Abstract
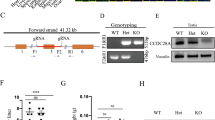
Ornithine decarboxylase antizyme 1 (OAZ1) regulates both polyamine biosynthesis and transport and acts as a tumor suppressor because of its functions in the regulation of cell proliferation, apoptosis and growth. However, the roles of OAZ1 in the regulation of polyamine metabolism, cell proliferation, apoptosis and hormone receptor expression in granulosa cells (GCs) from geese have not been determined. To define the roles of OAZ1 in primary GCs, we constructed and transfected an Oaz1 overexpression vector with a thymidine deletion into primary GCs. Oaz1 overexpression induced 65 and 30% decreases in the putrescine and spermidine contents, respectively, 225% increase in the spermine content and up-regulated the expression levels of genes associated with polyamine metabolism (but Oaz2). The Ccnd1, Pcna, Bax and Caspase 3 mRNA expression levels in GCs overexpressing Oaz1 were significantly higher than the expression levels in the vehicle group (p < 0.05). Additionally, a slight increase in the E2 concentration in the culture medium and enhanced Er, Fshr and Lhr expression were observed in the GCs 24 h after Oaz1 overexpression (p < 0.05). These data suggested that Oaz1 up-regulated the expression of genes related to polyamine metabolic enzymes and reproductive hormone receptors, disturbed intracellular polyamine homeostasis, and affected the transcription of genes associated to cell proliferation and apoptosis in goose primary GCs. Taken together, our results indicate that modifying polyamine metabolism by Oaz1 may interfere with the expression of genes involved in cell proliferation, apoptosis and responsiveness of ovary toward hormones in goose GCs.





Similar content being viewed by others
Abbreviations
- AZIN:
-
Ornithine decarboxylase antizyme inhibitor
- OAZ:
-
Ornithine decarboxylase antizyme
- ODC:
-
Ornithine decarboxylase
- DFMO:
-
α-Difluoromethylornithine
- SAMDC:
-
S-adenosylmethionine decarboxylase
- SPDS:
-
Spermidine synthase
- SPMS:
-
Spermine synthase
- SSAT:
-
Spermidine/spermine-N1-acetyltransferase
- APAO:
-
N1-acetylpolyamine oxidase
- SMO:
-
Spermine oxidase
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GC:
-
Granulosa cell
References
Acosta-Andrade C, Lambertos A, Urdiales JL, Sanchez-Jimenez F, Penafiel R, Fajardo I (2016) A novel role for antizyme inhibitor 2 as a regulator of serotonin and histamine biosynthesis and content in mouse mast cells. Amino Acids 48:2411–2421
An XP et al (2012) Analysis of differentially expressed genes in ovaries of polytocous versus monotocous dairy goats using suppressive subtractive hybridization. Reprod Domest Anim 47:498–503
Bastida CM, Cremades A, Castells MT, Lopez-Contreras AJ, Lopez-Garcia C, Tejada F, Penafiel R (2005) Influence of ovarian ornithine decarboxylase in folliculogenesis and luteinization. Endocrinology 146:666–674
Coffino P (2001) Regulation of cellular polyamines by antizyme. Nat Rev Mol Cell Biol 2:188–194
Cui PF et al (2016) Polyamine metabolism-based dual functional gene delivery system to synergistically inhibit the proliferation of cancer. Int J Pharm 506:79–86
Dewailly D, Robin G, Peigne M, Decanter C, Pigny P, Catteau-Jonard S (2016) Interactions between androgens, FSH, anti-Mullerian hormone and estradiol during folliculogenesis in the human normal and polycystic ovary. Hum Reprod Update 22:709–724
Feith DJ, Origanti S, Shoop PL, Sass-Kuhn S, Shantz LM (2006) Tumor suppressor activity of ODC antizyme in MEK-driven skin tumorigenesis. Carcinogenesis 27:1090–1098
Feith DJ, Shantz LM, Shoop PL, Keefer KA, Prakashagowda C, Pegg AE (2007) Mouse skin chemical carcinogenesis is inhibited by antizyme in promotion-sensitive and promotion-resistant genetic backgrounds. Mol Carcinog 46:453–465
Gilbert AB, Evans AJ, Perry MM, Davidson MH (1977) A method for separating the granulosa cells, the basal lamina and the theca of the preovulatory ovarian follicle of the domestic fowl (Gallus domesticus). J Reprod Fertil 50:179–181
