Abstract
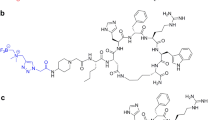
The melanocortin-1 receptor (MC1R) plays an important role in melanoma growth, angiogenesis and metastasis, and is overexpressed in melanoma cells. α-Melanocyte stimulating hormone (α-MSH) and derivatives are known to bind with high affinity at this receptor that provides the potential for selective targeting of melanoma. In this study, one linear α-MSH-derived peptide Nle-Asp-His-D-Phe-Arg-Trp-Gly-NH2 (NAP-NS1) without linker and with εAhx-β-Ala linker, and a cyclic α-MSH derivative, [Lys-Glu-His-D-Phe-Arg-Trp-Glu]-Arg-Pro-Val-NH2 (NAP-NS2) with εAhx-β-Ala linker were conjugated with p-SCN-Bn-NOTA and labeled with 64Cu. Radiochemical and radiopharmacological investigations were performed with regard to transchelation, stability, lipophilicity and in vitro binding assays as well as biodistribution in healthy rats. No transchelation reactions, but high metabolic stability and water solubility were demonstrated. The linear derivatives showed higher affinity than the cyclic one. [64Cu]Cu-NOTA-εAhx-β-Ala-NAP-NS1 ([64Cu]Cu-2) displayed rapid cellular association and dissociation in murine B16F10 cell homogenate. All [64Cu]Cu-labeled conjugates exhibited affinities in the low nanomolar range in B16F10. [64Cu]Cu-2 showed also high affinity in human MeWo and TXM13 cell homogenate. In vivo studies suggested that [64Cu]Cu-2 was stable, with about 85 % of intact peptide in rat plasma at 2 h p.i. Biodistribution confirmed the renal pathway as the major elimination route. The uptake of [64Cu]Cu-2 in the kidney was 5.9 % ID/g at 5 min p.i. and decreased to 2.0 % ID/g at 60 min p.i. Due to the prospective radiochemical and radiopharmacological properties of the linear α-MSH derivative [64Cu]Cu-2, this conjugate is a promising candidate for tracer development in human melanoma imaging.








Similar content being viewed by others
References
American Cancer Society (2015) Cancer Facts & Figures. http://www.cancer.org/acs/groups/content/@editorial/documents/document/acspc-044552.pdf. Assessed 13 Jan 2015
Ashton H, Millman JE, Telford R, Thompson JW, Davies TF, Hall R, Shuster S, Thody AJ, Coy DH, Kastin AJ (1977) Psychopharmacological and endocrinological effects of melanocyte stimulating hormones in normal man. Psychopharmacology 55:165–172
Bapst JP, Froidevaux S, Calame M, Tanner H, Eberle AN (2007) Dimeric DOTA-alpha- melanocyte-stimulating hormone analogs: synthesis and in vivo characteristics of radiopeptides with high in vitro activity. J Recept Signal Transduct Res 27:383–409
Bapst JP, Calame M, Tanner H, Eberle AN (2009) Glycosylated DOTA-alpha-melanocyte-stimulating hormone analogues for melanoma targeting: influence of the site of glycosylation on in vivo biodistribution. Bioconjug Chem 20:984–993
Behr TM, Goldenberg DM, Becker W (1998) Reducing the renal uptake of radiolabeled antibody fragments and peptides for diagnosis and therapy: present status, future prospects and limitations. Eur J Nucl Med 25:201–2011
Biondi B, Bolzati C, Carta D, Salvarese N, Refosco F, Calderan A, Ruzza P (2010) The [Tc(N)PNP] metal fragment labeled peptide for MC1 receptors imaging: preliminary studies. J Peptide Sci 16(Suppl 1):182
Castrucci AM, Hadley ME, Sawyer TK, Hruby VJ (1984) Enzymological studies of melanotropins. Comp Biochem Physiol B 78:519–524
Chen J, Cheng Z, Hoffman TJ, Jurisson SS, Quinn TP (2000) Melanoma-targeting properties of (99 m)technetium-labeled cyclic alpha-melanocyte-stimulating hormone peptide analogues. Cancer Res 60:5649–5658
