Abstract
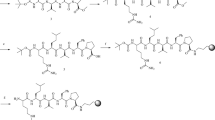
Two new chiral monochloro-s-triazines (MCT) were synthesized [viz N-(4-chloro-6-piperidinyl-[1,3,5]-triazine-2-yl)-l-leucine amide and N-(4-chloro-6-piperidinyl-[1,3,5]-triazine-2-yl)-l-leucine) (CDR 1 and 2, respectively)] by the nucleophilic displacement of chlorine atoms in s-triazine moiety. One of the Cl atoms was replaced with piperidine, and the second Cl atom in the 6-piperidinyl derivative was replaced with amino acid amide (viz l-Leu–NH2) and amino acid (l-Leu). These reagents were characterized and used as CDRs for chiral separation of protein and non-protein amino acids, and were separated on a reversed-phase C18 column. The reaction conditions were optimized for the synthesis of diastereomers using one MCT reagent. The separation method was validated for limit of detection, linearity, accuracy, precision, and recovery.



Similar content being viewed by others
References
Ali HMS, Pätzold R, Brückner H (2010) Gas chromatographic determination of amino acid enantiomers in bottled and aged wines. Amino Acids 38:951–958
B’Hymer C, Montes-Bayon M, Caruso JA (2003) Marfey’s reagent: past, present, and future uses of 1-fluoro-2, 4-dinitrophenyl-5-l-alanine amide. J Sep Sci 26:7–19
Bhushan R, Agarwal C (2010) Application of (S)-N-(4-nitrophenoxycarbonyl) phenylalanine methoxyethyl ester as a chiral derivatizing reagent for reversed-phase high-performance liquid chromatographic separation of diastereomers of amino alcohols, non-protein amino acids and PenA. Amino Acids. doi:10.1007/s00726-010-0472-z
Bhushan R, Brückner H (2004) Marfey’s reagent for chiral amino acid analysis: a review. Amino Acids 27:231–247
Bhushan R, Kumar V (2008) Indirect enantioseparation of α-amino acids by reversed-phase liquid chromatography using new chiral derivatizing reagents synthesized from s-triazine chloride. J Chromatogr A 1201:35–42
Bhushan R, Kumar V, Tanwar S (2009) Chromatographic separation of enantiomers of non-protein α-amino acids after derivatization with Marfey’s reagent and its four variants. Amino Acids 36:571–579
Brückner H, Keller-Hoehl C (1990) HPLC separation of dl-amino acids derivatized with N2-(5-fluoro-2, 4-dinitrophenyl)-l-amino acid amides. Chromatographia 30:621–629
Brückner H, Wachsmann M (1996) Liquid chromatographic separation of amino acid enantiomers on a silica-bonded chiral s-triazine column. J Chromatogr 728:447–454
Brückner H, Wachsmann M (2003) Design of chiral monochloro-s-triazine reagents for the liquid chromatographic separation of amino acid enantiomers. J Chromatogr 998:73–82
De Zotti M, Damato F, Formaggio F, Cirsma M, Schievano E, Mammi S, Kaptein B, Broxterman QB, Felock PJ, Hazuda DJ, Singh SB, Kirschbaum J, Brückner H, Toniolo C (2010) Total synthesis, characterization, and conformational analysis of the naturally occurring hexadecapeptide integramide A and a diastereomer. Chem Eur J 16:316–327
Fujii K, Ikai Y, Mayumi T, Oka H, Suzuki M, Harada K (1997) A nonempirical method using LC/MS for determination of the absolute configuration of constituent amino acids in a peptide: elucidation of limitations of Marfey’s method and of its separation mechanism. Anal Chem 69:3346–3352
Hunt S (1985) In: Barrett GC (ed) Chemistry and Biochemistry of the amino acids. Chapman and Hall, London, p 55
Lin J-Y, Yang M-H (1993) High-performance liquid chromatographic separation of enantiomeric amino acids on bis[carbamoyl(alkyl)methylamino]-6-chloro-s-triazine-derived chiral stationary phases. J Chromatogr 644:277–283
Marfey P (1984) Determination of d-amino acids. II. Use of a bifunctional reagent, 1,5-difluoro-2,4-dinitrobenzene. Carlsberg Res Comm 49:591–596
Wachsmann M, Brückner H (1998) Ligand-exchange chromatographic separation of dl-amino acids on aminopropylsilica-bonded chiral s-triazines. Chromatographia 47:637–642
Acknowledgments
The authors are thankful to the Ministry of Human Resources Development, Government of India, New Delhi for the award of a research assistantship (to C.A.) and to the Alexander von Humboldt Foundation, Bonn, Germany for donating Knauer HPLC equipment and to the Council of Scientific and Industrial Research, New Delhi, India for financial assistance [to R.B.; research grant No. 01(2334)/09/EMR-II].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhushan, R., Agarwal, C. Reversed-phase liquid chromatographic resolution of diastereomers of protein and non-protein amino acids prepared with newly synthesized chiral derivatizing reagents based on cyanuric chloride. Amino Acids 40, 403–409 (2011). https://doi.org/10.1007/s00726-010-0650-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-010-0650-z