Abstract
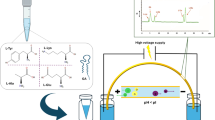
Qualitative and quantitative determination of amino acid composition using amino acid analysis (AAA) is an important quality attribute and considered an identity of therapeutic peptide drugs by the regulatory agencies. Although huge literature is available on pre- and post- column derivatization AAA methods, arriving at an appropriate hydrolysis protocol coupled with adequate separation of the derivatized/underivatized amino acids is always challenging. Towards achieving a facile and comprehensive protocol for AAA, the present work is geared towards developing a deeper understanding of the extent of hydrolysis of peptide, and the nature and stability of amino acids present in the peptide backbone. This defines the suitability of the method in meeting the end goals and the regulatory requirement. Analysis of historical data generated during the method optimization of AAA for icatibant acetate (ICT) using head space oven hydrolysis (HSOH) and microwave-assisted hydrolysis (MAH) methods helped in arriving at fast (< 1 h) and efficient hydrolysis (0.9–1.1 of theoretical residue) conditions. Better separations for the natural and unnatural amino acids were achieved using 3.45 ≤ pH ≤ 10.85, and a column oven gradient program. This approach was useful in meeting the method quality attributes [resolution (Rs) > 2.0; plate count (N) > 5600; and USP tailing factor < 1.2] with a target analytical method profile of relative amino acid mole ratios (RAAMR) in the range of 0.9–1.1 for Ser, Oic, Tic, Hyp, Ala (Thi), Gly and Pro, and between 2.7 and 3.3 for Arg. The developed method was validated as per the ICH guidelines and is precise, accurate, linear and robust.







Similar content being viewed by others
Abbreviations
- AAA:
-
Amino acid analysis
- ICT:
-
Icatibant acetate
- HSOH:
-
Head space oven hydrolysis
- MAH:
-
Microwave-assisted hydrolysis
- RAAMR:
-
Relative amino acid mole ratios
- R s :
-
Resolution
- USP:
-
United States pharmacopeia
- Ser:
-
Serine
- Gly:
-
Glycine
- Ala(thi):
-
Thienylalanine
- Arg:
-
Arginine
- Hyp:
-
Hydroxy-proline
- Pro:
-
Proline
- Oic:
-
Octahydro-1H-indole-2-carboxylic acid
- Tic:
-
D-1, 2, 3, 4-Tetrahydroisoquinoline-3-carboxylic acid
- TP:
-
Therapeutic peptides
- NMR:
-
Nuclear magnetic resonance spectroscopy
- IUPAC:
-
International Union of Pure and Applied Chemists
- CHMP:
-
Committee for Medicinal Products for Human Use
- Pr-CDT:
-
Pre-column derivatization
- Po-CDT:
-
Post-column derivatization
- RSM:
-
Response surface methodology
- F-moc:
-
9-Fluorenylmethyl-chloroformate
- OPA:
-
Orthophthalaldehyde
- PITC:
-
Phenylisothiocyanate
- Tf:
-
Peak tailing
- Rs:
-
Resolution
- N:
-
Theoretical plate count
- FOM:
-
Figure of merit
- EOH:
-
Extent of hydrolysis
- HSGC:
-
Head space gas chromatography
- GC:
-
Gas chromatography
- ICH:
-
International Conference on Harmonization
- RSD:
-
Relative standard deviation
References
Adriaens P, Meesschaert B, Wuyts W, Vaderhaeghe H, Eyssen H (1977) Separation of ninhydrin-positive compounds on a single-column amino acid analyzer using lithium buffers. J Chromatogr 140:103–108
Akhlaghi Y, Ghaffari S, Attar H, Alamir HA (2015) A rapid hydrolysis method and DABS-Cl derivatization for complete amino acid analysis of octreotide acetate by reversed phase HPLC. Amino Acids 47(11):2255–2263
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Blundell G, Brydon WG (1987) High performance liquid chromatography of plasma aminoacids using orthophthalaldehyde derivatisation. Clin Chim Acta 170(1):79–83
Buiarelli F, Gallo V, Di Filippo P, Pomata D, Riccardi C (2013) Development of a method for the analysis of underivatized amino acids by liquid chromatography/tandem mass spectrometry: application on Standard Reference Material 1649a (urban dust). Talanta 115:966–972
Campanella L, Crescentini G, Avino P (1999) Simultaneous determination of cysteine, cystine and 18 other amino acids in various matrices by high-performance liquid chromatography. J Chromatogr A 833(2):137–145
Chinard FP (1952) Photometric estimation of proline and ornithine. J Biol Chem 199(1):91–95
Csapó J, Albert C, Lóki K, Csapó-Kiss Z (2008) Separation and determination of the amino acids by ion exchange column chromatography applying postcolumn derivatization. Acta Universitatis Sapientiae Alimentaria 1(5–2):9
