7.pdf. Accessed 23 July 2010
Kuchuk NO, van Schoor NM, Pluijm SM, Chines A, Lips P (2009) Vitamin D status, parathyroid function, bone turnover and bone mineral density in postmenopausal women with osteoporosis in global perspective. J Bone Miner Res 24:693–701
Bischoff-Ferrari HA, Dietrich T, Orav EJ, Dawson-Hughes B (2004) Positive association between 25-hydroxy vitamin D levels and bone mineral density: a population-based study of younger and older adults. Am J Med 116:634–639
Chandran M, Hoeck HC, Wong HC, Zhang RF, Dimai HP (2010) Vitamin D status and its relationship with bone mineral density and parathyroid hormone in South-East Asian adults with low bone density. Endocr Pract 17:226–234
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J, Gluer CC, Krueger K, Cohen FJ, Eckert S, Ensrud KE, Avioli LV, Lips P, Cummings SR, for the Multiple Outcomes of Raloxifene Evaluation (MORE) Investigators (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. JAMA 282:637–645
Bauer DC, Black DM, Garnero P, Hochberg M, Ott S, Orloff J, Thompson DE, Ewing SK, Delmas PD (2004) Change in bone turnover and hip, non-spine, and vertebral fracture in alendronate-treated women: the fracture intervention trial. J Bone Miner Res 19:1250–1258
Eastell R, Barton I, Hannon RA, Chines A, Garnero P, Delmas PD (2003) Relationship of early changes in bone resorption to the reduction in fracture risk with risedronate. J Bone Miner Res 18:1051–1056
Bjarnason NH, Sarkar S, Duong T, Mitlak B, Delmas PD, Christiansen C (2001) Six and twelve month changes in bone turnover are related to reduction in vertebral fracture risk during 3 years of raloxifene treatment in postmenopausal osteoporosis. Osteoporos Int 12:922–930
Ito M, Nishida A, Uetani M, Hayashi K (2001) Osteoporosis in the Japanese population. Semin Musculoskelet Radiol 5:121–126
Koh LK (2002) An Asian perspective to the problem of osteoporosis. Ann Acad Med Singapore 31:26–29
Acknowledgments
These studies and analysis were sponsored by Wyeth Research, Collegeville, PA, USA, and Wyeth K. K., Tokyo, Japan, which were acquired by Pfizer Inc in October 2009. Medical writing support was provided by Katie Gersh, PhD, of MedErgy and was funded by Pfizer Inc.
Conflict of interest
Dr. Itabashi reports serving as a consultant for Wyeth, Chugai, and Ohtsuka. Dr. Yoh reports serving as a consultant for Wyeth. Dr. Miki reports serving as a consultant for Wyeth. Dr. Takada and Dr. Sato have no relevant conflicts of interest to report. Dr. Gorai reports serving as a consultant for Wyeth. Dr. Sugimoto reports serving as a consultant for Wyeth, Asahi Kasei, and Daiichi-Sankyo. Dr. Mizunuma reports serving as a consultant for Wyeth, Chugai, Shiseido, Ono, and Aska. Dr. Ohta reports serving as a consultant for Pola-Pharma, Otsuka, Teijin Pharma, and Takeda. Dr. Ochi is a current employee of Pfizer Inc. Drs. Chines and Constantine were previously employees of Pfizer Inc. Dr. Chines is a stockholder in Pfizer Inc.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. A. Chines and G. D. Constantine are no longer affiliated with Wyeth/Pfizer.
About this article
Cite this article
Itabashi, A., Yoh, K., Chines, A.A. et al. Bridging analysis of the efficacy and safety of bazedoxifene in Japanese and global populations of postmenopausal women with osteoporosis. J Bone Miner Metab 33, 61–72 (2015). https://doi.org/10.1007/s00774-013-0554-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-013-0554-6