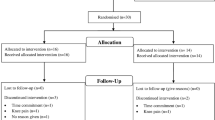
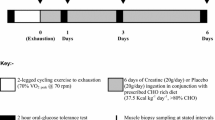
Abstract
Greater force produced with eccentric (ECC) compared to concentric (CONC) contractions, may comprise a stronger driver of muscle growth, which may be further augmented by protein supplementation. We investigated the effect of differentiated contraction mode with either whey protein hydrolysate and carbohydrate (WPH + CHO) or isocaloric carbohydrate (CHO) supplementation on regulation of anabolic signalling, muscle protein synthesis (MPS) and muscle hypertrophy. Twenty-four human participants performed unilateral isolated maximal ECC versus CONC contractions during exercise habituation, single-bout exercise and 12 weeks of training combined with WPH + CHO or CHO supplements. In the exercise-habituated state, p-mTOR, p-p70S6K, p-rpS6 increased by approximately 42, 206 and 213 %, respectively, at 1 h post-exercise, with resistance exercise per se; whereas, the phosphorylation was exclusively maintained with ECC at 3 and 5 h post-exercise. This acute anabolic signalling response did not differ between the isocaloric supplement types, neither did protein fractional synthesis rate differ between interventions. Twelve weeks of ECC as well as CONC resistance training augmented hypertrophy with WPH + CHO group compared to the CHO group (7.3 ± 1.0 versus 3.4 ± 0.8 %), independently of exercise contraction type. Training did not produce major changes in basal levels of Akt-mTOR pathway components. In conclusion, maximal ECC contraction mode may constitute a superior driver of acute anabolic signalling that may not be mirrored in the muscle protein synthesis rate. Furthermore, with prolonged high-volume resistance training, contraction mode seems less influential on the magnitude of muscle hypertrophy, whereas protein and carbohydrate supplementation augments muscle hypertrophy as compared to isocaloric carbohydrate supplementation .








Similar content being viewed by others
References
Aagaard P, Simonsen EB, Andersen JL, Magnusson SP, Halkjaer-Kristensen J, Dyhre-Poulsen P (2000) Neural inhibition during maximal eccentric and concentric quadriceps contraction: effects of resistance training. J Applied Physiol 89:2249–2257
Abbott BC, Bigland B, Ritchie JM (1952) The physiological cost of negative work. J Physiol 117:380–390
Andersen LL et al (2005) The effect of resistance training combined with timed ingestion of protein on muscle fiber size and muscle strength, Metab. Clin Exp 54:151–156. doi:10.1016/j.metabol.2004.07.012
Anthony JC, Anthony TG, Kimball SR, Vary TC, Jefferson LS (2000a) Orally administered leucine stimulates protein synthesis in skeletal muscle of post-absorptive rats in association with increased eIF4F formation. J Nutrition 130:139–145
Anthony JC, Yoshizawa F, Anthony TG, Vary TC, Jefferson LS, Kimball SR (2000b) Leucine stimulates translation initiation in skeletal muscle of post-absorptive rats via a rapamycin-sensitive pathway. J Nutr 130:2413–2419
Atherton PJ, Babraj J, Smith K, Singh J, Rennie MJ, Wackerhage H (2005) Selective activation of AMPK-PGC-1alpha or PKB-TSC2-mTOR signaling can explain specific adaptive responses to endurance or resistance training-like electrical muscle stimulation. FASEB J 19:786–788. doi:10.1096/fj.04-2179fje
Burd NA et al (2010a) Resistance exercise volume affects myofibrillar protein synthesis and anabolic signalling molecule phosphorylation in young men. J Physiol 588:3119–3130. doi:10.1113/jphysiol.2010.192856;10.1113/jphysiol.2010.192856
Burd NA et al (2010b) Low-load high volume resistance exercise stimulates muscle protein synthesis more than high-load low volume resistance exercise in young men. PloS One 5:e12033. doi:10.1371/journal.pone.0012033;10.1371/journal.pone.0012033
Burd NA et al (2012) Muscle time under tension during resistance exercise stimulates differential muscle protein sub-fractional synthetic responses in men. J Physiol 590:351–362. doi:10.1113/jphysiol.2011.221200;10.1113/jphysiol.2011.221200