Chen J, Cheng Z, Owen NK, Hoffman TJ, Miao Y, Jurisson SS, Quinn TP (2001) Evaluation of an (111)In-DOTA-rhenium cyclized alpha-MSH analog: a novel cyclic-peptide analog with improved tumor-targeting properties. J Nucl Med 42:1847–1855
Chen JQ, Cheng Z, Miao YB, Jurisson SS, Quinn TP (2002) Alpha-melanocyte- stimulating hormone peptide analogs labeled with technetium-99 m and indium-111 for malignant melanoma targeting. Cancer 94(4 Suppl):1196–1201
Cheng Z, Chen J, Miao Y, Owen NK, Quinn TP, Jurisson SS (2002) Modification of the structure of a metallopeptide: synthesis and biological evaluation of (111)In-labeled DOTA-conjugated rhenium-cyclized alpha-MSH analogues. J Med Chem 45:3048–3056
Cheng Z, Chen J, Quinn TP, Jurisson SS (2004a) Radioiodination of rhenium cyclized alpha-melanocyte-stimulating hormone resulting in enhanced radioactivity localization and retention in melanoma. Cancer Res 64:1411–1418
Cheng Z, Chen J, Quinn TP, Jurisson SS (2004b) Radioiodination of rhenium cyclized alpha-melanocyte-stimulating hormone resulting in enhanced radioactivity localization and retention in melanoma. Cancer Res 64:1411–1418
Cheng Z, Xiong ZM, Subbarayan M, Chen XY, Gambhir SS (2007a) Cu-64-labeled alpha-melanocyte-stimulating hormone analog for MicroPET imaging of melanocortin 1 receptor expression. Bioconjug Chem 18:765–772
Cheng Z, Zhang L, Graves E, Xiong Z, Dandekar M, Chen X, Gambhir SS (2007b) Small-animal PET of melanocortin 1 receptor expression using a 18F-labeled alpha-melanocyte- stimulating hormone analog. J Nucl Med 48:987–994
Chluba-de Tapia J, Bagutti C, Cotti R, Eberle AN (1996) Induction of constitutive melanogenesis in amelanotic mouse melanoma cells by transfection of the human melanocortin-1 receptor gene. J Cell Sci 109(Pt 8):2023–2030
Cooper MS, Ma MT, Sunassee K, Shaw KP, Williams JD, Paul RL, Donnelly PS, Blower PJ (2012) Comparison of (64)Cu-complexing bifunctional chelators for radioimmunoconjugation: labeling efficiency, specific activity, and in vitro/in vivo stability. Bioconjug Chem 23:1029–1039. doi:10.1021/bc300037w
Eberle AN (1988) 10. Secretion, Distribution and inactivation of MSH. In: Eberle AN (ed) The melanotropins. Chemistry, physiology and mechanisms of action, 1st edn. Karger, Basel, pp 173–209
Eberle AN, Verin VJ, Solca F, Siegrist W, Küenlin C, Bagutti C, Stutz S, Girard J (1991) Biologically active monoiodinated alpha-MSH derivatives for receptor binding studies using human melanoma cells. J Recept Res 11:311–312
Eberle AN, Bapst JP, Calame M, Tanner H, Froidevaux S (2010) MSH radiopeptides for targeting melanoma metastases. Adv Exp Med Biol 681:133–142. doi:10.1007/978-1-4419-6354-3_11
Fani M, Maecke HR (2012) Radiopharmaceutical development of radiolabelled peptides. Eur J Nucl Med Mol Imaging 39:S11–S30. doi:10.1007/s00259-011-2001-z
Flook AM, Yang J, Miao Y (2013) Evaluation of new Tc-99 m-labeled Arg-X-Asp-conjugated α-melanocyte stimulating hormone peptides for melanoma imaging. Mol Pharm 10:3417–3424. doi:10.1021/mp400248f
Froidevaux S, Calame-Christe M, Tanner H, Sumanovski L, Eberle AN (2002) A novel DOTA-alpha-melanocyte-stimulating hormone analog for metastatic melanoma diagnosis. J Nucl Med 43:1699–1706
Froidevaux S, Calame-Christe M, Schuhmacher J, Tanner H, Saffrich R, Henze M, Eberle AN (2004) A gallium-labeled DOTA-alpha-melanocyte-stimulating hormone analog for PET Imaging of melanoma metastases. J Nucl Med 45:116–123
