Abstract
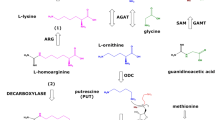
l-Homoarginine (hArg) and guanidinoacetate (GAA) are produced from l-arginine (Arg) by the catalytic action of arginine:glycine amidinotransferase. Guanidinoacetate methyltransferase methylates GAA on its non-guanidine N atom to produce creatine. Arg and hArg are converted by nitric oxide synthase (NOS) to nitric oxide (NO). NO is oxidized to nitrite and nitrate which circulate in the blood and are excreted in the urine. Asymmetric dimethylarginine (ADMA), an NOS inhibitor, is widely accepted to be exclusively produced after asymmetric N G-methylation of Arg residues in proteins and their regular proteolysis. Low circulating and urinary hArg concentrations and high circulating concentrations of ADMA emerged as risk markers in the human renal and cardiovascular systems. While ADMA’s distribution and metabolism are thoroughly investigated, such studies on hArg are sparse. The aim of the present pilot study was to investigate the distribution of exogenous hArg in plasma, liver, kidney, lung, and heart in a rat model of takotsubo cardiomyopathy (TTC). hArg hydrochloride solutions in physiological saline were injected intra-peritoneally at potentially pharmacological, non-toxic doses of 20, 220, or 440 mg/kg body weight. Vehicle (saline) served as control. As hArg has been reported to be a pro-oxidant, plasma and tissue malondialdehyde (MDA) was measured as a biomarker of lipid peroxidation. hArg administration resulted in dose-dependent maximum plasma hArg concentrations and distribution in all investigated organs. hArg disappeared from plasma with an elimination half-life ranging between 20 and 40 min. hArg administration resulted in relatively small changes in the plasma and tissue content of Arg, GAA, ADMA, creatinine, and of the NO metabolites nitrite and nitrate. Remarkable changes were observed for tissue GAA, notably in the kidney. Plasma and tissue MDA concentration did not change upon hArg administration, suggesting that even high-dosed hArg is not an oxidant. The lowest hArg dose of 20 mg/kg bodyweight increased 25-fold the mean hArg maximum plasma concentration. This hArg dose seems to be useful as the upper limit in forthcoming studies on the putative cardioprotective effects of hArg in our rat model of TTC.





Similar content being viewed by others
Abbreviations
- ADMA:
-
Asymmetric dimethylarginine (N G,N G-dimethyl-l-arginine)
- AGAT:
-
Arginine:glycine amidinotransferase
- DDAH:
-
Dimethylarginine dimethylaminohydrolase
- GAA:
-
Guanidinoacetate
- GAMT:
-
Guanidinoacetate methyltransferase
- GC–MS:
-
Gas chromatography–mass spectrometry
- hArg:
-
Homoarginine
- MDA:
-
Malondialdehyde
- N G :
-
Guanidine nitrogen
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- PBS:
-
Phosphate buffered saline
- SIC:
-
Stress-induced cardiomyopathy
- TTC:
-
Takotsubo cardiomyopathy
References
Alessandrì MG, Celati L, Battini R, Casarano M, Cioni G (2005) Gas chromatography/mass spectrometry assay for arginine: glycine-amidinotransferase deficiency. Anal Biochem 343:356–358
Alesutan I, Feger M, Tuffaha R, Castor T, Musculus K, Buehling SS et al (2016) Augmentation of phosphate-induced osteo-/chondrogenic transformation of vascular smooth muscle cells by homoarginine. Cardiovasc Res 110:408–418
Atzler D, Schönhoff M, Cordts K, Ortland I, Hoppe J, Hummel FC, Gerloff C, Jaehde U, Jagodzinski A, Böger RH, Choe CU, Schwedhelm E (2016) Oral supplementation with l-homoarginine in young volunteers. Br J Clin Pharmacol 82:1477–1485
Bernstein HG, Jäger K, Dobrowolny H, Steiner J, Keilhoff G, Bogerts B, Laube G (2015) Possible sources and functions of l-homoarginine in the brain: review of the literature and own findings. Amino Acids 47:1729–1740
