Abstract
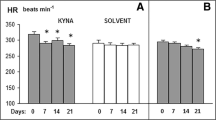
S-adenosyl-l-methionine (SAM) is an amino acid involved in a number of physiological processes in the nervous system. Some evidence suggests a therapeutic potential of SAM in hypertension. In this study we investigated the effect of intracerebroventricular (ICV) infusions of SAM on arterial blood pressure in rats. Mean arterial blood pressure (MABP) and heart rate (HR) were measured at baseline and during ICV infusion of either SAM or vehicle (aCSF; controls) in conscious, male normotensive Wistar Kyoto rats (WKY) and Spontaneously Hypertensive Rats (SHR). MABP and HR were not affected by the vehicle. WKY rats infused with SAM (10 μM, 100 μM and 1 mM) showed a biphasic hemodynamic response i.e., mild hypotension and bradycardia followed by a significant increase in MABP and HR. On the contrary, SHR infused with SAM showed a dose-dependent hypotensive response. In separate series of experiments, pretreatment with hexamethonium, a ganglionic blocker as well as pretreatment with glibenclamide, a KATP channel blocker reduced the hemodynamic effects of SAM. SAM may affect the nervous control of arterial blood pressure via the autonomic nervous system and KATP channel-dependent mechanisms.




Similar content being viewed by others
References
Ashcroft FM, Gribble FM (1998) Correlating structure and function in ATP-sensitive K+ channels. Trends Neurosci 21(7):288–294
Asor E, Stempler S, Avital A, Klein E, Ruppin E, Ben-Shachar D (2015) The role of branched chain amino acid and tryptophan metabolism in rat’s behavioral diversity: Intertwined peripheral and brain effects. Eur Neuropsychopharmacol 25(10):1695–1705. doi:10.1016/j.euroneuro.2015.07.009
Bencze M, Behuliak M, Zicha J (2013) The impact of four different classes of anesthetics on the mechanisms of blood pressure regulation in normotensive and spontaneously hypertensive rats. Physiol Res 62(5):471–478
Bottiglieri T (2002) S-Adenosyl-l-methionine (SAMe): from the bench to the bedside–molecular basis of a pleiotrophic molecule. Am J Clin Nutr 76(5):1151S–1157S
Brown MD, Dudeja PK, Brasitus TA (1988) S-adenosyl-l-methionine modulates Na+ + K+ -ATPase activity in rat colonic basolateral membranes. Biochem J 251(1):215–222
De Berardis D, Orsolini L, Serroni N, Girinelli G, Iasevoli F, Tomasetti C, de Bartolomeis A, Mazza M, Valchera A, Fornaro M, Perna G, Piersanti M, Di Nicola M, Cavuto M, Martinotti G, Di Giannantonio M (2016) A comprehensive review on the efficacy of S-Adenosyl-l-methionine in Major Depressive Disorder. CNS Neurol Disord Drug Targets 15(1):35–44
Ding YA, Chang ST, Shieh SM, Huang WC (1988) Antihypertensive and renal effects of cilazapril and their reversal by angiotensin in renovascular hypertensive rats. Clin Sci (Lond) 74(4):365–372
Duan XC, Guo R, Liu SY, Xiao L, Xue HM, Guo Q, Jin S, Wu YM (2015) Gene transfer of cystathionine beta-synthase into RVLM increases hydrogen sulfide-mediated suppression of sympathetic outflow via KATP channel in normotensive rats. Am J Physiol Heart Circ Physiol 308(6):H603–H611. doi:10.1152/ajpheart.00693.2014
Eto K, Kimura H (2002) A novel enhancing mechanism for hydrogen sulfide-producing activity of cystathionine beta-synthase. J Biol Chem 277(45):42680–42685. doi:10.1074/jbc.M205835200
Fan Y, Lan L, Zheng L, Ji X, Lin J, Zeng J, Huang R, Sun J (2015) Spontaneous white matter lesion in brain of stroke-prone renovascular hypertensive rats: a study from MRI, pathology and behavior. Metab Brain Dis 30(6):1479–1486. doi:10.1007/s11011-015-9722-9
Furujo M, Kinoshita M, Nagao M, Kubo T (2012) Methionine adenosyltransferase I/III deficiency: neurological manifestations and relevance of S-adenosylmethionine. Mol Genet Metab 107(3):253–256. doi:10.1016/j.ymgme.2012.08.002
Grillo MA, Colombatto S (2008) S-adenosylmethionine and its products. Amino Acids 34(2):187–193. doi:10.1007/s00726-007-0500-9
Guo Q, Jin S, Wang XL, Wang R, Xiao L, He RR, Wu YM (2011) Hydrogen sulfide in the rostral ventrolateral medulla inhibits sympathetic vasomotor tone through ATP-sensitive K+ channels. J Pharmacol Exp Ther 338(2):458–465. doi:10.1124/jpet.111.180711