Churchward-Venne TA et al (2012) Supplementation of a suboptimal protein dose with leucine or essential amino acids: effects on myofibrillar protein synthesis at rest and following resistance exercise in men. J Physiol 590:2751–2765. doi:10.1113/jphysiol.2012.228833;10.1113/jphysiol.2012.228833
Churchward-Venne TA et al (2014) Leucine supplementation of a low-protein mixed macronutrient beverage enhances myofibrillar protein synthesis in young men: a double-blind, randomized trial. Am J Clin Nutr 99:276–286. doi:10.3945/ajcn.113.068775
Crozier SJ, Kimball SR, Emmert SW, Anthony JC, Jefferson LS (2005) Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J Nutr 135:376–382
Cuthbertson DJ, Babraj J, Smith K, Wilkes E, Fedele MJ, Esser K, Rennie M (2006) Anabolic signaling and protein synthesis in human skeletal muscle after dynamic shortening or lengthening exercise American journal of physiologyEndocrinology and metabolism 290:E731–E738. doi:10.1152/ajpendo.00415.2005
Dean D et al (2000) Exercise diminishes the activity of acetyl-CoA carboxylase in human muscle. Diabetes 49:1295–1300
Dibble CC, Manning BD (2013) Signal integration by mTORC1 coordinates nutrient input with biosynthetic output. Nat Cell Biol 15:555–564. doi:10.1038/ncb276310.1038/ncb2763
Dickinson JM et al (2011) Mammalian target of rapamycin complex 1 activation is required for the stimulation of human skeletal muscle protein synthesis by essential amino acids. J Nutr 141:856–862. doi:10.3945/jn.111.139485;10.3945/jn.111.139485
Eliasson J, Elfegoun T, Nilsson J, Kohnke R, Ekblom B, Blomstrand E (2006) Maximal lengthening contractions increase p70 S6 kinase phosphorylation in human skeletal muscle in the absence of nutritional supply. Am J Physiologyendocrinol Metab 291:E1197–1205. doi:10.1152/ajpendo.00141.2006
Farup J et al (2013) Whey protein hydrolysate augments tendon and muscle hypertrophy independent of resistance exercise contraction mode. Scand J Med Sci Sports. doi:10.1111/sms.12083;10.1111/sms.12083
Friden J, Lieber RL (2001) Eccentric exercise-induced injuries to contractile and cytoskeletal muscle fibre components. Acta Physiol Scand 171:321–326
Fujita S et al (2007) Nutrient signalling in the regulation of human muscle protein synthesis. J Physiol 582:813–823. doi:10.1113/jphysiol.2007.134593
Gannon MC, Nuttall FQ (2010) Amino acid ingestion and glucose metabolism–a review. IUBMB Life 62:660–668. doi:10.1002/iub.375;10.1002/iub.375
Gehlert S et al (2012) Intense resistance exercise induces early and transient increases in ryanodine receptor 1 phosphorylation in human skeletal muscle. PLoS One 7:e49326. doi:10.1371/journal.pone.004932610.1371/journal.pone.0049326
Gurtler A et al (2013) Stain-Free technology as a normalization tool in Western blot analysis. Anal Biochem 433:105–111. doi:10.1016/j.ab.2012.10.010;10.1016/j.ab.2012.10.010
Gwinn DM et al (2008) AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell 30:214–226. doi:10.1016/j.molcel.2008.03.003;10.1016/j.molcel.2008.03.003
Hartman JW, Tang JE, Wilkinson SB, Tarnopolsky MA, Lawrence RL, Fullerton AV, Phillips SM (2007) Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am J Clin Nutr 86:373–381
Holm L, van Hall G, Rose AJ, Miller BF, Doessing S, Richter EA, Kjaer M (2010) Contraction intensity and feeding affect collagen and myofibrillar protein synthesis rates differently in human skeletal muscle. Am J Physiologyendocrinol Metab 298:E257–E269. doi:10.1152/ajpendo.00609.2009;10.1152/ajpendo.00609.2009
Hornberger TA (2011) Mechanotransduction and the regulation of mTORC1 signaling in skeletal muscle. Int J Biochem Cell Biol 43:1267–1276. doi:10.1016/j.biocel.2011.05.007;10.1016/j.biocel.2011.05.007
Hornberger TA, Chien S (2006) Mechanical stimuli and nutrients regulate rapamycin-sensitive signaling through distinct mechanisms in skeletal muscle. J Cell Biochem 97:1207–1216. doi:10.1002/jcb.20671