Di L (2015) Strategic approaches to optimizing peptide ADME properties. AAPS J 17(1):134–143
Duncan M, Poljak A (1998) AAA of peptides and proteins on the femtomole scale by gas chromatography/mass spectrometry. Anal Chem 70:890–896
Eggen I, Gregg B, Verlander M, Swietlow A, Rode H, Szajek A (2014) Control strategies for synthetic therapeutic peptide APIs—part I: analytical consideration. BioPharm Int 27(3):16–21
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1):70–77
Fekkes D, Voskuilen-Kooyman A, Jankie R, Huijmans J (2000) Precise analysis of primary amino acids in urine by an automated high-performance liquid chromatography method: comparison with ion-exchange chromatography. J Chromatogr B Biomed Sci Appl 744(1):183–188
Fierabracci V, Masiello P, Novelli M, Bergamini E (1991) Application of amino acid analysis by high-performance liquid chromatography with phenyl isothiocyanate derivatization to the rapid determination of free amino acids in biological samples. J Chromatogr 570(2):285–291
Fosgerau K, Hoffmann T (2015) Peptide therapeutics: current status and future directions. Drug Discov Today 20(1):122–128
Friedmann M (2004) Applications of the ninhydrin reaction for analysis of amino acids, peptides and proteins to agricultural and biomedical sciences. J Agric Food Chem 52:385–406
Fürst P, Pollack L, Graser TA, Godel H, Stehle P (1990) Appraisal of four pre-column derivatization methods for the high-performance liquid chromatographic determination of free amino acids in biological materials. J Chromatogr 499:557–569
Guthals A, Bandeira N (2012) Peptide identification by tandem mass spectrometry with alternate fragmentation modes. Mol Cell Proteom 11(9):550–557
Heftmann E (2004) Chromatography: fundamentals and applications of chromatography and related differential migration methods—part A: fundamentals and techniques, vol 69A, 6th edn. Elsevier Science, Amsterdam, Netherlands
Hesse A, Weller MG (2016) Protein quantification by derivatization-free high-performance liquid chromatography of aromatic amino acids. J Amino Acids 2016:7374316
Hsieh HP, Wu YT, Chen ST, Wang KT (1999) Direct solid-phase synthesis of octreotide conjugates: precursors for use as tumor-targeted radiopharmaceuticals. Bioorg Med Chem Lett 7:1797–1803
ICH Q2A Text on Validation of Analytical Procedures (1995) In: International conference on harmonization of technical requirements for registration of pharmaceuticals for human use, Geneva, Switzerland, 1995
Ishida Y, Fujita T, Asai K (1981) New detection and separation method for amino acids by high-performance liquid chromatography. J Chromatogr 204:143–148
Kataoka H, Tanaka H, Fujimoto A, Noguchi I, Makita M (1994) Determination of sulphur amino acids by gas chromatography with flame photometric detection. Biomed Chromatogr 8(3):119–124
Kataoka H, Matsumura S, Makita M (1997) Determination of amino acids in biological fluid by capillary gas chromatography with nitrogen phosphorus selective detection. J Pharm Biomed Anal 15:1271–1279
Lau JL, Dunn MK (2018) Therapeutic peptides: historical perspectives, current development trends, and future directions. Bioorg Med Chem 26(10):2700–2707
Le Boucher J, Charret C, Coudray-Lucas C, Giboudeau J, Cynober L (1997) Amino acid determination in biological fluids by automated ion-exchange chromatography: performance of Hitachi L-8500A. Clin Chem 43(8):1421–1428
Lewis AM, Waterhouse C, Jacob LS (1980) Whole blood and plasma amino acid analysis- Gas chromatography and ion exchange chromatography compared. Clin Chem 26(2):271–276
Lobas AA, Verenchikov AN, Goloborodko AA, Levitsky LI, Gorshkov MV (2013) Combination of Edman degradation of peptides with liquid chromatography/mass spectrometry workflow for peptide identification in bottom-up proteomics. Rapid Commun Mass Spectrom 27(3):391–400
Matsumura S, Kataoka H, Makita M (1996) Determination of amino acids in human serum by capillary gas chromatography with nitrogen phosphorus selective detection. J Chromatogr B 681:375–380
Moodie IM, Hough BJ, Labadarios D (1989) Determination of amino acids in urine by gas chromatography. J Sep Sci 12(7):437–441
Moore S, Stein WH (1948) Photometric ninhydrin method for use in the chromatography of amino acids. J Biol Chem 176(1):367–388
