Abstract
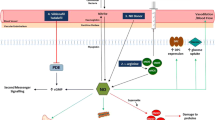
The l-arginine/nitric oxide (L-Arg/NO) pathway regulates endothelial function and may play an important role in the pathogenesis of Duchenne muscular dystrophy (DMD). Yet, this pathway is poorly investigated in children suffering from DMD. Endothelial dysfunction can affect the perfusion of contracting muscles, thus leading to ischemia and hypoxia. In the present study, we tested the hypothesis that reduced NO production due to elevated synthesis of N G,N G-dimethyl-l-arginine (asymmetric dimethylarginine, ADMA), an endogenous inhibitor of NO synthesis, is a possible pathophysiological mechanism for progressive intramuscular muscle ischemia and disturbed endothelial function in children with DMD. Given the possible antagonistic action of homoarginine (hArg) on ADMA, we also analyzed this amino acid. We investigated 55 male patients with DMD and 54 healthy male controls (HC; aged 11.9 ± 4.8 vs. 11.1 ± 4.9 years, mean ± SD). Urinary creatinine and metabolites of the L-Arg/NO pathway were measured in plasma and urine by GC–MS or GC–MS/MS. Urine levels of ADMA and its major urinary metabolite dimethylamine (DMA), nitrite and nitrate (P < 0.001 for all) and hArg (P = 0.002) were significantly higher in DMD patients compared to HC, while the urinary DMA/ADMA molar ratio was lower (P = 0.002). In plasma, nitrate (P < 0.001), hArg (P = 0.002) and the hArg/ADMA ratio (P < 0.001) were lower in DMD than in HC. In plasma, ADMA (631 ± 119 vs. 595 ± 129 nM, P = 0.149), arginine and nitrite did not differ between DMD and HC. In DMD, positive correlations between ADMA, DMA or nitrate excretion and the stage of disease (according to Vignos and Thompson) were found. In DMD patients on steroid medication, lower concentrations of ADMA in plasma, and of DMA, ADMA, nitrate and hArg in urine were observed compared to non-treated patients. The L-Arg/NO pathway is impaired in DMD patients, with the disease progression being clinically negatively correlated with the extent of impairment. One of the underlying mechanisms in DMD may involve insufficient antagonism of ADMA by hArg. Steroids, but not creatine supplementation, seems to improve the L-Arg/NO pathway in DMD.

Similar content being viewed by others
Abbreviations
- ACE:
-
Angiotensin-converting enzyme
- ADMA:
-
Asymmetric dimethylarginine (N G,N G-dimethyl-l-arginine)
- AGAT:
-
Arginineglycine amidinotransferase
- DDAH:
-
Dimethylarginine dimethylaminohydrolase
- DMA:
-
Dimethylamine
- DMD:
-
Duchenne muscular dystrophy
- EDRF:
-
Endothelium-derived relaxing factor
- GAMT:
-
Guanidinoacetate N-methyltransferase
- GC–MS:
-
Gas chromatography–mass spectrometry
- GC–MS/MS:
-
Gas chromatography–tandem mass spectrometry
- hArg:
-
Homoarginine
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- SDMA:
-
Symmetric dimethylarginine (N G,N G´-dimethyl-l-arginine)
- sVCAM-1:
-
Soluble vascular cell adhesion molecule-1
References
Aartsma-Rus A, Van Deutekom JC, Fokkema IF, Van Ommen GJ, Den Dunnen JT (2006) Entries in the Leiden Duchenne muscular dystrophy mutation database: an overview of mutation types and paradoxical cases that confirm the reading-frame rule. Muscle Nerve 34:135–144
Achan V, Broadhead M, Malaki M, Whitley G, Leiper J, MacAllister R, Vallance P (2003) Asymmetric dimethylarginine causes hypertension and cardiac dysfunction in humans and is actively metabolized by dimethylarginine dimethylaminohydrolase. Arterioscler Thromb Vasc Biol 23:1455–1459
