Abstract
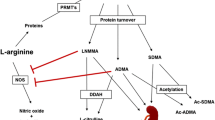
Free, non-protein bound asymmetrically guanidine-dimethylated arginine (ADMA) is an endogenous inhibitor of nitric oxide (NO) synthesis. Human erythrocytic membrane comprises considerable amounts of large (>50 kDa) ADMA-containing proteins. Location in the erythrocyte membrane and identity and physiological functions of ADMA-containing proteins are unrevealed. In healthy subjects, the concentration of free ADMA in heparinised plasma is almost identical to that of serum. We hypothesised that the robustness of free ADMA concentration in human blood is due to a remarkable resistance of erythrocytic ADMA-containing proteins against proteases. In vivo, we investigated the course of the concentration of ADMA in serum and EDTA plasma of a critically ill patient with acute kidney injury during extended haemodialysis. In vitro, we studied the effects of thermolysin, a useful experimental proteolytic enzyme of erythrocyte membrane proteins, on erythrocytic ADMA. The protein binding (PB) of ADMA to human serum albumin (HSA) was also determined. In these studies, ADMA was measured by a previously reported, fully validated GC–MS/MS method. We measured almost identical ADMA concentrations in plasma and serum samples of the patient. During dialysis, the circulating ADMA concentration decreased slowly and moderately indicating removal of this substance, which was however much less than expected from its low molecular weight (202 Da) and high water solubility. After dialysis, circulating ADMA concentration increased again, a phenomenon called rebound, and ADMA reached higher levels compared to the baseline. The PB value of ADMA to HSA was about 30 %. This surprisingly high PB value of ADMA to HSA may be an explanation for the rather poor dialysance of ADMA. Washed human erythrocytes suspended in phosphate-buffered physiological saline were found not to release appreciable amounts of free and ADMA-containing proteins. The lack of effect of coagulation or anticoagulation on the concentration of circulating free ADMA in humans is likely to be due to a remarkable resistance of ADMA-containing proteins in the erythrocyte membrane against proteases in vivo in humans. Our study suggests that free ADMA is released in the circulating blood at relatively high rates. The considerable PB of ADMA to HSA is likely to add to the apparently poor dialysability of ADMA. Other contributing factors could be redistribution of free ADMA between plasma and erythrocytes in favour of plasma ADMA and parallel formation of free ADMA from erythrocytic ADMA-containing proteins during haemodialysis.






Similar content being viewed by others
Abbreviations
- ADMA:
-
Asymmetric dimethylarginine (N G,N G-dimethyl-l-arginine)
- CID:
-
Collision-induced dissociation
- DDAH:
-
Dimethylarginine dimethylaminohydrolase
- DMA:
-
Dimethylamine
- ECNICI:
-
Electron-capture negative-ion chemical ionization
- GC–MS/MS:
-
Gas chromatography-tandem mass spectrometry
- HSA:
-
Human serum albumin
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- PB:
-
Protein binding
- PBS:
-
Phosphate buffer saline
- SDMA:
-
Symmetric dimethylarginine (N G,N′ G-dimethyl-l-arginine)
References
Adekoya OA, Sylte I (2009) The thermolysin family (M4) of enzymes: therapeutic and biotechnological potential. Chem Biol Drug Des 73:7–16
Araujo P (2009) Key aspects of analytical method validation and linearity evaluation. J Chromatogr B 877:2224–2234
Billecke SS, Kitzmiller LA, Northrup JJ, Whitesall SE, Kimoto M, Hinz AV, D´Alecy LG (2006) Contribution of whole blood to the control of plasma asymmetrical dimethylarginine. Am J Physiol Heart Circ Physiol 291:H1788–H1796
Billecke SS, D`Alecy LG, Platel R, Whitesall SE, Jamerson KA, Perlman RL, Gadegbeku CA (2009) Blood content of asymmetric dimethylarginine: new insights into its dysregulation in renal disease. Nephrol Dial Transplant 24:489–496
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1:307–310
Böhmer A, Großkopf H, Jordan J, Tsikas D (2012a) Human hemoglobin does not contain asymmetric dimethylarginine (ADMA). Nitric Oxide 27:72–74
Böhmer A, Beckmann B, Sandmann J, Tsikas D (2012b) Doubts concerning functional endothelial nitric oxide synthase in human erythrocytes. Blood 119:1322–1323
Bouteldja N, Woodman RJ, Hewitson CL, Domingo E, Barbara JA, Mangoni AA (2013) Methylated arginines and nitric oxide in end-stage renal disease: impact of inflammation, oxidative stress and haemodialysis. Biomarkers 18:357–364
