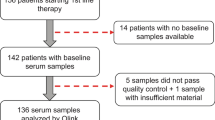
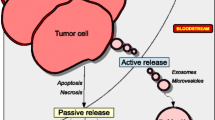
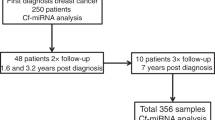
Abstract
Breast cancer is a multifactorial disease classified by several sub-types which differ from each other by risk factors, specific molecular promoters and severity of outcomes. Tumour aggressiveness and metastatic disease are the key determinants of breast cancer outcomes. Tumour cell ability to degrade the extracellular matrix and to be motile is the hallmark of invasion and essential step in a development of breast cancer metastatic disease. Therefore, a coordinated action between cell motility and ability to degrade the extracellular matrix is currently under extensive investigation focused on molecular targets for both diagnostic and therapeutic purposes. Contextually, our current study was dedicated to patient stratification utilising MMP-9 serum activity levels and RhoA expression patterns measured in circulating leucocytes. Biomarker patterns were “masked” in non-stratified patient groups. In contrast, the multiparametric stratification approach led to highly improved clinical utility of biomarker patterns. Presented stratification system is recommended for population screening as a cost-effective non-invasive approach to facilitate predictive diagnostics of breast cancer predisposition, pre-lesions and early stages, when the pathology can be effectively prevented or cured. Proposed approach might be particularly useful for early and predictive breast cancer diagnostics applied to certain phenotypes such as premenopausal rather than postmenopausal women, women with dense breast tissue, where highly increased RhoA/MMPs activities are utilised for effective proteolysis of the matrix and cancer cell migration into dense matrices, as well as for breast cancer of unclear origin such as particularly aggressive triple-negative sub-type.




Similar content being viewed by others
References
Abraham J-A, Golubnitschaja O (2016) Time for paradigm change in management of hepatocellular carcinoma: is an personalized approach on the horizon? Pers Med 13:455–467. doi:10.2217/pme-2016-0013
Bhandary L, Whipple RA, Vitolo MI et al (2015) ROCK inhibition promotes microtentacles that enhance reattachment of breast cancer cells. Oncotarget 6:6251–6266. doi:10.18632/oncotarget.3360
Braun M, Fountoulakis M, Papadopoulou A et al (2009) Down-regulation of microfilamental network-associated proteins in leukocytes of breast cancer patients: potential application to predictive diagnosis. Cancer Genom Proteom 6:31–40
Cáceres M, Guerrero J, Martínez J (2005) Overexpression of RhoA-GTP induces activation of the epidermal growth factor receptor, dephosphorylation of focal adhesion kinase and increased motility in breast cancer cells. Exp Cell Res 309:229–238. doi:10.1016/j.yexcr.2005.05.020
Camara O, Kavallaris A, Nöschel H et al (2006) Seeding of epithelial cells into circulation during surgery for breast cancer: the fate of malignant and benign mobilized cells. World J Surg Oncol 4:67. doi:10.1186/1477-7819-4-67
Carr HS, Zuo Y, Oh W, Frost JA (2013) Regulation of focal adhesion kinase activation, breast cancer cell motility, and amoeboid invasion by the RhoA guanine nucleotide exchange factor Net1. Mol Cell Biol 33:2773–2786. doi:10.1128/MCB.00175-13
Chang Y-WE, Marlin JW, Chance TW, Jakobi R (2006) RhoA mediates cyclooxygenase-2 signaling to disrupt the formation of adherens junctions and increase cell motility. Cancer Res 66:11700–11708. doi:10.1158/0008-5472.CAN-06-1818
Cheng H, Wang L, Mollica M et al (2014) Nitric oxide in cancer metastasis. Cancer Lett 353:1–7. doi:10.1016/j.canlet.2014.07.014
Covington KR, Brusco L, Barone I et al (2013) Metastasis tumor-associated protein 2 enhances metastatic behavior and is associated with poor outcomes in estrogen receptor-negative breast cancer. Breast Cancer Res Treat 141:375–384. doi:10.1007/s10549-013-2709-5
Cox TR, Rumney RMH, Schoof EM et al (2015) The hypoxic cancer secretome induces pre-metastatic bone lesions through lysyl oxidase. Nature 522:106–110. doi:10.1038/nature14492