Gruendler C, Lin Y, Farley J, Wang T (2001) Proteasomal degradation of Smad1 induced by bone morphogenetic proteins. J Biol Chem 276:46533–46543
Hayashi S, Murakami Y, Matsufuji S (1996) Ornithine decarboxylase antizyme: a novel type of regulatory protein. Trends Biochem Sci 21:27–30
He H, Kang B, Jiang D, Ma R, Bai L (2014) Molecular cloning and mRNA expression analysis of ornithine decarboxylase antizyme 2 in ovarian follicles of the Sichuan white goose (Anser cygnoides). Gene 545:247–252
Huang Y et al (2006) Polyamine analogues down-regulate estrogen receptor alpha expression in human breast cancer cells. J Biol Chem 281:19055–19063
Kahana C (2009) Antizyme and antizyme inhibitor, a regulatory tango. Cell Mol Life Sci 66:2479–2488
Kang B, Guo JR, Yang HM, Zhou RJ, Liu JX, Li SZ, Dong CY (2009) Differential expression profiling of ovarian genes in prelaying and laying geese. Poult Sci 88:1975–1983
Kang B, Jiang DM, Bai L, He H, Ma R (2014) Molecular characterisation and expression profiling of the ENO1 gene in the ovarian follicle of the Sichuan white goose. Mol Biol Rep 41:1927–1935
Katayama H, Zhou H, Li Q, Tatsuka M, Sen S (2001) Interaction and feedback regulation between STK15/BTAK/Aurora-A kinase and protein phosphatase 1 through mitotic cell division cycle. J Biol Chem 276:46219–46224
Keren-Paz A, Bercovich Z, Porat Z, Erez O, Brener O, Kahana C (2006) Overexpression of antizyme-inhibitor in NIH3T3 fibroblasts provides growth advantage through neutralization of antizyme functions. Oncogene 25:5163–5172
Kogo H, Johnson DC, Dey SK, Takeo S (1993) A comparison of the effects of estradiol and 2- and 4-hydroxyestradiol on uterine ornithine decarboxylase activity in immature rats. Jpn J Pharmacol 61:65–67
Koike C, Chao DT, Zetter BR (1999) Sensitivity to polyamine-induced growth arrest correlates with antizyme induction in prostate carcinoma cells. Can Res 59:6109–6112
Kurian L, Palanimurugan R, Godderz D, Dohmen RJ (2011) Polyamine sensing by nascent ornithine decarboxylase antizyme stimulates decoding of its mRNA. Nature 477:490–494
Lefevre PL, Palin MF, Murphy BD (2011) Polyamines on the reproductive landscape. Endocr Rev 32:694–712
Lewis JS, Vijayanathan V, Thomas TJ, Pestell RG, Albanese C, Gallo MA, Thomas T (2005) Activation of cyclin D1 by estradiol and spermine in MCF-7 breast cancer cells: a mechanism involving the p38 MAP kinase and phosphorylation of ATF-2. Oncol Res 15:113–128
Li WD et al (2015) Involvement of Antizyme Characterized from the Small Abalone Haliotis diversicolor in Gonadal Development. PLoS One 10:e0135251
Lim SK, Gopalan G (2007) Antizyme1 mediates AURKAIP1-dependent degradation of Aurora-A. Oncogene 26:6593–6603
Limon A, Mamdani F, Hjelm BE, Vawter MP, Sequeira A (2016) Targets of polyamine dysregulation in major depression and suicide: activity-dependent feedback, excitability, and neurotransmission. Neurosci Biobehav Rev 66:80–91
Lin Y et al (2002) A novel link between the proteasome pathway and the signal transduction pathway of the bone morphogenetic proteins (BMPs). BMC Cell Biol 3:15
Liu GY et al (2006) Antizyme, a natural ornithine decarboxylase inhibitor, induces apoptosis of haematopoietic cells through mitochondrial membrane depolarization and caspases’ cascade. Apoptosis 11:1773–1788
Ma R, Jiang D, Kang B, Bai L, He H, Chen Z, Yi Z (2015) Molecular cloning and mRNA expression analysis of antizyme inhibitor 1 in the ovarian follicles of the Sichuan white goose. Gene 568:55–60
Mackintosh CA, Feith DJ, Shantz LM, Pegg AE (2000) Overexpression of antizyme in the hearts of transgenic mice prevents the isoprenaline-induced increase in cardiac ornithine decarboxylase activity and polyamines, but does not prevent cardiac hypertrophy. Biochem J 350(Pt 3):645–653