Hawkins RA, O’Kane RL, Simpson IA, Vina JR (2006) Structure of the blood-brain barrier and its role in the transport of amino acids. J Nutr 136(1 Suppl):218S–226S
Jordi J, Herzog B, Camargo SM, Boyle CN, Lutz TA, Verrey F (2013) Specific amino acids inhibit food intake via the area postrema or vagal afferents. J Physiol 591(Pt 22):5611–5621. doi:10.1113/jphysiol.2013.258947
Kamoun P (2004) Endogenous production of hydrogen sulfide in mammals. Amino Acids 26(3):243–254. doi:10.1007/s00726-004-0072-x
Kampfer AJ, Balog EM (2010) S-adenosyl-l-methionine regulation of the cardiac ryanodine receptor involves multiple mechanisms. Biochemistry 49(35):7600–7614. doi:10.1021/bi100599b
Laffin LJ, Bakris GL (2015) Hypertension and new treatment approaches targeting the sympathetic nervous system. Curr Opin Pharmacol 21:20–24. doi:10.1016/j.coph.2014.12.006
Lake KD, Martin BR, Kunos G, Varga K (1997) Cardiovascular effects of anandamide in anesthetized and conscious normotensive and hypertensive rats. Hypertension 29(5):1204–1210
Lakhi S, Snow W, Fry M (2013) Insulin modulates the electrical activity of subfornical organ neurons. Neuroreport 24(6):329–334. doi:10.1097/WNR.0b013e32835ffc14
Li DP, Chen SR, Pan HL (2010) Adenosine inhibits paraventricular pre-sympathetic neurons through ATP-dependent potassium channels. J Neurochem 113(2):530–542. doi:10.1111/j.1471-4159.2010.06618.x
Liu WQ, Chai C, Li XY, Yuan WJ, Wang WZ, Lu Y (2011) The cardiovascular effects of central hydrogen sulfide are related to K(ATP) channels activation. Physiol Res 60(5):729–738
Losada ME, Rubio MC (1989) Acute effects of S-adenosyl-l-methionine on catecholaminergic central function. Eur J Pharmacol 163(2–3):353–356
Lu SC, Mato JM (2012) S-adenosylmethionine in liver health, injury, and cancer. Physiol Rev 92(4):1515–1542. doi:10.1152/physrev.00047.2011
Montgomery SE, Sepehry AA, Wangsgaard JD, Koenig JE (2014) The effect of S-adenosylmethionine on cognitive performance in mice: an animal model meta-analysis. PLoS One 9(10):e107756. doi:10.1371/journal.pone.0107756
Otero-Losada ME, Rubio MC (1989) Acute changes in 5-HT metabolism after S-adenosyl-l-methionine administration. Gen Pharmacol 20(4):403–406
Panza F, Frisardi V, Capurso C, D’Introno A, Colacicco AM, Di Palo A, Imbimbo BP, Vendemiale G, Capurso A, Solfrizzi V (2009) Polyunsaturated fatty acid and S-adenosylmethionine supplementation in predementia syndromes and Alzheimer’s disease: a review. Sci World J 9:373–389. doi:10.1100/tsw.2009.48
Qiao W, Yang L, Li XY, Cao N, Wang WZ, Chai C, Lu Y (2011) The cardiovascular inhibition functions of hydrogen sulfide within the nucleus tractus solitarii are mediated by the activation of KATP channels and glutamate receptors mechanisms. Pharmazie 66(4):287–292
Roy A, Khan AH, Islam MT, Prieto MC, Majid DS (2012) Interdependency of cystathione gamma-lyase and cystathione beta-synthase in hydrogen sulfide-induced blood pressure regulation in rats. Am J Hypertens 25(1):74–81. doi:10.1038/ajh.2011.149
Sarris J, Papakostas GI, Vitolo O, Fava M, Mischoulon D (2014) S-Adenosyl methionine (SAMe) versus escitalopram and placebo in major depression RCT: efficacy and effects of histamine and carnitine as moderators of response. J Affect Disord 164:76–81. doi:10.1016/j.jad.2014.03.041
Sikora M, Drapala A, Ufnal M (2014) Exogenous hydrogen sulfide causes different hemodynamic effects in normotensive and hypertensive rats via neurogenic mechanisms. Pharmacol Rep 66(5):751–758. doi:10.1016/j.pharep.2014.04.004
Stark R, Reichenbach A, Andrews ZB (2015) Hypothalamic carnitine metabolism integrates nutrient and hormonal feedback to regulate energy homeostasis. Mol Cell Endocrinol 418(Pt 1):9–16. doi:10.1016/j.mce.2015.08.002
Szczepanska-Sadowska E (2006) Neuropeptides in neurogenic disorders of the cardiovascular control. J Physiol Pharmacol 57(Suppl 11):31–53
Szczepanska-Sadowska E, Cudnoch-Jedrzejewska A, Ufnal M, Zera T (2010) Brain and cardiovascular diseases: common neurogenic background of cardiovascular, metabolic and inflammatory diseases. J Physiol Pharmacol 61(5):509–521