Froidevaux S, Calame-Christe M, Tanner H, Eberle AN (2005) Melanoma targeting with DOTA-alpha-melanocyte-stimulating hormone analogs: structural parameters affecting tumor uptake and kidney uptake. J Nucl Med 46:887–895
Funasaka Y, Sato H, Chakraborty AK, Ohashi A, Chrousos GP, Ichihashi M (1999) Expression of proopiomelanocortin, corticotropin-releasing hormone (CRH), and CRH receptor in melanoma cells, nevus cells, and normal human melanocytes. J Investig Dermatol Symp Proc 4:105–109
Fung S, Hruby VJ (2005) Design of cyclic and other templates for potent and selective peptide alpha-MSH analogues. Curr Opin Chem Biol 9:352–358
Garcia MF, Zhang X, Gallazzi F, Fernandez M, Moreno M, Gambini JP, Porcal W, Cabral P, Quinn TP (2014) Evaluation of tricine and EDDA as Co-ligands for 99mTc-labeled HYNIC-MSH analogs for melanoma imaging. Anticancer Agents Med Chem 15:122–130
Giblin MF, Wang N, Hoffman TJ, Jurisson SS, Quinn TP (1998) Design and characterization of alpha-melanotropin peptide analogs cyclized through rhenium and technetium metal coordination. Proc Natl Acad Sci U S A 95:12814–12818
Guo H, Miao Y (2012) Cu-64-labeled lactam bridge-cyclized α-MSH peptides for PET imaging of melanoma. Mol Pharm 9:2322–2330. doi:10.1021/mp300246j
Guo H, Yang J, Gallazzi F, Prossnitz ER, Sklar LA, Miao Y (2009a) Effect of DOTA position on melanoma targeting and pharmacokinetic properties of 111In-labeled lactam bridge-cyclized alpha-melanocyte stimulating hormone peptide. Bioconjug Chem 20:2162–2168. doi:10.1021/bc9003475
Guo H, Yang J, Shenoy N, Miao Y (2009b) Gallium-67-labeled lactam bridge-cyclized alpha-melanocyte stimulating hormone peptide for primary and metastatic melanoma imaging. Bioconjug Chem 20:2356–2363
Guo H, Yang J, Gallazzi F, Miao Y (2010) Reduction of the ring size of radiolabeled lactam bridge-cyclized alpha-MSH peptide, resulting in enhanced melanoma uptake. J Nucl Med 51:418–426
Kim MH, Park JA, Woo SK, Lee KC, An GI, Kim BS, Kim KI, Lee TS, Kim CW, Kim KM, Kang JH, Lee YJ (2015) Evaluation of a 64Cu-labeled 1,4,7-triazacyclononane, 1-glutaric acid-4,7 acetic acid (NODAGA)-galactose-bombesin analogue as a PET imaging probe in a gastrin-releasing peptide receptor-expressing prostate cancer xenograft model. Int J Oncol 46:1159–1168. doi:10.3892/ijo.2015.2832
Kopanchuk S, Veiksina S, Petrovska R, Mutule I, Szardenings M, Rinken A, Wikberg JE (2005) Co-operative regulation of ligand binding to melanocortin receptor subtypes: evidence for interacting binding sites. Eur J Pharmacol 512:85–95
Lang L, Li W, Guo N, Ma Y, Zhu L, Kiesewetter DO, Shen B, Niu G, Chen X (2011) Comparison study of [18F]FAl-NOTA-PRGD2, [18F]FPPRGD2, and [68Ga]Ga-NOTA-PRGD2 for PET imaging of U87MG tumors in mice. Bioconjug Chem 22:2415–2422. doi:10.1021/bc200197h
Lim JC, Hong YD, Kim JJ, Choi SM, Baek HS, Choi SJ (2012) Synthesis and biological evaluation of a novel (177)Lu-DOTA-[Gly(3)-cyclized(Dap(4), (d)-Phe(7), Asp(10))-Arg(11)]α-MSH(3-13) analogue for melanocortin-1 receptor-positive tumor targeting. Cancer Biother Radiopharm 27:464–472
Lin MJ, Mar V, McLean C, Wolfe R, Kelly JW (2014) Diagnostic accuracy of malignant melanoma according to subtype. Australas J Dermatol 55:35–42. doi:10.1111/ajd.12121
Lindskog Jonsson A, Granqvist A, Elvin J, Johansson ME, Haraldsson B, Nyström J (2014) Effects of melanocortin 1 receptor agonists in experimental nephropathies. PLoS ONE 9:e87816. doi:10.1371/journal.pone.0087816
