Abstract
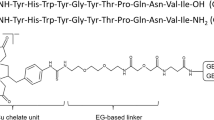
The urokinase-type plasminogen activator receptor (uPAR) is overexpressed in several cancers including glioblastoma (GBM) and is an established biomarker for metastatic potential. The uPAR-targeting peptide AE105-NH2 (Ac-Asp-Cha-Phe-(D)Ser-(D)Arg-Tyr-Leu-Trp-Ser-CONH2) is a promising candidate for non-invasive positron emission tomography (PET) imaging of uPAR. Despite the optimal physical properties of 68Ga for peptide-based PET imaging, low tumor uptakes have previously been reported using 68Ga-labeled AE105-NH2-based tracers. In an attempt to optimize the tumor uptake, we developed three novel tracers with alkane (AOC) and polyethylene glycol (PEG) spacers inserted between AE105-NH2 and the radio metal chelator 2-(4,7-bis(carboxymethyl)-1,4,7-triazonan-1-yl)pentanedioic acid (NODAGA). The resulting tracers NODAGA-AOC-AE105-NH2, NODAGA-PEG3-AE105-NH2 and NODAGA-PEG8-AE105-NH2 were compared to the non-spacer version, NODAGA-AE105-NH2. Following radiolabeling with 68Ga, we evaluated the in vitro and in vivo performance in mice bearing subcutaneous tumors derived from the uPAR-expressing human GBM cell line U87MG. In vivo PET/CT imaging showed that introduction of PEG spacers more than doubled the in vivo tumor uptake after 1 h compared with the non-spacer version: 68Ga-NODAGA-PEG3-AE105-NH2 (2.08 ± 0.37%ID/g) and 68Ga-NODAGA-PEG8-AE105-NH2 (2.01 ± 0.22%ID/g) vs. 68Ga-NODAGA-AE105-NH2 (0.70 ± 0.40%ID/g), p < 0.05. In addition, 68Ga-NODAGA-PEG8-AE105-NH2 showed significantly higher (p < 0.05) tumor-to-background contrast (3.68 ± 0.23) than the other tracers. The specific tumor-targeting property of 68Ga-NODAGA-PEG8-AE105-NH2 was established by effectively blocking the tumor uptake with co-injection of unlabeled AE105-NH2 (1 h: unblocked 2.01 ± 0.22%ID/g vs. blocked 1.24 ± 0.09%ID/g, p < 0.05). Ex vivo biodistribution confirmed the improved tumor uptakes of the PEG-modified tracers. 68Ga-NODAGA-PEG8-AE105-NH2 is thus a promising candidate for human translation for PET imaging of GBM.










Similar content being viewed by others
Abbreviations
- AE105-NH2 :
-
Ac-Asp-Cha-Phe-(D)Ser-(D)Arg-Tyr-Leu-Trp-Ser-CONH2
- AE105-NH2-mut :
-
Arg-Cha-Glu-(D)Ser-(D)-Arg-Tyr-Leu-Glu-Ser-CONH2
- AOC:
-
Amino-octanoic acid
- Azido-PEG3 :
-
11-azido-3,6,9-trioxaundecan-1-amine
- Azido-PEG8 :
-
O-(2-Aminoethyl)-O′-(2-azidoethyl)heptaethylene glycol
- BCN-NHS:
-
(1R,8S,9 s)-Bicyclo[6.1.0]non-4-yn-9-ylmethyl N-succinimidyl carbonate
- DIPEA:
-
N,N-diisopropylethylamine
- Fmoc-AOC:
-
8-(Fmoc-amino)octanoic acid
- GBM:
-
Glioblastoma
- NODAGA-NHS:
-
2,2′-(7-(1-Carboxy-4-((2,5-dioxopyrrolidin-1-yl)oxy)-4-oxobutyl)-1,4,7-triazonane-1,4-diyl)diacetic acid
- PEG3 :
-
Tri-ethylene glycol
- PEG8 :
-
Octa-ethylene glycol
- p.i.:
-
Post-injection
- ROI:
-
Region of interest
- uPAR:
-
Urokinase-type plasminogen activator receptor
- %ID/g:
-
Percent injected dose per gram
References
Autio A, Henttinen T, Sipilä HJ et al (2011) Mini-PEG spacering of VAP-1-targeting 68Ga-DOTAVAP-P1 peptide improves PET imaging of inflammation. EJNMMI Res 1:10. doi:10.1186/2191-219X-1-10