Annexstad EJ, Lund-Petersen I, Rasmussen M (2014) Duchenne muscular dystrophy. Tidsskr Nor Laegeforen 134:1361–1364
Arahata K, Ishiura S, Ishiguro T, Tsukahara T, Suhara Y, Eguchi C, Ishihara T, Nonaka I, Ozawa E, Sugita H (1988) Immunostaining of skeletal and cardiac muscle surface membrane with antibody against Duchenne muscular dystrophy peptide. Nature 333(6176):861–863
Banerjee B, Sharma U, Balasubramanian K, Kalaivani M, Kalra V, Jagannathan NR (2010) Effect of creatine monohydrate in improving cellular energetics and muscle strength in ambulatory Duchenne muscular dystrophy patients: a randomized, placebo-controlled 31P MRS study. Magn Reson Imaging 28(5):698–707
Barnes PJ (2010) Mechanisms and resistance in glucocorticoid control of inflammation. J Steroid Biochem Mol Biol 120:76–85
Böger RH, Bode-Böger SM, Thiele W, Junker W, Alexander K, Frölich JC (1997) Biochemical evidence for impaired nitric oxide synthesis in patients with peripheral arterial occlusive disease. Circulation 95:2068–2074
Böhmer A, Gambaryan S, Tsikas D (2014) Human blood platelets lack nitric oxide synthase activity. Platelets. doi:10.3109/09537104.2014.974024
Bonati U, Hafner P, Erne B, Thomas E, Rutz E, Frank S, Hilker C, Deuster S, Gloor M, Bieri O, Sinnreich M, Fischmann A, Fischer D (2015) Improved muscle function in Duchenne muscular dystrophy using a combination of l-arginine and metformin. Neuropedoatrics 46:PS02-34. doi:10.1055/s-0035-1550745
Brenman JE, Chao DS, Xia H, Aldape K, Bredt DS (1995) Nitric oxide synthase complexed with dystrophin and absent from skeletal muscle sarcolemma in Duchenne muscular dystrophy. Cell 82:743–752
Chavoshan B, Sander M, Sybert T, Hansen J, Victor R, Thomas G (2002) Nitric oxide-dependent modulation of sympathetic neural control of oxygenation in exercising human skeletal muscle. J Physiol 540(Pt 1):377–386
Chobanyan-Jürgens K, Fuchs AJ, Tsikas D, Kanzelmeyer N, Das AM, Illsinger S, Vaske B, Jordan J, Lücke T (2012a) Increased asymmetric dimethylarginine (ADMA) dimethylaminohydrolase (DDAH) activity in childhood hypercholesterolemia type II. Amino Acids 43:805–811
Chobanyan-Jürgens K, Schwarz A, Böhmer A, Beckmann B, Gutzki FM, Michaelsen JT, Stichtenoth DO, Tsikas D (2012b) Renal carbonic anhydrases are involved in the reabsorption of endogenous nitrite. Nitric Oxide 26:126–131
Cooke JP (2000) Does ADMA cause endothelial dysfunction? Arterioscler Thromb Vasc Biol 20:2032–2037
Cutolo M, Seriolo B, Pizzorni C, Secchi ME, Soldano S, Paolino S, Montagna P, Sulli A (2008) Use of glucocorticoids and risk of infections. Autoimmun Rev 8:153–155
da Silva RP, Clow K, Brosnan JT, Brosnan ME (2014) Synthesis of guanidinoacetate and creatine from amino acids by rat pancreas. Br J Nutr 111:571–577
De Paepe B, De Bleecker JL (2013) Cytokines and chemokines as regulators of skeletal muscle inflammation: presenting the case of Duchenne muscular dystrophy. Mediators Inflamm 2013:540370
Derave W, Marescau B, Vanden Eede E, Eijnde BO, De Deyn PP, Hespel P (2004) Plasma guanidino compounds are altered by oral creatine supplementation in healthy humans. J Appl Physiol 97:852–857
Evans NP, Misyak SA, Robertson JL, Bassaganya-Riera J, Grange RW (2009) Dysregulated intracellular signaling and inflammatory gene expression during initial disease onset in Duchenne muscular dystrophy. Am J Phys Med Rehabil 88:502–522