Matsufuji S, Matsufuji T, Miyazaki Y, Murakami Y, Atkins JF, Gesteland RF, Hayashi S (1995) Autoregulatory frameshifting in decoding mammalian ornithine decarboxylase antizyme. Cell 80:51–60
Newman RM et al (2004) Antizyme targets cyclin D1 for degradation. A novel mechanism for cell growth repression. J Biol Chem 279:41504–41511
Ocon-Grove OM, Poole DH, Johnson AL (2012) Bone morphogenetic protein 6 promotes FSH receptor and anti-Mullerian hormone mRNA expression in granulosa cells from hen prehierarchal follicles. Reproduction 143:825–833
Pietila M, Dhungana H, Uimari A, Sironen R, Alhonen L (2014) Systemic overexpression of antizyme 1 in mouse reduces ornithine decarboxylase activity without major changes in tissue polyamine homeostasis. Transgenic Res 23:153–163
Ramani D, De Bandt JP, Cynober L (2014) Aliphatic polyamines in physiology and diseases. Clin Nutr 33:14–22
Ray RM, Bhattacharya S, Bavaria MN, Viar MJ, Johnson LR (2014) Antizyme (AZ) regulates intestinal cell growth independent of polyamines. Amino Acids 46:2231–2239
Swift TA, Dias JA (1986) Effects of the polyamine spermine on binding of follicle-stimulating hormone to membrane-bound immature bovine testis receptors. Biochim Biophys Acta 885:221–230
Tao Y, Liu XJ (2013) Deficiency of ovarian ornithine decarboxylase contributes to aging-related egg aneuploidy in mice. Aging Cell 12:42–49
Thyssen SM, Becu-Villalobos D, Lacau-Mengido IM, Libertun C (1997) alpha-difluoromethylornithine modifies gonadotropin-releasing hormone release and follicle-stimulating hormone secretion in the immature female rat. Proc Soc Exp Biol Med 215:192–197
Wei C et al (2016) Exogenous spermine inhibits hypoxia/ischemia-induced myocardial apoptosis via regulation of mitochondrial permeability transition pore and associated pathways. Exp Biol Med (Maywood) 241:1505–1515
Wilson SM, Hawel L 3rd, Pastorian KE, Byus CV (2005) A stable, inducible, dose-responsive ODC overexpression system in human cell lines. Biochim Biophys Acta 1732:103–110
Wu HY et al (2015) Structural basis of antizyme-mediated regulation of polyamine homeostasis. Proc Natl Acad Sci USA 112:11229–11234
Xiao S et al (2016) Genome-scale RNA interference screen identifies antizyme 1 (OAZ1) as a target for improvement of recombinant protein production in mammalian cells. Biotechnol Bioeng 113:2403–2415
Zhao YC et al (2008) Polyamines are essential in embryo implantation: expression and function of polyamine-related genes in mouse uterus during peri-implantation period. Endocrinology 149:2325–2332
Zhu Q, Jin L, Casero RA, Davidson NE, Huang Y (2012) Role of ornithine decarboxylase in regulation of estrogen receptor alpha expression and growth in human breast cancer cells. Breast Cancer Res Treat 136:57–66
Acknowledgements
This work was financially supported by Grants 31201798 from the National Natural Science Foundation of China and 20105103120003 from the Specialized Research Fund for the Doctoral Program of Higher Education.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This study was funded by Grants 31201798 from the National Natural Science Foundation of China and 20105103120003 from the Specialized Research Fund for the Doctoral Program of Higher Education.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involving human participants and/or animals
This article does not contain any studies that involve human participants. All procedures performed in studies involving animals were in accordance with the ethical standards of the Animal Ethics Committee of the College of Animal Science and Technology, Sichuan Agricultural University or practice at which the studies were conducted.
Additional information
Handling Editor: E. Agostinelli.
B. Kang and D. Jiang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kang, B., Jiang, D., He, H. et al. Effect of Oaz1 overexpression on goose ovarian granulosa cells. Amino Acids 49, 1123–1132 (2017). https://doi.org/10.1007/s00726-017-2411-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-017-2411-8