Abstract
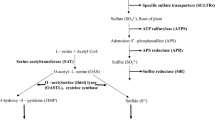
Mobilization of the l-cysteine sulfur for the persulfuration of the rhodanese of Azotobacter vinelandii, RhdA, can be mediated by the A. vinelandii cysteine desulfurases, IscS and NifS. The amount of cysteine was higher in mutant strains lacking rhdA (MV474) than in wild type. The diazotrophic growth of MV474 was impaired. Taking into account the functional results about rhodanese-like proteins and RhdA itself, it is suggested that RhdA-dependent modulation of l-cysteine levels must deal with a redox-related process.




Similar content being viewed by others
References
Behshad E, Parkin SE, Bollinger JM Jr (2004) Mechanism of cysteine desulfurase Slr0387 from Synechocystis sp. PCC 6803: kinetic analysis of cleavage of the persulfide intermediate by chemical reductants. Biochemistry 43:12220–12226
Beinert H (2000) A tribute to sulfur. Eur J Biochem 267:5657–5664
Bordo D, Deriu D, Colnaghi R, Carpen A, Pagani S, Bolognesi M (2000) The crystal structure of a sulfurtransferase from Azotobacter vinelandii highlights the evolutionary relationship between the rhodanese and phosphatase enzyme families. J Mol Biol 298:691–704
Bordo D, Forlani F, Spallarossa A, Colnaghi R, Carpen A, Bolognesi M, Pagani S (2001) A persulfurated cysteine promotes active site reactivity in Azotobacter vinelandii rhodanese. Biol Chem 382:1245–1252
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cereda A, Forlani F, Iametti S, Bernhardt R, Ferranti P, Picariello G, Pagani S, Bonomi F (2003) Molecular recognition between Azotobacter vinelandii rhodanese and a sulfur acceptor protein. Biol Chem 384:1473–1481
Cereda A, Carpen A, Picariello G, Iriti M, Faoro F, Ferranti P, Pagani S (2007) Effects of the deficiency of the rhodanese-like protein RhdA in Azotobacter vinelandii. FEBS Lett 581:1625–1630
Cereda A, Carpen A, Picariello G, Tedeschi G, Pagani S (2009) The lack of rhodanese RhdA affects the sensitivity of Azotobacter vinelandii to oxidative events. Biochem J 418:135–143
Colnaghi R, Pagani S, Kennedy C, Drummond M (1996) Cloning, sequence analysis and overexpression of the rhodanese gene of Azotobacter vinelandii. Eur J Biochem 236:240–248
Dos Santos PC, Johnson DC, Ragle BE, Unciuleac MC, Dean DR (2007) Controlled expression of nif and isc iron-sulfur protein maturation components reveals target specificity and limited functional replacement between the two systems. J Bacteriol 189:2854–2862
Flint DH (1996) Escherichia coli contains a protein that is homologous in function and N-terminal sequence to the protein encoded by the nifS gene of Azotobacter vinelandii and that can participate in the synthesis of the Fe–S cluster of dihydroxy-acid dehydratase. J Biol Chem 271:16068–16074
Florczyk MA, McCue LA, Stack RF, Hauer CR, McDonough KA (2001) Identification and characterization of mycobacterial proteins differentially expressed under standing and shaking culture conditions, including Rv2623 from a novel class of putative ATP-binding proteins. Infect Immun 69:5777–5785
Forlani F, Carpen A, Pagani S (2003) Evidence that elongation of the catalytic loop of the Azotobacter vinelandii rhodanese changed selectivity from sulfur- to phosphate-containing substrates. Protein Eng 16:515–519
Forlani F, Cereda A, Freuer A, Nimtz M, Leimkuhler S, Pagani S (2005) The cysteine-desulfurase IscS promotes the production of the rhodanese RhdA in the persulfurated form. FEBS Lett 579:6786–6790
Giuliani MC, Tron P, Leroy G, Aubert C, Tauc P, Giudici-Orticoni MT (2007) A new sulfurtransferase from the hyperthermophilic bacterium Aquifex aeolicus. Being single is not so simple when temperature gets high. FEBS J 274:4572–4587
Ikeuchi Y, Shigi N, Kato J, Nishimura A, Suzuki T (2006) Mechanistic insights into sulfur relay by multiple sulfur mediators involved in thiouridine biosynthesis at tRNA wobble positions. Mol Cell 21:97–108