Bode-Böger SM (2006) Effect of l-arginine supplementation on NO production in man. Eur J Clin Pharmacol 62:91–99
Cathelineau L, Saudubray JM, Charpentier C, Polonovski C (1974) Letter: the presence of the homoanalogues of substrates of the urea cycle in the presence of argininosuccinate synthetase deficiency. Pediatr Res 8:857
Chappell JB (1963) The effect of alkylguanidines on mitochondrial metabolism. J Biol Chem 238:410–417
Choe CU, Atzler D, Wild PS, Carter AM, Boger RH, Ojeda F et al (2013a) Homoarginine levels are regulated by arginine:glycine amidinotransferase and affect stroke outcome: results from human and murine studies. Circulation 128:1451–1461
Choe CU, Nabuurs C, Stockebrand MC, Neu A, Nunes P, Morellini F et al (2013b) Arginine:glycine amidinotransferase deficiency protects from metabolic syndrome. Hum Mol Genet 22:110–123
Cullen ME, Yuen AH, Felkin LE, Smolenski RT, Hall JL, Grindle S et al (2006) Myocardial expression of the arginine:glycine amidinotransferase gene is elevated in heart failure and normalized after recovery: potential implications for local creatine synthesis. Circulation 114:I16–I20
Davids M, Ndika JD, Salomons GS, Blom HJ, Teerlink T (2012) Promiscuous activity of arginine:glycine amidinotransferase is responsible for the synthesis of the novel cardiovascular risk factor homoarginine. FEBS Lett 586:3653–3657
Dellera F, Ganzetti GS, Froio A, Manzini S, Busnelli M, Meinitzer A, Sirtori CR, Chiesa G, Parolini C (2016) l-Homoarginine administration reduces neointimal hyperplasia in balloon-injured rat carotids. Thromb Haemost 116:400–402
Gambaryan S, Tsikas D (2015) A review and discussion of platelet nitric oxide and nitric oxide synthase: do blood platelets produce nitric oxide from l-arginine or nitrite? Amino Acids 47:1779–1793
Hanff E, Böhmer A, Zinke M, Gambaryan S, Schwarz A, Supuran CT et al (2016a) Carbonic anhydrases are producers of S-nitrosothiols from inorganic nitrite and modulators of soluble guanylyl cyclase in human platelets. Amino Acids 48:1695–1706
Hanff E, Kayacelebi AA, Yanchev GR, Maassen N, Haghikia A, Tsikas D (2016b) Simultaneous stable-isotope dilution GC–MS measurement of homoarginine, guanidinoacetate and their common precursor arginine in plasma and their interrelationships in healthy and diseased humans. Amino Acids 48:721–732
Hanff E, Lützow M, Kayacelebi AA, Finkel A, Maassen M, Yanchev GR, Haghikia A, Bavendiek U, Buck A, Lücke T, Maassen N, Tsikas D (2017) Simultaneous GC–ECNICI–MS measurement of nitrite, nitrate and creatinine in human urine and plasma in clinical settings. J Chromatogr B 1047:207–2014
Hibbs JB Jr, Westenfelder C, Taintor R, Vavrin Z, Kablitz C, Baranowski RL, Ward JH, Menlove RL, McMurry MP, Kushner JP, Samlowski WE (1992) Evidence for cytokine-inducible nitric oxide synthesis from l-arginine in patients receiving interleukin-2 therapy. J Clin Invest 89:867–877
Holecek M, Sispera L (2016) Effects of arginine supplementation on amino acid profiles in blood and tissues in fed and overnight-fasted rats. Nutrients 8:206
Hollunger G (1955) Guanidines and oxidative phosporylations. Acta Pharmacol Toxicol 11(Suppl 1):1–84
Hou Y, Hu S, Jia S, Nawaratna G, Che D, Wang F et al (2016) Whole-body synthesis of l-homoarginine in pigs and rats supplemented with l-arginine. Amino Acids 48:993–1001
Jones JD, Burnett PC (1972) Implication of creatinine and gut flora in the uremic syndrome: induction of “creatininase” in colon contents of the rat by dietary creatinine. Clin Chem 18:280–284
