Summary.
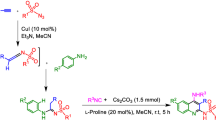
Chloranil and 2,3-dichloro-1,4-naphthoquinone have been linked to different natural and unnatural amino acids via a vinylic spacer. Two routes were developed for the facile preparation of these novel modified amino acids: the direct method, which can only be applied to secondary amines, and the indirect method (transamination reaction), which can be applied to any amino acid or ester.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received June 16, 2000 Accepted August 28, 2000
Rights and permissions
About this article
Cite this article
Alnabari, M., Bittner, S. New quinone-amino acid conjugates linked via a vinylic spacer. Amino Acids 20, 381–387 (2001). https://doi.org/10.1007/s007260170034
Issue Date:
DOI: https://doi.org/10.1007/s007260170034