Loir B, Pérez Sánchez C, Ghanem G, Lozano JA, García-Borrón JC, Jiménez- Cervantes G (1999) Expression of the MC1 receptor gene in normal and malignant human melanocytes. A semiquantitative RT-PCR study. Cell Mol Biol 45:1083–1092
López MN, Pereda C, Ramírez M, Mendoza-Naranjo A, Serrano A, Ferreira A, Poblete R, Kalergis AM, Kiessling R, Salazar-Onfray F (2007) Melanocortin 1 receptor is expressed by uveal malignant melanoma and can be considered a new target for diagnosis and immunotherapy. Invest Ophthalmol Vis Sci 48:1219–1227
Lunec J, Pieron C, Thody AJ (1992) MSH receptor expression and the relationship to melanogenesis and metastatic activity in B16 melanoma. Melanoma Res 2:5–12
Maack T, Johnson V, Kau ST, Figueiredo J, Sigulem D (1979) Renal filtration, transport, and metabolism of low-molecular-weight proteins: a review. Kidney Int 16:251–270
Miao Y, Owen NK, Whitener D, Gallazzi F, Hoffman TJ, Quinn TP (2002) In vivo evaluation of 188Re-labeled alpha-melanocyte stimulating hormone peptide analogs for melanoma therapy. Int J Cancer 101:480–487
Miao Y, Whitener D, Feng W, Owen NK, Chen J, Quinn TP (2003) Evaluation of the human melanoma targeting properties of radiolabeled alpha-melanocyte stimulating hormone peptide analogues. Bioconjug Chem 14:1177–1184
Miao Y, Gallazzi F, Guo H, Quinn TP (2008) In-111-labeled lactam bridge-cyclized alpha-melanocyte stimulating hormone peptide analogues for melanoma imaging. Bioconjug Chem 19:539–547. doi:10.1021/bc700317w
Morais M, Oliveira BL, Correia JD, Oliveira MC, Jiménez MA, Santos I, Raposinho PD (2013) Influence of the bifunctional chelator on the pharmacokinetic properties of 99mTc(CO)3-labeled cyclic α-melanocyte stimulating hormone analog. J Med Chem 56:1961–1973. doi:10.1021/jm301647t
Ni XP, Bhargava A, Pearce D, Humphreys MH (2006) Modulation by dietary sodium intake of melanocortin 3 receptor mRNA and protein abundance in the rat kidney. Am J Physiol Regul Integr Comp Physiol 290:R560–R567
Petrangolini G, Pratesi G, De Cesare M, Supino R, Pisano C, Marcellini M, Giordano V, Laccabue D, Lanzi C, Zunino F (2003) Antiangiogenic effects of the novel camptothecin ST1481 (gimatecan) in human tumor xenografts. Mol Cancer Res 1:863–870
Raposinho PD, Xavier C, Correia JDG, Falcao S, Gomes P, Santos I (2008) Melanoma targeting with alpha-melanocyte stimulating hormone analogs labeled with fac-[Tc-99m(CO)(3)](+): effect of cyclization on tumor-seeking properties. J Biol Inorgan Chem 13:449–459. doi:10.1007/s00775-007-0338-3
Ringholm A, Klovins J, Rudzish R, Phillips S, Rees JL, Schiöth HB (2004) Pharmacological characterization of loss of function mutations of the human melanocortin 1 receptor that are associated with red hair. J Invest Dermatol 123:917–923
Rudman D, Hollins BM, Kutner MH, Moffitt SD, Lynn MJ (1983) Three types of alpha-melanocyte-stimulating hormone: bioactivities and half-lives. Am J Physiol 245:E47–E54
Salazar-Onfray F, López M, Lundqvist A, Aguirre A, Escobar A, Serrano A, Korenblit C, Petersson M, Chhajlani V, Larsson O, Kiessling R (2002) Tissue distribution and differential expression of melanocortin 1 receptor, a malignant melanoma marker. Br J Cancer 87:414–422
Sawyer TK, Sanfilippo PJ, Hruby VJ, Engel MH, Heward CB, Burnett JB, Hadley ME (1980) 4-Norleucine, 7-d-phenylalanine-alpha-melanocyte-stimulating hormone: a highly potent alpha-melanotropin with ultralong biological activity. Proc Natl Acad Sci U S A 77:5754–5758
