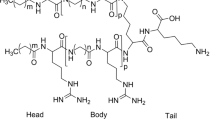
Abstract
The necessity to develop therapeutic agents and strategies to abate the spread of antibiotic-resistant pathogens is prominent. Antimicrobial peptides (AMPs) provide scaffolds and inspiration for antibiotic development. As an AMP of shorter scaffold, eight dilipid ultrashort cationic lipopeptides (dUSCLs) were prepared consisting of only four amino acids and varying dilipids. Lipids were acylated at the peptide N-terminus and the ε-amine side chain of the N-terminal l-lysine. Compounds that possess aliphatic dilipids of ≥ 11 carbons-long showed significant hemolysis and therefore limited therapeutic application. Several non-hemolytic dUSCLs were identified to enhance the activity of chloramphenicol and other conventional antibiotics against Gram-negative bacteria. Compounds 2 and 6 have a short peptide sequence of KKKK and KKGK, respectively, and are both acylated with an aliphatic dilipid of nine carbons-long potentiated chloramphenicol against MDR clinical isolates of Pseudomonas aeruginosa, Acinetobacter baumannii and Enterobacteriaceae. Both dUSCLs showed comparable adjuvant potency in combination with chloramphenicol. However, dUSCL 2 synergized with a wider span of antibiotic classes against P. aeruginosa relative to dUSCL 6 that included rifampicin, trimethoprim, minocycline, fosfomycin, piperacillin, ciprofloxacin, levofloxacin, moxifloxacin, linezolid and vancomycin. Our data revealed that dUSCLs can indirectly disrupt active efflux of chloramphenicol in P. aeruginosa. This along with their membrane-permeabilizing properties may explain the dUSCLs synergistic combination with conventional antibiotics against Gram-negative bacteria.




Similar content being viewed by others
References
Ahn M, Jacob B, Gunasekaran P et al (2014) Poly-lysine peptidomimetics having potent antimicrobial activity without hemolytic activity. Amino Acids 46:2259–2269. https://doi.org/10.1007/s00726-014-1778-z
Dawgul MA, Greber KE, Bartoszewska S et al (2017) In vitro evaluation of cytotoxicity and permeation study on lysine- and arginine-based lipopeptides with proven antimicrobial activity. Molecules. https://doi.org/10.3390/molecules22122173
Domalaon R, Yang X, O’Neil J et al (2014) Structure-activity relationships in ultrashort cationic lipopeptides: the effects of amino acid ring constraint on antibacterial activity. Amino Acids 46:2517–2530. https://doi.org/10.1007/s00726-014-1806-z
Domalaon R, Zhanel GG, Schweizer F (2016) Short antimicrobial peptides and peptide scaffolds as promising antibacterial agents. Curr Top Med Chem 16:1217–1230. https://doi.org/10.2174/1568026615666150915112459
Domalaon R, Berry L, Tays Q et al (2018a) Development of dilipid polymyxins: investigation on the effect of hydrophobicity through its fatty acyl component. Bioorg Chem. https://doi.org/10.1016/j.bioorg.2018.07.018
Domalaon R, Idowu T, Zhanel GG, Schweizer F (2018b) Antibiotic hybrids: the next generation of agents and adjuvants against Gram-negative pathogens? Clin Microbiol Rev. https://doi.org/10.1128/cmr.00077-17
Domalaon R, Sanchak Y, Koskei LC et al (2018c) Short proline-rich lipopeptide potentiates minocycline and rifampin against multidrug- and extensively drug-resistant Pseudomonas aeruginosa. Antimicrob Agents Chemother. https://doi.org/10.1128/aac.02374-17
Feng Q, Huang Y, Chen M et al (2015) Functional synergy of alpha-helical antimicrobial peptides and traditional antibiotics against Gram-negative and Gram-positive bacteria in vitro and in vivo. Eur J Clin Microbiol Infect Dis 34:197–204. https://doi.org/10.1007/s10096-014-2219-3
Findlay B, Zhanel GG, Schweizer F (2012) Investigating the antimicrobial peptide “window of activity” using cationic lipopeptides with hydrocarbon and fluorinated tails. Int J Antimicrob Agents 40:36–42. https://doi.org/10.1016/j.ijantimicag.2012.03.013
Fjell CD, Hiss JA, Hancock REW, Schneider G (2011) Designing antimicrobial peptides: form follows function. Nat Rev Drug Discov 11:37–51. https://doi.org/10.1038/nrd3591
Greber KE, Dawgul M, Kamysz W et al (2014) Biological and surface-active properties of double-chain cationic amino acid-based surfactants. Amino Acids 46:1893–1898. https://doi.org/10.1007/s00726-014-1744-9
Greber KE, Dawgul M, Kamysz W, Sawicki W (2017) Cationic net charge and counter ion type as antimicrobial activity determinant factors of short lipopeptides. Front Microbiol 8:123. https://doi.org/10.3389/fmicb.2017.00123