Jacobson MR, Cash VL, Weiss MC, Laird NF, Newton WE, Dean DR (1989) Biochemical and genetic analysis of the nifUSVWZM cluster from Azotobacter vinelandii. Mol Gen Genet 219:49–57
Johnson DC, Dean DR, Smith AD, Johnson MK (2005) Structure, function, and formation of biological iron-sulfur clusters. Annu Rev Biochem 74:247–281
Johnson DC, Unciuleac MC, Dean DR (2006) Controlled expression and functional analysis of iron-sulfur cluster biosynthetic components within Azotobacter vinelandii. J Bacteriol 188:7551–7561
Kambampati R, Lauhon CT (1999) IscS is a sulfurtransferase for the in vitro biosynthesis of 4-thiouridine in Escherichia coli tRNA. Biochemistry 38:16561–16568
Kambampati R, Lauhon CT (2000) Evidence for the transfer of sulfane sulfur from IscS to ThiI during the in vitro biosynthesis of 4-thiouridine in Escherichia coli tRNA. J Biol Chem 275:10727–10730
Kato S, Mihara H, Kurihara T, Takahashi Y, Tokumoto U, Yoshimura T, Esaki N (2002) Cys-328 of IscS and Cys-63 of IscU are the sites of disulfide bridge formation in a covalently bound IscS/IscU complex: implications for the mechanism of iron–sulfur cluster assembly. Proc Natl Acad Sci USA 99:5948–5952
Kessler D (2006) Enzymatic activation of sulfur for incorporation into biomolecules in prokaryotes. FEMS Microbiol Rev 30:825–840
Kosower NS, Kosower EM (1987) Thiol labeling with bromobimanes. Methods Enzymol 143:76–84
Krivobok S, Kuony S, Meyer C, Louwagie M, Willison JC, Jouanneau Y (2003) Identification of pyrene-induced proteins in Mycobacterium sp. strain 6PY1: evidence for two ring-hydroxylating dioxygenases. J Bacteriol 185:3828–3841
Lauhon CT, Skovran E, Urbina HD, Downs DM, Vickery LE (2004) Substitutions in an active site loop of Escherichia coli IscS result in specific defects in Fe–S cluster and thionucleoside biosynthesis in vivo. J Biol Chem 279:19551–19558
Matthies A, Rajagopalan KV, Mendel RR, Leimkuhler S (2004) Evidence for the physiological role of a rhodanese-like protein for the biosynthesis of the molybdenum cofactor in humans. Proc Natl Acad Sci USA 101:5946–5951
Matthies A, Nimtz M, Leimkuhler S (2005) Molybdenum cofactor biosynthesis in humans: identification of a persulfide group in the rhodanese-like domain of MOCS3 by mass spectrometry. Biochemistry 44:7912–7920
Mihara H, Esaki N (2002) Bacterial cysteine desulfurases: their function and mechanisms. Appl Microbiol Biotechnol 60:12–23
Mueller EG (2006) Trafficking in persulfides: delivering sulfur in biosynthetic pathways. Nat Chem Biol 2:185–194
Nagahara N (2008) A novel mercaptopyruvate sulfurtransferase thioredoxin-dependent redox-sensing molecular switch: a mechanism for the maintenance of cellular redox equilibrium. Mini Rev Med Chem 8:585–589
Nandi DL, Westley J (1998) Reduced thioredoxin as a sulfur-acceptor substrate for rhodanese. Int J Biochem Cell Biol 30:973–977
Nandi DL, Horowitz PM, Westley J (2000) Rhodanese as a thioredoxin oxidase. Int J Biochem Cell Biol 32:465–473
Pagani S, Forlani F, Carpen A, Bordo D, Colnaghi R (2000) Mutagenic analysis of Thr-232 in rhodanese from Azotobacter vinelandii highlighted the differences of this prokaryotic enzyme from the known sulfurtransferases. FEBS Lett 472:307–311
Palenchar PM, Buck CJ, Cheng H, Larson TJ, Mueller EG (2000) Evidence that ThiI, an enzyme shared between thiamin and 4-thiouridine biosynthesis, may be a sulfurtransferase that proceeds through a persulfide intermediate. J Biol Chem 275:8283–8286
Park S, Imlay JA (2003) High levels of intracellular cysteine promote oxidative DNA damage by driving the fenton reaction. J Bacteriol 185:1942–1950
Ray WK, Zeng G, Potters MB, Mansuri AM, Larson TJ (2000) Characterization of a 12-kilodalton rhodanese encoded by glpE of Escherichia coli and its interaction with thioredoxin. J Bacteriol 182:2277–2284
Riemenschneider A, Nikiforova V, Hoefgen R, De Kok LJ, Papenbrock J (2005) Impact of elevated H(2)S on metabolite levels, activity of enzymes and expression of genes involved in cysteine metabolism. Plant Physiol Biochem 43:473–483