Schiöth HB, Chhajlani V, Muceniece R, Klusa V, Wikberg JE (1996) Major pharmacological distinction of the ACTH receptor from other melanocortin receptors. Life Sci 59:797–801
Schiöth HB, Yook P, Muceniece R, Wikberg JE, Szardenings M (1998) Chimeric melanocortin MC1 and MC3 receptors: identification of domains participating in binding of melanocyte-stimulating hormone peptides. Mol Pharmacol 54:154–161
Schlesinger J, Rajander J, Ihalainen JA, Ramesh D, Eklund P, Fagerholm V, Nuutila P, Solin O (2011) Isomerism of [64Cu-NOTA-Bn]-labeled radiotracers: separation of two complex isomers and determination of their interconversion energy barrier using ion pair chromatography. Inorg Chem 50:4260–4271. doi:10.1021/ic101775q
Schottelius M, Wester HJ (2009) Molecular imaging targeting peptide receptors. Methods 48:161–177. doi:10.1016/j.ymeth.2009.03.012
Shao Y, Liang W, Kang F, Yang W, Ma X, Li G, Zong S, Chen K, Wang J (2014) 68Ga-labeled cyclic NGR peptide for microPET imaging of CD13 receptor expression. Molecules 19:11600–11612. doi:10.3390/molecules190811600
Si J, Ge Y, Zhuang S, Wang LJ, Chen S, Gong R (2013) Adrenocorticotropic hormone ameliorates acute kidney injury by steroidogenic-dependent and -independent mechanisms. Kidney Int 83:635–646. doi:10.1038/ki.2012.447
Siegrist W, Eberle AN (1995) Melanocortins and their implication in melanoma. Trends Endocrinol Metab 6:115–120
Siegrist W, Oestreicher M, Stutz S, Girard J, Eberle AN (1988) Radioreceptor assay for alpha-MSH using mouse B16 melanoma cells. J Recept Res 8:323–343
Siegrist W, Solca F, Stutz S, Giuffre L, Carrel S, Girard J, Eberle AN (1989) Characterization of Receptors for Alpha-Melanocyte-Stimulating Hormone on Human-Melanoma Cells. Cancer Res 49:6352–6358
Siegrist W, Willard DH, Wilkison WO, Eberle AN (1996) Agouti protein inhibits growth of B16 melanoma cells in vitro by acting through melanocortin receptors. Biochem Biophys Res Commun 218:171–175
Tandler N, Mosch B, Pietzsch J (2012) Protein and non-protein biomarkers in melanoma: a critical update. Amino Acids 43:2203–2230. doi:10.1007/s00726-012-1409-5
Tatro JB, Entwistle ML, Lester BR, Reichlin S (1990) Melanotropin receptors of murine melanoma characterized in cultured cells and demonstrated in experimental tumors in situ. Cancer Res 50:1237–1242
Yang J, Guo H, Padilla RS, Berwick M, Miao Y (2010) Replacement of the Lys linker with an Arg linker resulting in improved melanoma uptake and reduced renal uptake of Tc-99 m-labeled Arg-Gly-Asp-conjugated alpha-melanocyte stimulating hormone hybrid peptide. Bioorg Med Chem 18:6695–6700
Acknowledgments
Feng Gao thanks the German Academic Exchange Service (DAAD) for financial support (A/11/94299, 91540213). The excellent technical assistance of Regina Herrlich, Utta Herzog and Andrea Suhr is greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The animal experiments were performed in accordance to the guidelines of the German Regulations of Animal Welfare. The protocol was approved by the local Ethical Committee for Animal Experiments (reference number 24-9168.21-4/2004-1).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gao, F., Sihver, W., Jurischka, C. et al. Radiopharmacological characterization of 64Cu-labeled α-MSH analogs for potential use in imaging of malignant melanoma. Amino Acids 48, 833–847 (2016). https://doi.org/10.1007/s00726-015-2131-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-015-2131-x