Kato T, Sano M, Mizutani N, Hayakawa C (1988) Homocitrullinuria and homoargininuria in hyperargininaemia. J Inherit Metab Dis 11:261–265
Kayacelebi AA, Nguyen TH, Neil C, Horowitz JD, Jordan J, Tsikas D (2014) Homoarginine and 3-nitrotyrosine in patients with takotsubo cardiomyopathy. Int J Cardiol 173:546–547
Kayacelebi AA, Langen J, Weigt-Usinger K, Chobanyan-Jurgens K, Mariotti F, Schneider JY et al (2015a) Biosynthesis of homoarginine (hArg) and asymmetric dimethylarginine (ADMA) from acutely and chronically administered free l-arginine in humans. Amino Acids 47:1893–1908
Kayacelebi AA, Knöffel AK, Beckmann B, Hanff E, Warnecke G, Tsikas D (2015b) Measurement of unlabeled and stable isotope-labeled homoarginine, arginine and their metabolites in biological samples by GC–MS and GC–MS/MS. Amino Acids 47:2023–2034
Lacza Z, Kozlov AV, Pankotai E, Csordás A, Wolf G, Redl H, Kollai M, Szabó C, Busija DW, Horn TF (2006a) Mitochondria produce reactive nitrogen species via an arginine-independent pathway. Free Radic Res 40:369–378
Lacza Z, Pankotai E, Csordás A, Gero D, Kiss L, Horváth EM, Kollai M, Busija DW, Szabó C (2006b) Mitochondrial NO and reactive nitrogen species production: does mtNOS exist? Nitric Oxide 14:162–168
Levin B, Oberholzer VG, Palmer T (1974) Letter: the high levels of lysine, homocitrulline, and homoarginine found in argininosuccinate synthetase deficiency. Pediatr Res 8:857–858
Mansuy D, Boucher JL (2004) Alternative nitric-oxide producing substrates for NO synthases. Free Radic Biol Med 37:1105–1121
März W, Meinitzer A, Drechsler C, Pilz S, Krane V, Kleber ME, Fischer J, Winkelmann BR, Böhm BO, Ritz E, Wanner C (2010) Homoarginine, cardiovascular risk, and mortality. Circulation 122:967–975
McGuire DM, Tormanen CD, Segal IS, Van Pilsum JF (1980) The effect of growth hormone and thyroxine on the amount of l-arginine:glycine amidinotransferase in kidneys of hypophysectomized rats. Purification and some properties of rat kidney transamidinase. J Biol Chem 255:1152–1159
O’Neal RM, Chen CH, Reynolds CS, Meghal SK, Koeppe RE (1968) The ‘neurotoxicity’ of l-2,4-diaminobutyric acid. Biochem J 106:699–706
Pentyala J, Rao SLN (1999) Sustained nitric oxide generation with l-homoarginine. Res Commun Biochem Cell Mol Biol 3:223–232
Pilz S, Meinitzer A, Gaksch M, Grübler M, Verheyen N, Drechsler C, Hartaigh BÓ, Lang F, Alesutan I, Voelkl J, März W, Tomaschitz A (2015) Homoarginine in the renal and cardiovascular systems. Amino Acids 47:1703–1713
Redfors B, Oras J, Shao Y, Seemann-Lodding H, Ricksten SE, Omerovic E (2014) Cardioprotective effects of isoflurane in a rat model of stress-induced cardiomyopathy (takotsubo). Int J Cardiol 176:815–821
Ryan WL, Wells IC (1964) Homocitrulline and homoarginine synthesis from lysine. Science 144:1122–1127
Ryan WL, Barak AJ, Johnson RJ (1968) Lysine, homocitrulline, and homoarginine metabolism by the isolated perfused rat liver. Arch Biochem Biophys 123:294–297
Ryan WL, Johnson RJ, Dimari S (1969) Homoarginine synthesis by rat kidney. Arch Biochem Biophys 131:521–526
Sase A, Nawaratna G, Hu S, Wu G, Lubec G (2016) Decreased hippocampal homoarginine and increased nitric oxide and nitric oxide synthase levels in rats parallel training in a radial arm maze. Amino Acids 48:2197–2204
Sasso S, Dalmedico L, Magro DD, Pereira EM, Wyse AT, de Lima DD (2015) Differential in vitro effects of homoarginine on oxidative stress in plasma, erythrocytes, kidney and liver of rats in the absence and in the presence alpha-tocopherol, ascorbic acid or l-NAME. Amino Acids 47:1931–1939