Bernstein LR (1998) Mechanisms of therapeutic activity for gallium. Pharmacol Rev 50:665–682
Cekanova M, Rathore K (2014) Animal models and therapeutic molecular targets of cancer: utility and limitations. Drug Des Devel Ther 8:1911–1921. doi:10.2147/DDDT.S49584
Dass K, Ahmad A, Azmi AS et al (2008) Evolving role of uPA/uPAR system in human cancers. Cancer Treat Rev 34:122–136. doi:10.1016/j.ctrv.2007.10.005
Duffy MJ (2004) The urokinase plasminogen activator system: role in malignancy. Curr Pharm Des 10:39–49
Knör S, Sato S, Huber T et al (2008) Development and evaluation of peptidic ligands targeting tumour-associated urokinase plasminogen activator receptor (uPAR) for use in alpha-emitter therapy for disseminated ovarian cancer. Eur J Nucl Med Mol Imaging 35:53–64. doi:10.1007/s00259-007-0582-3
Kriegbaum MC, Persson M, Haldager L et al (2011) Rational targeting of the urokinase receptor (uPAR): development of antagonists and non-invasive imaging probes. Curr Drug Targets 12:1711–1728
Li Z-B, Niu G, Wang H et al (2008) Imaging of urokinase-type plasminogen activator receptor expression using a 64Cu-labeled linear peptide antagonist by microPET. Clin Cancer Res 14:4758–4766. doi:10.1158/1078-0432.CCR-07-4434
Lin L, Gårdsvoll H, Huai Q et al (2010) Structure-based engineering of species selectivity in the interaction between urokinase and its receptor: implication for preclinical cancer therapy. J Biol Chem 285:10982–10992. doi:10.1074/jbc.M109.093492
Lund IK, Illemann M, Thurison T et al (2011) uPAR as anti-cancer target: evaluation of biomarker potential, histological localization, and antibody-based therapy. Curr Drug Targets 12:1744–1760
Persson M, Madsen J, Østergaard S et al (2012a) 68 Ga-labeling and in vivo evaluation of a uPAR binding DOTA- and NODAGA-conjugated peptide for PET imaging of invasive cancers. Nucl Med Biol 39:560–569. doi:10.1016/j.nucmedbio.2011.10.011
Persson M, Madsen J, Østergaard S et al (2012b) Quantitative PET of human urokinase-type plasminogen activator receptor with 64Cu-DOTA-AE105: implications for visualizing cancer invasion. J Nucl Med 53:138–145. doi:10.2967/jnumed.110.083386
Persson M, Hosseini M, Madsen J et al (2013a) Improved PET imaging of uPAR expression using new (64)Cu-labeled cross-bridged peptide ligands: comparative in vitro and in vivo studies. Theranostics 3:618–632. doi:10.7150/thno.6810
Persson M, Liu H, Madsen J et al (2013b) First (18)F-labeled ligand for PET imaging of uPAR: in vivo studies in human prostate cancer xenografts. Nucl Med Biol 40:618–624. doi:10.1016/j.nucmedbio.2013.03.001
Persson M, Skovgaard D, Brandt-Larsen M et al (2015) First-in-human uPAR PET: imaging of cancer aggressiveness. Theranostics 5:1303–1316. doi:10.7150/thno.12956
Persson M, Nedergaard MK, Brandt-Larsen M et al (2016) Urokinase-type plasminogen activator receptor as a potential PET biomarker in glioblastoma. J Nucl Med 57:272–278. doi:10.2967/jnumed.115.161703
Ploug M (2013) Structure-driven design of radionuclide tracers for non-invasive imaging of uPAR and targeted radiotherapy. The tale of a synthetic peptide antagonist. Theranostics 3:467–476. doi:10.7150/thno.3791