Förstermann U, Closs EI, Pollock JS, Nakane M, Schwarz P, Gath I, Kleinert H (1994) Nitric oxide synthase isozymes. Characterization, purification, molecular cloning, and functions. Hypertension 23 (6 Pt 2):1121–1131
Furchgott RF, Zawadzki JV (1980) The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 288(5789):373–376
Furchgott RF, Cherry PD, Zawadzki JV, Jothianandan D (1984) Endothelial cells as mediators of vasodilation of arteries. J Cardiovasc Pharmacol 6(Suppl 2):S336–S343
Goonasekera CD, Rees DD, Woolard P, Frend A, Shah V, Dillon MJ (1997) Nitric oxide synthase inhibitors and hypertension in children and adolescents. J Hypertens 15:901–909
Gücüyener K, Ergenekon E, Erbas D, Pinarli G, Serdaroğlu A (2000) The serum nitric oxide levels in patients with Duchenne muscular dystrophy. Brain Dev 22:181–183
Inoue R, Miyake M, Kanazawa A, Sato M, Kakimoto Y (1979) Decrease of 3-methylhistidine and increase of NG, NG-dimethylarginine in the urine of patients with muscular dystrophy. Metabolism 28:801–804
Ito A, Tsao PS, Adimoolam S, Kimoto M, Ogawa T, Cooke JP (1999) Novel mechanism for endothelial dysfunction: dysregulation of dimethylarginine dimethylaminohydrolase. Circulation 99:3092–3095
Kakimoto Y, Akazawa S (1970) Isolation and identification of N-G, N-G- and N-G, N’-G-dimethyl-arginine, N-epsilon-mono-, di-, and trimethyllysine, and glucosylgalactosyl- and galactosyl-delta-hydroxylysine from human urine. J Biol Chem 245:5751–5758
Kanzelmeyer N, Tsikas D, Chobanyan-Jürgens K, Beckmann B, Vaske B, Illsinger S, Das AM, Lücke T (2012) Asymmetric dimethylarginine in children with homocystinuria or phenylketonuria. Amino Acids 42:1765–1772
Kanzelmeyer NK, Pape L, Chobanyan-Jürgens K, Tsikas D, Hartmann H, Fuchs AJ, Vaske B, Das AM, Haubitz M, Jordan J, Lücke T (2014) l-arginine/NO pathway is altered in children with haemolytic-uraemic syndrome (HUS). Oxid Med Cell Longev 2014:203512
Kasai T, Abeyama K, Hashiguchi T, Fukunaga H, Osame M, Maruyama I (2004) Decreased total nitric oxide production in patients with Duchenne muscular dystrophy. J Biomed Sci 11:534–537
Kayacelebi AA, Nguyen TH, Neil C, Horowitz JD, Jordan J, Tsikas D (2014a) Homoarginine and 3-nitrotyrosine in patients with takotsubo cardiomyopathy. Int J Cardiol 173:546–547
Kayacelebi AA, Beckmann B, Gutzki FM, Jordan J, Tsikas D (2014b) GC-MS and GC-MS/MS measurement of the cardiovascular risk factor homoarginine in biological samples. Amino Acids 46:2205–2217
Kayacelebi AA, Knöfel A-K, Beckmann B, Hanff E, Warnecke G, Tsikas D (2015) Measurement of unlabeled and stable isotope-labeled homoarginine, arginine and their metabolites in biological samples by GC–MS and GC–MS/MS. Amino Acids. doi:10.1007/s00726-015-1984-3
Kielstein JT, Böger RH, Bode-Böger SM, Schäffer J, Barbey M, Koch KM, Frölich JC (1999) Asymmetric dimethylarginine plasma concentrations differ in patients with end-stage renal disease: relationship to treatment method and atherosclerotic disease. J Am Soc Nephrol 10:594–600
Kielstein A, Tsikas D, Galloway GP, Mendelson JE (2007) Asymmetric dimethylarginine (ADMA)–a modulator of nociception in opiate tolerance and addiction? Nitric Oxide 17:55–59
Kley RA, Tarnopolsky MA, Vorgerd M (2013) Creatine for treating muscle disorders. Cochrane Database Syst Rev 6:CD004760. doi:10.1002/14651858.CD004760.pub4
Koenig M, Hoffman EP, Bertelson CJ, Monaco AP, Feener C, Kunkel LM (1987) Complete cloning of the Duchenne muscular dystrophy (DMD) cDNA and preliminary genomic organization of the DMD gene in normal and affected individuals. Cell 50:509–517