Burkhardt O, Hafer C, Langhoff A, Kaever V, Kumar V, Welte T, Haller H, Fliser D, Kielstein JT (2009) Pharmacokinetics of ertapenem in critically ill patients with acute renal failure undergoing extended daily dialysis. Nephrol Dial Transplant 24:267–271
D`Alecy LG, Billecke SS (2010) Massive quantities of asymmetric dimethylarginine (ADMA) are incorporated in red blood cell proteins and may be released by proteolysis following hemolytic stress. Blood Cells Mol Dis 45:40
Damoglou AP, Lindley H, Stapleton IW (1971) The hydrolysis by thermolysin of dipeptide derivatives that contain substituted cysteine. Biochem J 123:379–384
Davids M, van Hell AJ, Visser M, Nijveldt RJ, van Leeuwen PAM, Teerlink T (2012) Role of the human erythrocyte in generation and storage of asymmetric dimethylarginine. Am J Physiol Heart Circ Physiol 302:H1762–H1770
Davids M, Peters JHC, de Jong S, Teerlink T (2013) Measurement of nitric oxide-related amino acids in serum and plasma: effects of blood clotting and type of anticoagulant. Clin Chim Acta 421C:164–167
Eksborg S (1981) Evaluation of method-comparison data. Clin Chem 27:1311–1312
Ellmerer M, Schaupp L, Brunner GA, Sendlhofer G, Wutte A, Wach P, Pieber TR (2000) Measurement of interstitial albumin in human skeletal muscle and adipose tissue by open-flow microperfusion. Am J Physiol Endocrinol Metab 278:E352–E356
Feder J, Garrett LR, Wildi BS (1971) Studies on the role of calcium in thermolysin. Biochemistry 10:4552–4556
Girg R, Rudolph R, Jaenicke R (1981) Limited proteolysis of porcine-muscle lactic dehydrogenase by thermolysin during reconstitution yields dimers. Eur J Biochem 119:301–305
Großkopf H, Böhmer A, Tsikas D (2012) Letter to the editor: “Role of the human erythrocyte in generation and storage of asymmetric dimethylarginine”. Am J Physiol Heart Circ Physiol 303:751–752
Horowitz JD, Heresztyn T (2007) An overview of plasma concentrations of asymmetric dimethylarginine (ADMA) in health and disease and in clinical studies: methodological considerations. J Chromatogr B 851:42–50
Ikizler TA (2005) Effects of haemodialysis on protein metabolism. J Ren Nutr 15:39–43
Jenkins RE, Tanner JA (1977) The structure of the major protein of the human erythrocyte membrane. Characterization of the intact protein and major fragments. Biochem J 161:139–147
Kang ES, Cates TB, Harper DN, Chiang TM, Myers LK, Acchiardo SR, Kimoto M (2011) An enzyme hydrolyzing methylated inhibitors of nitric oxide synthase is present in circulating human red blood cells. Free Rad Res 35:693–707
Karim AR, Rees WD, Holman GD (1987) Binding of cytochalasin B to trypsin and thermolysin fragments of the human erythrocyte hexose transporter. Biochim Biophys Acta 902:402–405
Kielstein JT, Böger RH, Bode-Böger SM, Schäffer J, Barbey M, Koch KM, Frölich JC (1999) Asymmetric dimethylarginine plasma concentrations differ in patients with end-stage renal disease: relationship to treatment method and atherosclerotic disease. J Am Soc Nephrol 10:594–600
Kielstein JT, Böger RH, Bode-Böger SM, Martens-Lobenhoffer J, Lonnemann G, Frölich JC, Haller H, Fliser D (2004) Low dialysance of asymmetric dimethylarginine (ADMA)—in vivo and in vitro evidence of significant protein binding. Clin Nephrol 62:295–300
Kielstein A, Tsikas D, Galloway GP, Mendelson JE (2007) Asymmetric dimethylarginine (ADMA)—a modulator of nociception in opiate tolerance and addiction? Nitric Oxide 17:55–59
Kleinbongard P, Schulz R, Rassaf T, Lauer T, Dejam A, Jax T, Kumara I, Gharini P, Kabanova S, Ozüyaman B, Schnürch HG, Gödecke A, Weber AA, Robenek M, Robenek H, Bloch W, Rösen P, Kelm M (2006) Red blood cells express a functional endothelial nitric oxide synthase. Blood 107:2943–2951
Komori Y, Nonogaki T, Nikai T (2001) Hemorrhagic activity and muscle damaging effect of Pseudomonas aeruginosa metalloproteinase (elastase). Toxicon 39:1327–1332
Kramer E, Routh JI (1973) The binding of salicylic acid and acetylsalicylic acid to human serum albumin. Clin Biochem 6:98–105
Kurz H, Trunk H, Weitz B (1977) Evaluation of methods to determine protein-binding of drugs: equilibrium dialysis, ultrafiltration, ultracentrifugation, gel filtration. Arzneimittelforschung 27:1373–1380
Leiper JM, Vallance P (2006) The synthesis and metabolism of asymmetric dimethylarginine (ADMA). Eur J Clin Pharmacol 26:33–38
Lenzen H, Tsikas T, Böger RH (2006) Asymmetric dimethylarginine (ADMA) and the risk for coronary heart disease. The multicenter CARDIAC study. Eur J Clin Pharmacol 62:45–49