Takemoto Y (1990) Amino acids with central pressor effect in conscious rats. Jpn J Physiol 40(4):561–565
Takemoto Y (1991) Central depressor effects of amino acids in conscious normotensive and two-kidney, one-clip renovascular hypertensive rats. Jpn J Physiol 41(5):717–724
Takemoto Y (2012) Amino acids that centrally influence blood pressure and regional blood flow in conscious rats. J Amino Acids 2012:831759. doi:10.1155/2012/831759
Takemoto Y (2013) Pressor response to l-cysteine injected into the cisterna magna of conscious rats involves recruitment of hypothalamic vasopressinergic neurons. Amino Acids 44(3):1053–1060. doi:10.1007/s00726-012-1440-6
Takemoto Y (2014a) Cardiovascular actions of l-cysteine and l-cysteine sulfinic acid in the nucleus tractus solitarius of the rat. Amino Acids 46(7):1707–1713. doi:10.1007/s00726-014-1733-z
Takemoto Y (2014b) Functional cardiovascular action of l-cysteine microinjected into pressor sites of the rostral ventrolateral medulla of the rat. Amino Acids 46(4):863–872. doi:10.1007/s00726-013-1651-5
Takemoto Y (2014c) l-Cysteine and L-AP4 microinjections in the rat caudal ventrolateral medulla decrease arterial blood pressure. Auton Neurosci 186:45–53. doi:10.1016/j.autneu.2014.09.018
Toal CB, Leenen FH (1985) Blood pressure responsiveness during the development of hypertension in the conscious spontaneously hypertensive rat. Can J Physiol Pharmacol 63(10):1258–1262
Ufnal M, Sikora M (2011) The role of brain gaseous transmitters in the regulation of the circulatory system. Curr Pharm Biotechnol 12(9):1322–1333
Ufnal M, Skrzypecki J (2014) Blood borne hormones in a cross-talk between peripheral and brain mechanisms regulating blood pressure, the role of circumventricular organs. Neuropeptides 48(2):65–73. doi:10.1016/j.npep.2014.01.003
Ufnal M, Sikora M, Dudek M (2008) Exogenous hydrogen sulfide produces hemodynamic effects by triggering central neuroregulatory mechanisms. Acta Neurobiol Exp (Wars) 68(3):382–388
Vina JR, Davis DW, Hawkins RA (1986) The influence of nitrous oxide on methionine, S-adenosylmethionine, and other amino acids. Anesthesiology 64(4):490–495
Wang R (2012) Physiological implications of hydrogen sulfide: a whiff exploration that blossomed. Physiol Rev 92(2):791–896. doi:10.1152/physrev.00017.2011
Yan J, Li L, Khatibi NH, Yang L, Wang K, Zhang W, Martin RD, Han J, Zhang J, Zhou C (2011) Blood-brain barrier disruption following subarchnoid hemorrhage may be faciliated through PUMA induction of endothelial cell apoptosis from the endoplasmic reticulum. Exp Neurol 230(2):240–247. doi:10.1016/j.expneurol.2011.04.022
Yang Y, Rosenberg GA (2011) Blood-brain barrier breakdown in acute and chronic cerebrovascular disease. Stroke 42(11):3323–3328. doi:10.1161/STROKEAHA.110.608257
Yang HT, Chien YW, Tsen JH, Chang CC, Chang JH, Huang SY (2009) Taurine supplementation improves the utilization of sulfur-containing amino acids in rats continually administrated alcohol. J Nutr Biochem 20(2):132–139. doi:10.1016/j.jnutbio.2008.01.009
Young SN, Shalchi M (2005) The effect of methionine and S-adenosylmethionine on S-adenosylmethionine levels in the rat brain. J Psychiatry Neurosci 30(1):44–48
Zhang Y, Wang H, Sun HW, Chen YL, Ouyang JY, Wang Y, Wang L, Zhang XY (2014) Correlation between cystathionine beta-synthase T883C genetic polymorphism and primary hypertension. Exp Ther Med 8(3):713–718. doi:10.3892/etm.2014.1799
Acknowledgments
This study was funded by the National Science Center Grant No. 2011/01/N/NZ4/03682 and Medical University of Warsaw.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
This article does not contain any studies with human participants performed by any of the authors.
Additional information
Handling Editor: H. Jakubowski.
Rights and permissions
About this article
Cite this article
Sikora, M., Pham, K. & Ufnal, M. Hypotensive effect of S-adenosyl-l-methionine in hypertensive rats is reduced by autonomic ganglia and KATP channel blockers. Amino Acids 48, 1581–1590 (2016). https://doi.org/10.1007/s00726-016-2213-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-016-2213-4