Kruszelnicka O, Chyrchel B, Golay A, Surdacki A (2015) Differential associations of circulating asymmetric dimethylarginine and cell adhesion molecules with metformin use in patients with type 2 diabetes mellitus and stable coronary artery disease. Amino Acids. doi:10.1007/s00726-015-1976-3
Leiper J, Vallance P (2006) The synthesis and metabolism of asymmetric dimethylarginine (ADMA). Eur J Clin Pharmacol 62:33–38
Leung DG, Germain-Lee EL, Denger BE, Wagner KR (2011) Report on the second endocrine aspects of Duchenne muscular dystrophy conference December 1–2, 2010, Baltimore, Maryland, USA. Neuromuscul Disord 21:594–601
Lücke T, Tsikas D, Kanzelmeyer N, Vaske B, Das AM (2006a) Elevated plasma concentrations of the endogenous nitric oxide synthase inhibitor asymmetric dimethylarginine in citrullinemia. Metabolism 55:1599–1603
Lücke T, Tsikas D, Kanzelmeyer NK, Boerkoel CF, Clewing JM, Vaske B, Ehrich JH, Das AM (2006b) Vaso-occlusion in Schimke-immuno-osseous dysplasia: is the NO pathway involved? Horm Metab Res 38:678–682
Lücke T, Kanzelmeyer N, Kemper MJ, Tsikas D, Das AM (2007) Developmental changes in the l-arginine/nitric oxide pathway from infancy to adulthood: plasma asymmetric dimethylarginine levels decrease with age. Clin Chem Lab Med 45:1525–1530
Lücke T, Kanzelmeyer N, Chobanyan K, Tsikas D, Franke D, Kemper MJ, Ehrich JH, Das AM (2008) Elevated asymmetric dimethylarginine (ADMA) and inverse correlation between circulating ADMA and glomerular filtration rate in children with sporadic focal segmental glomerulosclerosis (FSGS). Nephrol Dial Transplant 23:734–740
Manzur AY, Kuntzer T, Pike M, Swan A (2008) Glucocorticoid corticosteroids for Duchenne muscular dystrophy. Cochrane Database Syst Rev (1):CD003725. doi:10.1002/14651858.CD003725.pub3
Marletta MA (1993) Nitric oxide synthase structure and mechanism. J Biol Chem 268:12231–12234
März W, Meinitzer A, Drechsler C, Pilz S, Krane V, Kleber ME, Fischer J, Winkelmann BR, Böhm BO, Ritz E, Wanner C (2010) Homoarginine, cardiovascular risk, and mortality. Circulation 122:967–975
Moali C, Boucher JL, Sari MA, Stuehr DJ, Mansuy D (1998) Substrate specificity of NO synthases: detailed comparison of l-arginine, homo-l-arginine, their N omega-hydroxy derivatives, and N omega-hydroxynor-l-arginine. Biochemistry 37(29):10453–10460
Moncada S, Higgs EA (2006) The discovery of nitric oxide and its role in vascular biology. Br J Pharmacol 147(Suppl 1):S193–S201
Mortier W (1994) Muskel- und Nervenerkrankungen im Kindesalter. Thieme, Stuttgart
Palmer RM, Ferrige AG, Moncada S (1987) Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 327(6122):524–526
Palmer RM, Ashton DS, Moncada S (1988) Vascular endothelial cells synthesize nitric oxide from l-arginine. Nature 333(6174):664–666
Pham VV, Stichtenoth DO, Tsikas D (2009) Nitrite correlates with 3-nitrotyrosine but not with the F(2)-isoprostane 15(S)-8-iso-PGF(2alpha) in urine of rheumatic patients. Nitric Oxide 21:210–215
Pilz S, Meinitzer A, Tomaschitz A, Drechsler C, Ritz E, Krane V, Wanner C, Boehm BO, März W (2011) Low homoarginine concentration is a novel risk factor for heart disease. Heart 97(15):1222–1227
Pilz S, Teerlink T, Scheffer PG, Meinitzer A, Rutters F, Tomaschitz A, Drechsler C, Kienreich K, Nijpels G, Stehouwer CD, März W, Dekker JM (2014) Homoarginine and mortality in an older population: the Hoorn study. Eur J Clin Invest 44:200–208