Hulmi JJ, Tannerstedt J, Selanne H, Kainulainen H, Kovanen V, Mero AA (2009) Resistance exercise with whey protein ingestion affects mTOR signaling pathway and myostatin in men. J Appl Physiol 106:1720–1729. doi:10.1152/japplphysiol.00087.2009 (Bethesda, Md: 1985)
Hulmi JJ, Lockwood CM, Stout JR (2010) Effect of protein/essential amino acids and resistance training on skeletal muscle hypertrophy: a case for whey protein. Nutrition Metab 7:51. doi:10.1186/1743-7075-7-51
Jewell JL, Guan KL (2013) Nutrient signaling to mTOR and cell growth. Trends Biochem Sci 38:233–242. doi:10.1016/j.tibs.2013.01.004;10.1016/j.tibs.2013.01.004
Kakigi R, Yoshihara T, Ozaki H, Ogura Y, Ichinoseki-Sekine N, Kobayashi H, Naito H (2014) Whey protein intake after resistance exercise activates mTOR signaling in a dose-dependent manner in human skeletal muscle. Eur J Appl Physiol. doi:10.1007/s00421-013-2812-7
Latres E et al (2005) Insulin-like growth factor-1 (IGF-1) inversely regulates atrophy-induced genes via the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin (PI3K/Akt/mTOR) pathway. J Biol Chem 280:2737–2744. doi:10.1074/jbc.M407517200
Li R et al (2013) Costamere remodeling with muscle loading and unloading in healthy young men. J Anat 223:525–536. doi:10.1111/joa.12101;10.1111/joa.12101
Lueders TN et al (2011) The alpha7beta1-integrin accelerates fiber hypertrophy and myogenesis following a single bout of eccentric exercise American journal of physiologyCell. Physiology 301:C938–C946. doi:10.1152/ajpcell.00515.2010;10.1152/ajpcell.00515.2010
Menon S et al (2014) Spatial Control of the TSC Complex Integrates Insulin and Nutrient Regulation of mTORC1 at the Lysosome. Cell 156:771–785
Mitchell CJ, Churchward-Venne TA, West DW, Burd NA, Breen L, Baker SK, Phillips SM (2012) Resistance exercise load does not determine training-mediated hypertrophic gains in young men. J Appl Physiol 113:71–77. doi:10.1152/japplphysiol.00307.2012 (Bethesda, Md: 1985)
Mitchell CJ et al (2014) Acute post-exercise myofibrillar protein synthesis is not correlated with resistance training-induced muscle hypertrophy in young men. PLoS One 9:e89431. doi:10.1371/journal.pone.0089431
Moberg M, Apro W, Ohlsson I, Ponten M, Villanueva A, Ekblom B, Blomstrand E (2014) Absence of leucine in an essential amino acid supplement reduces activation of mTORC1 signalling following resistance exercise in young females. Appl Physiol Nutr Metab 39:183–194. doi:10.1139/apnm-2013-0244
Moller AB, Vendelbo MH, Rahbek SK, Clasen BF, Schjerling P, Vissing K, Jessen N (2013) Resistance exercise, but not endurance exercise, induces IKKbeta phosphorylation in human skeletal muscle of training-accustomed individuals. Pflugers Arch 465:1785–1795. doi:10.1007/s00424-013-1318-9
Moore DR, Phillips SM, Babraj JA, Smith K, Rennie MJ (2005) Myofibrillar and collagen protein synthesis in human skeletal muscle in young men after maximal shortening and lengthening contractions. Am J Physiologyendocrinol Metab 288:E1153–E1159. doi:10.1152/ajpendo.00387.2004
Moore DR, Young M, Phillips SM (2012) Similar increases in muscle size and strength in young men after training with maximal shortening or lengthening contractions when matched for total work. Eur J Appl Physiol 112:1587–1592. doi:10.1007/s00421-011-2078-x;10.1007/s00421-011-2078-x
Nave BT, Ouwens M, Withers DJ, Alessi DR, Shepherd PR (1999) Mammalian target of rapamycin is a direct target for protein kinase B: identification of a convergence point for opposing effects of insulin and amino-acid deficiency on protein translation. Biochem J 344(Pt 2):427–431
O’Neil TK, Duffy LR, Frey JW, Hornberger TA (2009) The role of phosphoinositide 3-kinase and phosphatidic acid in the regulation of mammalian target of rapamycin following eccentric contractions. J Physiol 587:3691–3701. doi:10.1113/jphysiol.2009.173609
