Abstract
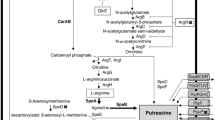
Properties of putrescine uptake by PotFGHI and PuuP and their physiological significance were studied using a polyamine biosynthesis and uptake deficient Escherichia coli KK3131 transformed with pACYC184 containing potFGHI or puuP. Putrescine uptake activity of E. coli KK3131 transformed with pACYC184-PotFGHI was higher than that of E. coli 3131 transformed with pACYC-PuuP when cells were cultured in the absence of putrescine. Putrescine uptake by PotFGHI was both ATP and membrane potential dependent, while that by PuuP was membrane potential dependent. Feedback inhibition by polyamines occurred at the PotFGHI uptake system but not at the PuuP uptake system. Expression of PuuP was reduced in the presence of PuuR, a negative regulator for PuuP, and expression of PuuR was positively regulated by glucose, which reduces the level of cAMP. The complex of cAMP and CRP (cAMP receptor protein) inhibited the expression of PuuR in the absence of glucose. Thus, the growth rate of E. coli KK3131 in the presence of both 0.4 % (22.2 mM) glucose and 10 mM putrescine was in the order of cells transformed with pACYC-PotFGHI > pACYC-PuuP > pACYC-PuuP + PuuR, which was parallel with the polyamine content in cells. The results indicate that PotFGHI is necessary for rapid cell growth in the presence of glucose as an energy source. When glucose in medium was depleted, however, PuuP was absolutely necessary for cell growth in the presence of putrescine, because accumulation of putrescine to a high level by PuuP was necessary for utilization of putrescine as an energy source.





Similar content being viewed by others
Abbreviations
- CadB:
-
Cadaverine-lysine antiporter
- CCCP:
-
Carbonyl cyanide m-chlorophenylhydrazone
- CRP:
-
cAMP receptor protein
- PotE:
-
Putrescine-ornithine antiporter
References
Bakker EP, Mangerich WE (1981) Interconversion of components of the bacterial proton motive force by electrogenic potassium transport. J Bacteriol 147:820–826
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Casero RA, Pegg AE (2009) Polyamine catabolism and disease. Biochem J 421:323–338
Cohen SS (1998) A guide to polyamines. Oxford University Press, New York
Cunningham-Rundles S, Maas WK (1975) Isolation, characterization, and mapping of Escherichia coli mutants blocked in the synthesis of ornithine decarboxylase. J Bacteriol 124:791–799
Emory SA, Belasco JG (1990) The ompA 5′ untranslated RNA segment functions in Escherichia coli as a growth-rate-regulated mRNA stabilizer whose activity is unrelated to translational efficiency. J Bacteriol 172:4472–4481
Higashi K, Ishigure H, Demizu R, Uemura T, Nishino K, Yamaguchi A, Kashiwagi K, Igarashi K (2008) Identification of a spermidine excretion protein complex (MdtJI) in Escherichia coli. J Bacteriol 190:872–878
Higashi K, Sakamaki Y, Herai E, Demizu R, Uemura T, Saroj SD, Zenda R, Terui Y, Nishimura K, Toida T, Kashiwagi K, Igarashi K (2010) Identification and functions of amino acid residues in PotB and PotC involved in spermidine uptake activity. J Biol Chem 285:39061–39069
Igarashi K, Kashiwagi K (2010a) Characteristics of cellular polyamine transport in prokaryotes and eukaryotes. Plant Physiol Biochem 48:506–512
Igarashi K, Kashiwagi K (2010b) Modulation of cellular function by polyamines. Int J Biochem Cell Biol 42:39–51
Igarashi K, Kashiwagi K, Hamasaki H, Miura A, Kakegawa T, Hirose S, Matsuzaki S (1986) Formation of a compensatory polyamine by Escherichia coli polyamine-requiring mutants during growth in the absence of polyamines. J Bacteriol 166:128–134
Kashiwagi K, Hosokawa N, Furuchi T, Kobayashi H, Sasakawa C, Yoshikawa M, Igarashi K (1990) Isolation of polyamine transport-deficient mutants of Escherichia coli and cloning of the genes for polyamine transport proteins. J Biol Chem 265:20893–20897
Kashiwagi K, Suzuki T, Suzuki F, Furuchi T, Kobayashi H, Igarashi K (1991) Coexistence of the genes for putrescine transport protein and ornithine decarboxylase at 16 min on Escherichia coli chromosome. J Biol Chem 266:20922–20927