Debald M, Yeghiazaryan K, Cebioglu M et al (2013) “Suspect molecular signature” in blood as the indicator of undiagnosed breast cancer, cancer risk and targeted prevention. EPMA J 4:22. doi:10.1186/1878-5085-4-22
Duffy MJ, McGowan PM, Harbeck N et al (2014) uPA and PAI-1 as biomarkers in breast cancer: validated for clinical use in level-of-evidence-1 studies. Breast Cancer Res 16:428. doi:10.1186/s13058-014-0428-4
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174. doi:10.1038/nrc745
Fagan-Solis KD, Schneider SS, Pentecost BT et al (2013) The RhoA pathway mediates MMP-2 and MMP-9-independent invasive behavior in a triple-negative breast cancer cell line. J Cell Biochem 114:1385–1394. doi:10.1002/jcb.24480
Friedl P, Wolf K (2010) Plasticity of cell migration: a multiscale tuning model. J Cell Biol 188:11–19. doi:10.1083/jcb.200909003
Fritz G, Just I, Kaina B (1999) Rho GTPases are over-expressed in human tumors. Int J Cancer 81:682–687
Girotra S, Yeghiazaryan K, Golubnitschaja O (2016) Potential biomarker panels in overall breast cancer management: advancements by multilevel diagnostics. Pers Med 13:469–484. doi:10.2217/pme-2016-0020
Goldfarb Y, Ben-Eliyahu S (2006) Surgery as a risk factor for breast cancer recurrence and metastasis: mediating mechanisms and clinical prophylactic approaches. Breast Dis 26:99–114
Golubnitschaja O, Sridhar KC (2016) Liver metastatic disease: new concepts and biomarker panels to improve individual outcomes. Clin Exp Metastasis. doi:10.1007/s10585-016-9816-8
Golubnitschaja O, Yeghiazaryan K, Costigliola V et al (2013) Risk assessment, disease prevention and personalised treatments in breast cancer: is clinically qualified integrative approach in the horizon? EPMA J 4:6. doi:10.1186/1878-5085-4-6
Golubnitschaja O, Debald M, Kuhn W et al (2016a) Flammer syndrome and potential formation of pre-metastatic niches: a multi-centred study on phenotyping, patient stratification, prediction and potential prevention of aggressive breast cancer and metastatic disease. EPMA J 7(Suppl 1):A25. doi:10.1186/s13167-016-0054-6
Golubnitschaja O, Debald M, Yeghiazaryan K et al (2016b) Breast cancer epidemic in the early twenty-first century: evaluation of risk factors, cumulative questionnaires and recommendations for preventive measures. Tumour Biol. doi:10.1007/s13277-016-5168-x
Golubnitschaja O, Yeghiazaryan K, Stricker H et al (2016c) Patients with hepatic breast cancer metastases demonstrate highly specific profiles of matrix metalloproteinases MMP-2 and MMP-9 after SIRT treatment as compared to other primary and secondary liver tumours. BMC Cancer 16:357. doi:10.1186/s12885-016-2382-2
Golubnitschaja-Labudova O, Liu R, Decker C et al (2000) Altered gene expression in lymphocytes of patients with normal-tension glaucoma. Curr Eye Res 21:867–876
Gou L, Wang W, Tong A et al (2011) Proteomic identification of RhoA as a potential biomarker for proliferation and metastasis in hepatocellular carcinoma. J Mol Med 89:817–827. doi:10.1007/s00109-011-0753-3
Harms W, Malter W, Krämer S et al (2014) Clinical significance of urokinase-type plasminogen activator (uPA) and its type-1 inhibitor (PAI-1) for metastatic sentinel lymph node involvement in breast cancer. Anticancer Res 34:4457–4462
Jacobs VR, Kates RE, Kantelhardt E et al (2013) Health economic impact of risk group selection according to ASCO-recommended biomarkers uPA/PAI-1 in node-negative primary breast cancer. Breast Cancer Res Treat 138:839–850. doi:10.1007/s10549-013-2496-z
Konieczka K, Ritch R, Traverso CE et al (2014) Flammer syndrome. EPMA J 5:11. doi:10.1186/1878-5085-5-11
Leppä S, Saarto T, Vehmanen L et al (2004) A high serum matrix metalloproteinase-2 level is associated with an adverse prognosis in node-positive breast carcinoma. Clin Cancer Res 10:1057–1063
Marino N, Woditschka S, Reed LT et al (2013) Breast cancer metastasis: issues for the personalization of its prevention and treatment. Am J Pathol 183:1084–1095. doi:10.1016/j.ajpath.2013.06.012