Pilz S, Meinitzer A, Gaksch M, Grübler M, Verheyen N, Drechsler C, Hartaigh BÓ, Lang F, Alesutan I, Voelkl J, März W, Tomaschitz A (2015) Homoarginine in the renal and cardiovascular systems. Amino Acids. doi:10.1007/s00726-015-1993-2
Radomski MW, Palmer RM, Moncada S (1990) Glucocorticoids inhibit the expression of an inducible, but not the constitutive, nitric oxide synthase in vascular endothelial cells. Proc Natl Acad Sci 87:10043–10047
Rees DD, Palmer RM, Moncada S (1989) Role of endothelium-derived nitric oxide in the regulation of blood pressure. Proc Natl Acad Sci 86:3375–3378
Roberts JJ, Walker JB (1985) Higher homolog and N-ethyl analog of creatine as synthetic phosphagen precursors in brain, heart, and muscle, repressors of liver amidinotransferase, and substrates for creatine catabolic enzymes. J Biol Chem 260:13502–13508
Ryan WL, Wells IC (1964) Homocitrulline and homoarginine synthesis from lysine. Science 144(3622):1122–1127
Ryan WL, Barak AJ, Johnson RJ (1968) Lysine, homocitrulline, and homoarginine metabolism by the isolated perfused rat liver. Arch Biochem Biophys 123:294–297
Sibal L, Agarwal SC, Home PD, Boger RH (2010) The role of asymmetric dimethylarginine (ADMA) in endothelial dysfunction and cardiovascular disease. Curr Cardiol Rev 6:82–90
Spencer MJ, Tidball JG (2001) Do immune cells promote the pathology of dystrophin-deficient myopathies? Neuromuscul Disord 11:556–564
Tarnopolsky MA, Mahoney DJ, Vajsar J, Rodriguez C, Doherty TJ, Roy BD, Biggar D (2004) Creatine monohydrate enhances strength and body composition in Duchenne muscular dystrophy. Neurology 62:1771–1777
Terrill JR, Radley-Crabb HG, Iwasaki T, Lemckert FA, Arthur PG, Grounds MD (2013) Oxidative stress and pathology in muscular dystrophies: focus on protein thiol oxidation and dysferlinopathies. FEBS J 280:4149–4164
Thomas GD, Sander M, Lau KS, Huang PL, Stull JT, Victor RG (1998) Impaired metabolic modulation of alpha-adrenergic vasoconstriction in dystrophin-deficient skeletal muscle. Proc Natl Acad Sci USA 95:15090–15095
Thompson RA, Vignos PJ Jr (1959) Serum aldolase in muscle disease. AMA archives of internal medicine 103(4):551–564
Tran CT, Leiper JM, Vallance P (2003) The DDAH/ADMA/NOS pathway. Atheroscler Suppl 4:33–40
Tsikas D (2000) Simultaneous derivatization and quantification of the nitric oxide metabolites nitrite and nitrate in biological fluids by gas chromatography/mass spectrometry. Anal Chem 72:4064–4072
Tsikas D (2005) Methods of quantitative analysis of the nitric oxide metabolites nitrite and nitrate in human biological fluids. Free Radic Res 39:797–815
Tsikas D (2008) A critical review and discussion of analytical methods in the l-arginine/nitric oxide area of basic and clinical research. Anal Biochem 379:139–163
Tsikas D (2015) Circulating and excretory nitrite and nitrate: their value as measures of nitric oxide synthesis, bioavailability and activity is inherently limited. Nitric Oxide 45:1–3
Tsikas D, Kayacelebi AA (2014) Do homoarginine and asymmetric dimethylarginine act antagonistically in the cardiovascular system? Circ J 78:2094–2095
Tsikas D, Sandmann J, Savva A, Luessen P, Böger RH, Gutzki FM, Mayer B, Frölich JC (2000) Assessment of nitric oxide synthase activity in vitro and in vivo by gas chromatography-mass spectrometry. J Chromatogr B 742:143–153
Tsikas D, Schubert B, Gutzki FM, Sandmann J, Frölich JC (2003) Quantitative determination of circulating and urinary asymmetric dimethylarginine (ADMA) in humans by gas chromatography-tandem mass spectrometry as methyl ester tri(N-pentafluoropropionyl) derivative. J Chromatogr B 798:87–99