Santos PM, Benndorf D, Sa-Correia I (2004) Insights into Pseudomonas putida KT2440 response to phenol-induced stress by quantitative proteomics. Proteomics 4:2640–2652
Sekowska A, Kung HF, Danchin A (2000) Sulfur metabolism in Escherichia coli and related bacteria: facts and fiction. J Mol Microbiol Biotechnol 2:145–177
Setubal JC, dos Santos P, Goldman BS, Ertesvag H, Espin G, Rubio LM, Valla S, Almeida NF, Balasubramanian D, Cromes L, Curatti L, Du Z, Godsy E, Goodner B, Hellner-Burris K, Hernandez JA, Houmiel K, Imperial J, Kennedy C, Larson TJ, Latreille P, Ligon LS, Lu J, Maerk M, Miller NM, Norton S, O’Carroll IP, Paulsen I, Raulfs EC, Roemer R, Rosser J, Segura D, Slater S, Stricklin SL, Studholme DJ, Sun J, Viana CJ, Wallin E, Wang B, Wheeler C, Zhu H, Dean DR, Dixon R, Wood D (2009) Genome sequence of Azotobacter vinelandii, an obligate aerobe specialized to support diverse anaerobic metabolic processes. J Bacteriol 191:4534–4545
Smith AD, Agar JN, Johnson KA, Frazzon J, Amster IJ, Dean DR, Johnson MK (2001) Sulfur transfer from IscS to IscU: the first step in iron-sulfur cluster biosynthesis. J Am Chem Soc 123:11103–11104
Sörbo BH (1953) Crystalline rhodanese. Purification and physicochemical examination. Acta Chem Scand 7:1129–1136
Stipanuk MH, Ueki I, Dominy JE Jr, Simmons CR, Hirschberger LL (2009) Cysteine dioxygenase: a robust system for regulation of cellular cysteine levels. Amino Acids 37:55–63
Urbina HD, Silberg JJ, Hoff KG, Vickery LE (2001) Transfer of sulfur from IscS to IscU during Fe/S cluster assembly. J Biol Chem 276:44521–44526
Venkatraman A, Landar A, Davis AJ, Chamlee L, Sanderson T, Kim H, Page G, Pompilius M, Ballinger S, Darley-Usmar V, Bailey SM (2004) Modification of the mitochondrial proteome in response to the stress of ethanol-dependent hepatotoxicity. J Biol Chem 279:22092–22101
Westrop GD, Georg I, Coombs GH (2009) The mercaptopyruvate sulfurtransferase of Trichomonas vaginalis links cysteine catabolism to the production of thioredoxin persulfide. J Biol Chem 284:33485–33494
Williams RA, Kelly SM, Mottram JC, Coombs GH (2003) 3-Mercaptopyruvate sulfurtransferase of Leishmania contains an unusual C-terminal extension and is involved in thioredoxin and antioxidant metabolism. J Biol Chem 278:1480–1486
Winyard PG, Moody CJ, Jacob C (2005) Oxidative activation of antioxidant defence. Trends Biochem Sci 30:453–461
Zheng LM, White RH, Cash VL, Jack RF, Dean DR (1993) Cysteine desulfurase activity indicates a role for Nifs in metallocluster biosynthesis. Proc Natl Acad Sci USA 90:2754–2758
Zheng LM, White RH, Cash VL, Dean DR (1994) Mechanism for the desulfurization of l-cysteine catalyzed by the Nifs gene-product. Biochemistry 33:4714–4720
Zheng L, Cash VL, Flint DH, Dean DR (1998) Assembly of iron-sulfur clusters. Identification of an iscSUA-hscBA-fdx gene cluster from Azotobacter vinelandii. J Biol Chem 273:13264–13272
Acknowledgments
Mobility of researchers between Italy and Germany was sponsored by Vigoni project n. 0815171 (2009-2010; Ateneo Italo-Tedesco, Deutscher Akademischer Austausch Dienst) to FF and JP. FF was funded by “Fondo interno ricerca scientifica e tecnologica” (2005, 2006; Università degli Studi di Milano). We thank Eleonora di Paolo, Marco Pavoni and Dr. Aristodemo Carpen for skillful technical assistance. We also thank Prof. L.E. Vickery for providing E. coli IscSC328A and Prof. D.R. Dean and Dr. D. Johnson for providing pDB943 and pDB551.
Author information
Authors and Affiliations
Corresponding author
Additional information
F. Cartini and W. Remelli contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cartini, F., Remelli, W., Dos Santos, P.C. et al. Mobilization of sulfane sulfur from cysteine desulfurases to the Azotobacter vinelandii sulfurtransferase RhdA. Amino Acids 41, 141–150 (2011). https://doi.org/10.1007/s00726-010-0529-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-010-0529-z