Moore S, Stein WH (1951) Chromatography of amino acids on sulfonated polystyrene resins. J Biol Chem 192(2):663–681
Moore S, Stein WH (1958) Chromatographie determination of amino acids by the use of automatic recording equipment. Methods Enzymol 6:819–831
Nakano Y, Konya Y, Taniguchi M, Fukusaki E (2017) Development of a liquid chromatography-tandem mass spectrometry method for quantitative analysis of trace d-amino acids. J Biosci Bioeng 123(1):134–138
Ng LT, Anderson GH (1998) Analysis of O-phthaldialdehyde precoloumn derivatized physiological amino acids. Malays Appl Biol 27(1–2):13–20
Omar MMA, Elbashir AA, Schmitz OJ (2017) Capillary electrophoresis method with UV-detection for analysis of free amino acids concentrations in food. Food Chem 214:300–307
Qureshi GA, Qureshi AR (1989) Determination of free amino acids in biological samples: problems of quantitation. J Chromatogr 491(2):281–289
Qvit N, Rubin SJS, Urban TJ, Mochly-Rosen D, Gross ER (2017) Peptidomimetic therapeutics: scientific approaches and opportunities. Drug Discov Today 22(2):454–462
Reason AJ (2003) Validation of amino acid analysis methods. Methods Mol Biol 211:181–194
Rigas PG (2013) Post-column labeling techniques in amino acid analysis by liquid chromatography. Anal Bioanal Chem 405(25):7957–7992
Rutherfurd SM, Gilani GS (2009) Amino acid analysis. Curr Protoc Protein Sci 58(1):11.9.1–11.9.37
Skelley AM, Mathies RA (2003) Chiral separation of fluorescamine-labeled amino acids using microfabricated capillary electrophoresis devices for extraterrestrial exploration. J Chromatogr A 1021:191–199
Troll W, Lindsley J (1955) A photometric method for the determination of proline. J Biol Chem 215:655–660
Wainer A (1967) The chromatography of ninhydrin negative compounds on an amino acid analyzer column. J Chromatogr 26(1):48–53
Williamson MP (1993) Peptide structure determination by NMR. Methods Mol Biol 17:69–85
Wu LC (2013) Regulatory considerations for peptide drug products. In: Paper presented at the 2013 GPhA/FDA CMC Workshop
Xia T, Gao S, Shu C, Wen Y, Yun Y, Tao X, Chen W, Zhang F (2016) Analysis of amino acids in human blood using UHPLC-MS/MS: potential interferences of storage time and vacutainer tube in pre-analytical procedure. Clin Biochem 49(18):1372–1378
Zahou E, Jornvall H, Bergman T (2000) AAA by capillary electrophoresis after phenylthiocarbamylation. Anal Biochem 281:115–122
Zhang L, Li Y, Zhou H, Li L, Wang Y, Zhang Y (2012) Determination of eight amino acids in mice embryonic stem cells by pre-column derivatization HPLC with fluorescence detection. J Pharm Biomed Anal 66:356–358
Zhao XE, You JM, Liu HZ, Suo YR (2007) Pre-column derivatization high performance liquid chromatography tandem mass spectrometric determination of trace level of amino acids in rat serum. Chin J Anal Chem 35(7):938–944
Zhong H, Marcus SL, Li L (2005) Microwave-assisted acid hydrolysis of proteins combined with liquid chromatography MALDI MS/MS for protein identification. J Am Soc Mass Spectrom 16(4):471–481
Zunić G, Spasić S (2008) Capillary electrophoresis method optimized with a factorial design for the determination of glutathione and amino acid status using human capillary blood. J Chromatogr B Anal Technol Biomed Life Sci 873(1):70–76
Acknowledgements
Two of the authors (KYKK and DVGR) thank the management of Dr. Reddy’s Laboratories Ltd., Hyderabad, India, for supporting this work. Support from colleagues in Research and Development, and Analytical Research and Development of Dr. Reddy’s Laboratories Ltd. is appreciated.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors Yagnakirankumar Komaravolu, Venugopala Rao Dama, and Thirumala Chary Maringanti declare that they have no conflicts of interest.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Handling Editor: F. Albericio.
Rights and permissions
About this article
Cite this article
Komaravolu, Y., Dama, V.R. & Maringanti, T.C. Novel, efficient, facile, and comprehensive protocol for post-column amino acid analysis of icatibant acetate containing natural and unnatural amino acids using the QbD approach. Amino Acids 51, 295–309 (2019). https://doi.org/10.1007/s00726-018-2665-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-018-2665-9