Ploug M, Østergaard S, Gårdsvoll H et al (2001) Peptide-derived antagonists of the urokinase receptor. affinity maturation by combinatorial chemistry, identification of functional epitopes, and inhibitory effect on cancer cell intravasation. Biochemistry 40:12157–12168
Roesch F, Riss PJ (2010) The renaissance of the 68Ge/68Ga radionuclide generator initiates new developments in 68Ga radiopharmaceutical chemistry. Curr Top Med Chem 10:1633–1668
Salajegheh M, Rudnicki A, Smith TW (2005) Expression of urokinase-type plasminogen activator receptor (uPAR) in primary central nervous system neoplasms. Appl Immunohistochem Mol Morphol AIMM 13:184–189
Sun Y, Ma X, Cheng K et al (2015) Strained cyclooctyne as a molecular platform for construction of multimodal imaging probes. Angew Chem Int Ed Engl 54:5981–5984. doi:10.1002/anie.201500941
Varasteh Z, Velikyan I, Lindeberg G et al (2013) Synthesis and characterization of a high-affinity NOTA-conjugated bombesin antagonist for GRPR-targeted tumor imaging. Bioconjug Chem 24:1144–1153. doi:10.1021/bc300659k
Varasteh Z, Rosenström U, Velikyan I et al (2014) The effect of mini-PEG-based spacer length on binding and pharmacokinetic properties of a 68Ga-labeled NOTA-conjugated antagonistic analog of bombesin. Molecules 19:10455–10472. doi:10.3390/molecules190710455
Zhang X, Tian YE, Sun F et al (2015) Imaging of human pancreatic cancer xenografts by single-photon emission computed tomography with (99m)Tc-Hynic-PEG-AE105. Oncol Lett 10:2253–2258. doi:10.3892/ol.2015.3504
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Conflict of interest
The authors declare that they have no conflicts of interest.
Funding
This work was partially supported by the Office of Science (BER), U.S. Department of Energy (DE-SC0008397), National Institute of Health In vivo Cellular Molecular Imaging Center (ICMIC) grant P50 CA114747, the Lundbeck Foundation Clinical Research Fellowship Grant, the Novo Nordisk Foundation, the Innovation Fund Denmark, the Research Foundation of Rigshospitalet, the Research Foundation of the Capital Region of Denmark, the Svend Andersen Foundation, the Arvid Nilsson Foundation and the Danish Cancer Society.
Additional information
M. D. Loft and Y. Sun contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
726_2017_2407_MOESM1_ESM.pdf
Fig. S1a MALDI-TOFMS data for NODAGA-AE105-NH2. Fig. S1b MALDI-TOFMS data for NODAGA-AOC-AE105-NH2. Fig. S1c MALDI-TOFMS data for NODAGA-PEG3-AE105-NH2. Fig. S1d MALDI-TOFMS data for NODAGA-PEG8-AE105-NH2. Fig. S2 Radio-HPLC data for 68Ga-NODAGA-X-AE105-NH2 (X = 0, AOC, PEG3 or PEG8). Fig. S3 PET imaging derived organ uptakes for 68Ga-NODAGA-PEG8-AE105-NH2 with and without blocking (co-injection of AE105-NH2). Table S1 60 min ex vivo biodistribution data for 68Ga-NODAGA-X-AE105-NH2 (X = 0, AOC, PEG3 or PEG8) (PDF 1529 kb)
Rights and permissions
About this article
Cite this article
Loft, M.D., Sun, Y., Liu, C. et al. Improved positron emission tomography imaging of glioblastoma cancer using novel 68Ga-labeled peptides targeting the urokinase-type plasminogen activator receptor (uPAR). Amino Acids 49, 1089–1100 (2017). https://doi.org/10.1007/s00726-017-2407-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-017-2407-4