Hoban DJ, Zhanel GG (2013) Introduction to the CANWARD study (2007-11). J Antimicrob Chemother 68(Suppl 1):i3–i5
Jorge P, Perez-Perez M, Perez Rodriguez G et al (2017) A network perspective on antimicrobial peptide combination therapies: the potential of colistin, polymyxin B and nisin. Int J Antimicrob Agents 49:668–676. https://doi.org/10.1016/j.ijantimicag.2017.02.012
Juhaniewicz-Dębińska J, Tymecka D, Sęk S (2018) Lipopeptide-induced changes in permeability of solid supported bilayers composed of bacterial membrane lipids. J Electroanal Chem 812:227–234. https://doi.org/10.1016/j.jelechem.2017.12.065
Makovitzki A, Avrahami D, Shai Y (2006) Ultrashort antibacterial and antifungal lipopeptides. Proc Natl Acad Sci USA 103:15997–16002. https://doi.org/10.1073/pnas.0606129103
Masuda N, Sakagawa E, Ohya S et al (2000) Substrate specificities of MexAB-OprM, MexCD-OprJ, and MexXY-oprM efflux pumps in Pseudomonas aeruginosa. Antimicrob Agents Chemother 44:3322–3327
Meletiadis J, Pournaras S, Roilides E, Walsh TJ (2010) Defining fractional inhibitory concentration index cutoffs for additive interactions based on self-drug additive combinations, Monte Carlo simulation analysis, and in vitro-in vivo correlation data for antifungal drug combinations against Aspergillus fumi. Antimicrob Agents Chemother 54:602–609. https://doi.org/10.1128/AAC.00999-09
Naghmouchi K, Le Lay C, Baah J, Drider D (2012) Antibiotic and antimicrobial peptide combinations: synergistic inhibition of Pseudomonas fluorescens and antibiotic-resistant variants. Res Microbiol 163:101–108. https://doi.org/10.1016/j.resmic.2011.11.002
Park Y, Park SN, Park S-C et al (2006) Synergism of Leu-Lys rich antimicrobial peptides and chloramphenicol against bacterial cells. Biochim Biophys Acta 1764:24–32. https://doi.org/10.1016/j.bbapap.2005.10.019
Schwarz S, Kehrenberg C, Doublet B, Cloeckaert A (2004) Molecular basis of bacterial resistance to chloramphenicol and florfenicol. FEMS Microbiol Rev 28:519–542. https://doi.org/10.1016/j.femsre.2004.04.001
Soren O, Brinch KS, Patel D et al (2015) Antimicrobial peptide novicidin synergizes with rifampin, ceftriaxone, and ceftazidime against antibiotic-resistant Enterobacteriaceae in vitro. Antimicrob Agents Chemother 59:6233–6240. https://doi.org/10.1128/AAC.01245-15
Steenbergen JN, Mohr JF, Thorne GM (2009) Effects of daptomycin in combination with other antimicrobial agents: a review of in vitro and animal model studies. J Antimicrob Chemother 64:1130–1138. https://doi.org/10.1093/jac/dkp346
The Clinical and Laboratory Standards Institute (2016) Performance standards for antimicrobial susceptibility testing CLSI supplement M100S, 26th edn. Clin. Lab. Stand Institute, Wayne, PA
World Health Organization (2015) The selection and use of essential medicines. Report of the WHO Expert Committee, 2015 (including the 19th WHO Model List of Essential Medicines and the 5th WHO Model List of Essential Medicines for Children)
Zhanel GG, DeCorby M, Laing N et al (2008) Antimicrobial-resistant pathogens in intensive care units in Canada: results of the Canadian National Intensive Care Unit (CAN-ICU) study, 2005-2006. Antimicrob Agents Chemother 52:1430–1437. https://doi.org/10.1128/AAC.01538-07
Acknowledgements
This work was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) and the Manitoba Health Research Council (MHRC). We thank Dr. A. Kumar (University of Manitoba) for generously providing the efflux-deficient P. aeruginosa PAO200 and PAO750 strains. We thank Dr. Richard Hodges (University of Manitoba) for generously providing fresh red blood cells from pigs in their Animal Care and Veterinary Services facility.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
All procedures performed in studies involving animals were in accordance with the ethical standards of the University of Manitoba which the studies were conducted.
Informed consent
Informed consent was obtained from all the individual participants included in the study.
Additional information
Handling Editor: J. Metcalf.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Domalaon, R., Brizuela, M., Eisner, B. et al. Dilipid ultrashort cationic lipopeptides as adjuvants for chloramphenicol and other conventional antibiotics against Gram-negative bacteria. Amino Acids 51, 383–393 (2019). https://doi.org/10.1007/s00726-018-2673-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-018-2673-9