Mehner C, Hockla A, Miller E et al (2014) Tumor cell-produced matrix metalloproteinase 9 (MMP-9) drives malignant progression and metastasis of basal-like triple negative breast cancer. Oncotarget 5:2736–2749. doi:10.18632/oncotarget.1932
Muñoz-Nájar UM, Neurath KM, Vumbaca F, Claffey KP (2006) Hypoxia stimulates breast carcinoma cell invasion through MT1-MMP and MMP-2 activation. Oncogene 25:2379–2392. doi:10.1038/sj.onc.1209273
Narumiya S, Tanji M, Ishizaki T (2009) Rho signaling, ROCK and mDia1, in transformation, metastasis and invasion. Cancer Metastasis Rev 28:65–76. doi:10.1007/s10555-008-9170-7
Provatopoulou X, Gounaris A, Kalogera E et al (2009) Circulating levels of matrix metalloproteinase-9 (MMP-9), neutrophil gelatinase-associated lipocalin (NGAL) and their complex MMP-9/NGAL in breast cancer disease. BMC Cancer 9:390. doi:10.1186/1471-2407-9-390
Ranuncolo SM, Armanasco E, Cresta C et al (2003) Plasma MMP-9 (92 kDa-MMP) activity is useful in the follow-up and in the assessment of prognosis in breast cancer patients. Int J Cancer 106:745–751. doi:10.1002/ijc.11288
Raviraj V, Fok S, Zhao J et al (2012) Regulation of ROCK1 via Notch1 during breast cancer cell migration into dense matrices. BMC Cell Biol 13:12. doi:10.1186/1471-2121-13-12
Sanz-Moreno V, Marshall CJ (2010) The plasticity of cytoskeletal dynamics underlying neoplastic cell migration. Curr Opin Cell Biol 22:690–696. doi:10.1016/j.ceb.2010.08.020
Sayagués JM, Corchete LA, Gutiérrez ML et al (2016) Genomic characterization of liver metastases from colorectal cancer patients. Oncotarget. doi:10.18632/oncotarget.12140
Sun K, Duan X, Cai H et al (2016) Curcumin inhibits LPA-induced invasion by attenuating RhoA/ROCK/MMPs pathway in MCF7 breast cancer cells. Clin Exp Med 16:37–47. doi:10.1007/s10238-015-0336-7
Sung H, Choi J-Y, Lee S-A et al (2012) The association between the preoperative serum levels of lipocalin-2 and matrix metalloproteinase-9 (MMP-9) and prognosis of breast cancer. BMC Cancer 12:193. doi:10.1186/1471-2407-12-193
Tabariès S, Ouellet V, Hsu BE et al (2015) Granulocytic immune infiltrates are essential for the efficient formation of breast cancer liver metastases. Breast Cancer Res 17:45. doi:10.1186/s13058-015-0558-3
Talvensaari-Mattila A, Pääkkö P, Turpeenniemi-Hujanen T (2003) Matrix metalloproteinase-2 (MMP-2) is associated with survival in breast carcinoma. Br J Cancer 89:1270–1275. doi:10.1038/sj.bjc.6601238
WHO (2012) WHO|cancer. In: WHO. http://www.who.int/mediacentre/factsheets/fs297/en/. Accessed 15 Jun 2016
Acknowledgements
The authors thank the European Association for Predictive, Preventive and Personalised Medicine (EPMA, Belgium) for professional and financial support of the project. Further, our cordial thanks belong to Prof. Dr. J. Flammer, University of Basel, Switzerland for professional considerations of potential implementing areas for the stratification system published here and critical reading of the manuscript. For the performance of “Western-blot” analysis, the authors thank Mrs. G. Windisch-Schuster, Department of Radiology, University of Bonn.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with animals performed by any of the authors.
Funding
The study funding has been performed by the Breast Cancer Research Centre, University of Bonn, Bonn, Germany. KY has been awarded with corresponding fellowship by the European Association for Predictive, Preventive and Personalised Medicine (EPMA, Belgium).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Handling Editor: G. J. Peters.
Rights and permissions
About this article
Cite this article
Golubnitschaja, O., Yeghiazaryan, K., Abraham, JA. et al. Breast cancer risk assessment: a non-invasive multiparametric approach to stratify patients by MMP-9 serum activity and RhoA expression patterns in circulating leucocytes. Amino Acids 49, 273–281 (2017). https://doi.org/10.1007/s00726-016-2357-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-016-2357-2