May M, Batkai S, Zörner AA, Tsikas D, Jordan J, Engeli S (2014) Clinical evaluation of extracellular ADMA concentrations in human blood and adipose tissue. Int J Mol Sci 15:1189–1200
May M, Kayacelebi AA, Batkai S, Jordan J, Tsikas D, Engeli S (2015) Plasma and tissue homoarginine concentrations in healthy and obese humans. Amino Acids. doi:10.1007/s00726-015-1922-4
Morris ME, Levy G (1984) Renal clearance and serum protein binding of acetaminophen and its major conjugates in humans. J Pharm Sci 73:1038–1041
Oldham PD (1968) Measurement in medicine: the interpretation of numerical data. English Universities Press, London
Rifai K, Bode-Boeger SM, Martens-Lobenhoffer J, Ernst T, Kretschmer U, Hafer C, Fliser D, Manns MP, Kielstein JT (2010) Removal of asymmetric dimethylarginine during artificial liver support using fractionated plasma separation and adsorption. Scand J Gastroenterol 45:1110–1115
Schepers E, Speer T, Bode-Böger SM, Fliser D, Kielstein JT (2014) Dimethylarginines ADMA and SDMA: the real water-soluble small toxins? Semin Nephrol 34:97–105
Teerlink T (2007) HPLC analysis of ADMA and other methylated l-arginine analogs inn biological samples. J Chromatogr B 851:21–29
Trettin A, Modun D, Madunic S, Vukovic J, Radman M, Batkai S, Thum T, Jordan J, Tsikas D (2014) LC–MS/MS and GC–MS/MS measurement of plasma and urine di-paracetamol and 3-nitro-paracetamol: proof-of-concept studies on a novel human model of oxidative stress based on oral paracetamol administration. J Chromatogr B 959:71–81
Tsikas D (2007) Analysis of nitrite and nitrate in biological fluids by assays based on the Griess reaction: appraisal of the Griess reaction in the l-arginine/nitric oxide area of research. J Chromatogr B 851:51–70
Tsikas D (2008) Determination of asymmetric dimethylarginine in biological fluids: a paradigm for a successful analytical story. Curr Opin Clin Nutr Metab Care 11:592–600
Tsikas D (2009) De novo synthesis of trideuteromethyl esters of amino acids for use in GC–MS and GC–tandem MS exemplified for ADMA in in human plasma and urine: standardization, validation, comparison and proof of evidence for their aptitude as internal standards. J Chromatogr B 877:2308–2320
Tsikas D, Beckmann B (2009) Albumin from human serum does not contain asymmetric dimethylarginine (ADMA). Clin Biochem 42:1739–1740
Tsikas D, Böger RH, Sandmann J, Bode-Böger SM, Frölich JC (2000) Endogenous nitric oxide synthase inhibitors are responsible for the l-arginine paradox. FEBS Lett 478:1–3
Tsikas D, Schubert B, Gutzki FM, Sandmann J, Schwedhelm E, Frölich JC (2003) Quantitative determination of circulating and urinary asymmetric dimethylarginine (ADMA) in humans by gas chromatography-tandem mass spectrometry as methyl ester tri(N-pentafluoropropionyl) derivative. J Chromatogr B 798:87–99
Tsikas D, Engeli S, Beckmann B, Jordan J (2010) Asymmetric dimethylarginine (ADMA) is present in plasma proteins of healthy subjects at the low nmol-per-g-level. Nitric Oxide 22:316–317
Tsikas D, Böhmer A, Großkopf H, Beckmann B, Dreißigacker U, Jordan J, Maassen N (2012) Clinical-chemistry laboratory relevant hemolysis is unlikely to compromise human plasma concentration of free asymmetric dimethylarginine (ADMA). Clin Biochem 45:1536–1538
Vallance P, Leone A, Calver A, Collier J, Moncada S (1992) Accumulation of an endogenous inhibitor of nitric oxide synthesis in chronic renal failure. Lancet 339(8793):572–575
Zinellu A, Sotgia S, Scanu B, Formato M, Deiana L, Carru C (2007) Assessment of protein-incorporated arginine methylation in biological specimens by CZE UV detection. Electrophoresis 28:4452–4458
Zinellu A, Sotgia S, Scanu B, Deiana L, Carru C (2008) Determination of proteinincorporated methylated arginine reference values in healthy subjects whole blood and evaluation of factors affecting protein methylation. Clin Biochem 41:1218–1223
Acknowledgments
This study was supported by the Deutsche Forschungsgemeinschaft (DFG; grant TS60/4-1). We thank F.-M. Gutzki for performing GC–MS/MS analyses.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
The studies involving human participants were approved by the Ethics Committee of the Hannover Medical School.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sitar, M.E., Kayacelebi, A.A., Beckmann, B. et al. Asymmetric dimethylarginine (ADMA) in human blood: effects of extended haemodialysis in the critically ill patient with acute kidney injury, protein binding to human serum albumin and proteolysis by thermolysin. Amino Acids 47, 1983–1993 (2015). https://doi.org/10.1007/s00726-015-1991-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-015-1991-4