Schneider JY, Rothmann S, Schröder F, Langen J, Lücke T, Mariotti F, Huneau JF, Frölich JC, Tsikas D (2015) Effects of chronic oral l-arginine administration on the l-arginine/NO pathway in patients with peripheral arterial occlusive disease or coronary artery disease: l-arginine prevents renal loss of nitrite, the major NO reservoir. Amino Acids 47:1961–1974
Stockebrand M, Hornig S, Neu A, Atzler D, Cordts K, Böger RH et al (2015) Homoarginine supplementation improves blood glucose in diet-induced obese mice. Amino Acids 47:1921–1927
Tsikas D (2000) Simultaneous derivatization and quantification of the nitric oxide metabolites nitrite and nitrate in biological fluids by gas chromatography/mass spectrometry. Anal Chem 72:4064–4072
Tsikas D (2015) Circulating and excretory nitrite and nitrate: their value as measures of nitric oxide synthesis, bioavailability and activity is inherently limited. Nitric Oxide 45:1–3
Tsikas D (2017) Assessment of lipid peroxidation by measuring malondialdehyde (MDA) and relatives in biological samples: analytical and biological challenges. Anal Biochem 524:13–30
Tsikas D, Kayacelebi AA (2014) Do homoarginine and asymmetric dimethylarginine act antagonistically in the cardiovascular system? Circ J 78:2094–2095
Tsikas D, Böger RH, Sandmann J, Bode-Böger SM, Frölich JC (2000) Endogenous nitric oxide synthase inhibitors are responsible for the l-arginine paradox. FEBS Lett 478:1–3
Tsikas D, Rothmann S, Schneider JY, Gutzki FM, Beckmann B, Frölich JC (2017) Simultaneous measurement of malondialdehyde (MDA) and 4-hydroxy-2-nonenal in human plasma: effects of long-term l-arginine addministration. Anal Biochem 524:31–44
Tsikas D, Wu G (2015) Homoarginine, arginine, and relatives: analysis, metabolism, tcharansport, physiology, and pathology. Amino Acids 47:1697–1702
Verhoeven NM, Schor DS, Roos B, Battini R, Stöckler-Ipsiroglu S, Salomons GS, Jakobs C (2003) Diagnostic enzyme assay that uses stable-isotope-labeled substrates to detect l-arginine:glycine amidinotransferase deficiency. Clin Chem 49:803–805
Vieira RP, Duarte AC, Claudino RC, Perini A, Santos AB, Moriya HT, Arantes-Costa FM, Martins MA, Carvalho CR, Dolhnikoff M (2007) Creatine supplementation exacerbates allergic lung inflammation and airway remodeling in mice. Am J Respir Cell Mol Biol 37:660–667
Wu G, Bazer FW, Davis TA, Kim SW, Li P, Marc Rhoads J et al (2009) Arginine metabolism and nutrition in growth, health and disease. Amino Acids 37:153–168
Wyse AT, Bavaresco CS, Hagen ME, Delwing D, Wannmacher CM, Severo Dutra-Filho C et al (2001) In vitro stimulation of oxidative stress in cerebral cortex of rats by the guanidino compounds accumulating in hyperargininemia. Brain Res 923:50–57
Yamashita K, Nakajima Y, Matsushita H, Nishiya Y, Yamazawa R, Wu YF, Matsubara F, Oyama H, Ito K, Yoshimoto T (2010) Substitution of Glu122 by glutamine revealed the function of the second water molecule as a proton donor in the binuclear metal enzyme creatininase. J Mol Biol 396:1081–1096
Yang Y, Wu Z, Jia S, Dahanayaka S, Feng S, Meininger CJ, McNeal CJ, Wu G (2015) Safety of long-term dietary supplementation with l-arginine in rats. Amino Acids 47:1909–1920
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical statement
Rats received humane care and the study protocol complied with the institutional guidelines of the Sahlgrenska University Hospital.
Additional information
Handling Editors: C.-A. A. Hu, Y. Yin, Y. Hou, G. Wu, Y. Teng.
Rights and permissions
About this article
Cite this article
Günes, D.N., Kayacelebi, A.A., Hanff, E. et al. Metabolism and distribution of pharmacological homoarginine in plasma and main organs of the anesthetized rat. Amino Acids 49, 2033–2044 (2017). https://doi.org/10.1007/s00726-017-2465-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-017-2465-7