Kashiwagi K, Innami A, Zenda R, Tomitori H, Igarashi K (2002) The ATPase activity and the functional domain of PotA, a component of the sermidine-preferential uptake system in Escherichia coli. J Biol Chem 277:24212–24219
Kurihara S, Oda S, Tsuboi Y, Kim HG, Oshida M, Kumagai H, Suzuki H (2008) γ-Glutamylputrescine synthetase in the putrescine utilization pathway of Escherichia coli K-12. J Biol Chem 283:19981–19990
Kurihara S, Tsuboi Y, Oda S, Kim HG, Kumagai H, Suzuki H (2009) The putrescine Importer PuuP of Escherichia coli K-12. J Bacteriol 191:2776–2782
Lin HH, Hsu CC, Yang CD, Ju YW, Chen YP, Tseng CP (2011) Negative effect of glucose on ompA mRNA stability: a potential role of cyclic AMP in the repression of hfq in Escherichia coli. J Bacteriol 193:5833–5840
Marton LJ, Pegg AE (1995) Polyamines as targets for therapeutic intervention. Annu Rev Pharmacol Toxicol 35:55–91
Meng SY, Bennett GN (1992) Nucleotide sequence of the Escherichia coli cad operon: a system for neutralization of low extracellular pH. J Bacteriol 174:2659–2669
Neely MN, Olson ER (1996) Kinetics of expression of the Escherichia coli cad operon as a function of pH and lysine. J Bacteriol 178:5522–5528
Nemoto N, Kurihara S, Kitahara Y, Asada K, Kato K, Suzuki H (2012) Mechanism for regulation of the putrescine utilization pathway by the transcription factor PuuR in Escherichia coli K-12. J Bacteriol 194:3437–3447
Oliver DB, Beckwith J (1982) Regulation of a membrane component required for protein secretion in Escherichia coli. Cell 30:311–319
Pistocchi R, Kashiwagi K, Miyamoto S, Nukui E, Sadakata Y, Kobayashi H, Igarashi K (1993) Characteristics of the operon for a putrescine transport system that maps at 19 minutes on the Escherichia coli chromosome. J Biol Chem 268:146–152
Sambrook J, Russell DW (2001) Transformation of E. coli by electroporation. In: Sambrook J, Russell DW (eds) Molecular Cloning: A Laboratory Manual 3rd Ed. Cold Spring Harbor Laboratory, NY, pp 1.119–111.122
Shimada T, Fujita N, Yamamoto K, Ishihama A (2011) Novel roles of cAMP receptor protein (CRP) in regulation of transport and metabolism of carbon sources. PLoS One 6:e20081
Soksawatmaekhin W, Kuraishi A, Sakata K, Kashiwagi K, Igarashi K (2004) Excretion and uptake of cadaverine by CadB and its physiological functions in Escherichia coli. Mol Microbiol 51:1401–1412
Takayama M, Ohyama T, Igarashi K, Kobayashi H (1994) Escherichia coli cad operon functions as a supplier of carbon dioxide. Mol Microbiol 11:913–918
Terui Y, Higashi K, Taniguchi S, Shigemasa A, Nishimura K, Yamamoto K, Kashiwagi K, Ishihama A, Igarashi K (2007) Enhancement of the synthesis of RpoN, Cra, and H-NS by polyamines at the level of translation in Escherichia coli cultured with glucose and glutamate. J Bacteriol 189:2359–2368
Tomitori H, Kashiwagi K, Igarashi K (2012) Structure and function of polyamine-amino acid antiporters CadB and PotE in Escherichia coli. Amino Acids 42:733–740
Watson N, Dunyak DS, Rosey EL, Slonczewski JL, Olson ER (1992) Identification of elements involved in transcriptional regulation of the Escherichia coli cad operon by external pH. J Bacteriol 174:530–540
Yoshida M, Kashiwagi K, Kawai G, Ishihama A, Igarashi K (2001) Polyamine enhancement of the synthesis of adenylate cyclase at the translational level and the consequential stimulation of the synthesis of the RNA polymerase σ28 subunit. J Biol Chem 276:16289–16295
Acknowledgments
We thank Drs. A. J. Michael and K. Williams for their help in preparing the manuscript. We also thank Dr. A. Ishihama for providing antibodies against CRP and RNA polymerase σ70 subunit. This work was supported by a Gran-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, Culture, and Technology, Japan. S. D. Saroj was supported by the Tokyo Biochemical Research Foundation Research Fellowship.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Terui and S. D. Saroj contributed equally to this work.
Rights and permissions
About this article
Cite this article
Terui, Y., Saroj, S.D., Sakamoto, A. et al. Properties of putrescine uptake by PotFGHI and PuuP and their physiological significance in Escherichia coli . Amino Acids 46, 661–670 (2014). https://doi.org/10.1007/s00726-013-1517-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-013-1517-x