Park SH, Gammon SR, Knippers JD, Paulsen SR, Rubink DS, Winder WW (2002) Phosphorylation-activity relationships of AMPK and acetyl-CoA carboxylase in muscle. J Appl Physiol 92:2475–2482. doi:10.1152/japplphysiol.00071.2002 (Bethesda, Md: 1985)
Pennings B, Boirie Y, Senden JM, Gijsen AP, Kuipers H, van Loon LJ (2011) Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am J Clin Nutr 93:997–1005. doi:10.3945/ajcn.110.008102
Phillips SM, Tipton KD, Aarsland A, Wolf SE, Wolfe RR (1997) Mixed muscle protein synthesis and breakdown after resistance exercise in humans. Am J Physiol 273:E99–E107
Proud CG (2014) Control of the translational machinery by amino acids. Am J Clin Nutr 99:231S–236S. doi:10.3945/ajcn.113.066753;10.3945/ajcn.113.066753
Rasmussen BB, Phillips SM (2003) Contractile and nutritional regulation of human muscle growth. Exerc Sport Sci Rev 31:127–131
Roig M, O’Brien K, Kirk G, Murray R, McKinnon P, Shadgan B, Reid WD (2009) The effects of eccentric versus concentric resistance training on muscle strength and mass in healthy adults: a systematic review with meta-analysis. Br J Sports Med 43:556–568. doi:10.1136/bjsm.2008.051417
Spangenburg EE, Le Roith D, Ward CW, Bodine SC (2008) A functional insulin-like growth factor receptor is not necessary for load-induced skeletal muscle hypertrophy. J Physiol 586:283–291. doi:10.1113/jphysiol.2007.141507
Stefanetti RJ et al (2014) Influence of divergent exercise contraction mode and whey protein supplementation on atrogin-1, MuRF1 and FOXO1/3A in human skeletal muscle. J Appl Physiol 116(11):1491–1502. doi:10.1152/japplphysiol.00136.2013
van Loon LJ, Saris WH, Verhagen H, Wagenmakers AJ (2000) Plasma insulin responses after ingestion of different amino acid or protein mixtures with carbohydrate. Am J Clin Nutr 72:96–105
Vissing K, Andersen JL, Schjerling P (2005) Are exercise-induced genes induced by exercise? FASEB J 19:94–96. doi:10.1096/fj.04-2084fje
Vissing K et al (2013) Effect of resistance exercise contraction mode and protein supplementation on members of the STARS signalling pathway. J Physiol 591:3749–3763. doi:10.1113/jphysiol.2012.249755;10.1113/jphysiol.2012.249755
Wernbom M, Augustsson J, Thomee R (2007) The influence of frequency, intensity, volume and mode of strength training on whole muscle cross-sectional area in humans. Sports medicine 37:225–264
Wilkinson SB, Phillips SM, Atherton PJ, Patel R, Yarasheski KE, Tarnopolsky MA, Rennie MJ (2008) Differential effects of resistance and endurance exercise in the fed state on signalling molecule phosphorylation and protein synthesis in human muscle. J Physiol 586:3701–3717. doi:10.1113/jphysiol.2008.153916;10.1113/jphysiol.2008.153916
Acknowledgments
We thank the participants for their participation in the project. Gitte Hartvigsen, and Janni Mosgaard Jensen (Section of Sport Science, Department of Public Health, Aarhus University, Denmark), Helle Zibrandtsen (at Reasearch Laboratory for Biochemical Pathology, Institute for Clinical Medicine, Aarhus University, Denmark) and Ann-Marie Sedstrøm (at Institute of Sports Medicine, Bispebjerg Hospital, Copenhagen, Denmark) are thanked for technical assistance. We thank Arla Foods Ingredients Group P/S DK for funding the project.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
All participants were informed about the purpose and the risks related to the study and gave written, informed consent to participate. The study was approved by The Central Denmark Region Committees on Health Research Ethics (j. no. M-20110003) and performed in accordance with the Declaration of Helsinki.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rahbek, S.K., Farup, J., Møller, A.B. et al. Effects of divergent resistance exercise contraction mode and dietary supplementation type on anabolic signalling, muscle protein synthesis and muscle hypertrophy. Amino Acids 46, 2377–2392 (2014). https://doi.org/10.1007/s00726-014-1792-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-014-1792-1