Tsikas D, Thum T, Becker T, Pham VV, Chobanyan K, Mitschke A, Beckmann B, Gutzki FM, Bauersachs J, Stichtenoth DO (2007) Accurate quantification of dimethylamine (DMA) in human urine by gas chromatography-mass spectrometry as pentafluorobenzamide derivative: evaluation of the relationship between DMA and its precursor asymmetric dimethylarginine (ADMA) in health and disease. J Chromatogr B 851:229–239
Tsikas D, Wolf A, Mitschke A, Gutzki FM, Will W, Bader M (2010a) GC-MS determination of creatinine in human biological fluids as pentafluorobenzyl derivative in clinical studies and biomonitoring: inter-laboratory comparison in urine with Jaffé, HPLC and enzymatic assays. J Chromatogr B 878:2582–2592
Tsikas D, Schwarz A, Stichtenoth DO (2010b) Simultaneous measurement of [15N]nitrate and [15N]nitrite enrichment and concentration in urine by gas chromatography mass spectrometry as pentafluorobenzyl derivatives. Anal Chem 82:2585–2587
Tsikas D, Niemann J, Flentje M, Schwarz A, Tossios P (2014) N-Acetylcysteine (NAC) inhibits renal nitrite and nitrate reabsorption in healthy subjects and in patients undergoing cardiac surgery: risk of nitric oxide (NO) bioavailability by NAC? Int J Cardiol 177:30–33
Valtonen P, Laitinen T, Lyyra-Laitinen T, Raitakari OT, Juonala M, Viikari JS, Heiskanen N, Vanninen E, Punnonen K, Heinonen S (2008) Serum L-homoarginine concentration is elevated during normal pregnancy and is related to flow-mediated vasodilatation. Circ J 72:1879–1884
van der Zwan LP, Scheffer PG, Dekker JM, Stehouwer CD, Heine RJ, Teerlink T (2011) Systemic inflammation is linked to low arginine and high ADMA plasma levels resulting in an unfavourable NOS substrate-to-inhibitor ratio: the Hoorn Study. Clin Sci (Lond) 121:71–78
Walker JB, Hannan JK (1976) Creatine biosynthesis during embryonic development. False feedback suppression of liver amidinotransferase by N-acetimidoylsarcosine and 1-carboxymethyl-2-iminoimidazolidine (cyclocreatine). Biochemistry 15:2519–2522
Warnes DM, Tomas FM, Ballard FJ (1981) Increased rates of myofibrillar protein breakdown in muscle-wasting diseases. Muscle Nerve 4:62–66
Zoccali C, Maas R, Cutrupi S, Pizzini P, Finocchiaro P, Cambareri F, Panuccio V, Martorano C, Schulze F, Enia G, Tripepi G, Boger R (2007) Asymmetric dimethyl-arginine (ADMA) response to inflammation in acute infections. Nephrol Dial Transplant 22:801–806
Acknowledgments
The authors thank A. Mitschke and M. T. Suchy for excellent laboratory assistance and F. M. Gutzki for performing GC–MS and GC–MS/MS analyses. We are also grateful for the support in recruiting DMD patients from the Department of Neuropediatrics in the University Hospital Essen.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
The Ethics Committees of the Faculty of Medicine at Ruhr-University Bochum and of the Hannover Medical School approved the study. Written consent was given by each participant and his parents or only by the participant if he was 18 years or older.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hörster, I., Weigt-Usinger, K., Carmann, C. et al. The l-arginine/NO pathway and homoarginine are altered in Duchenne muscular dystrophy and improved by glucocorticoids. Amino Acids 47, 1853–1863 (2015). https://doi.org/10.1007/s00726-015-2018-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-015-2018-x