Abstract
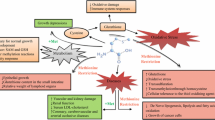
Sulfur amino acids (SAA), particularly methionine and cysteine, are critical for the gut to maintain its functions including the digestion, absorption and metabolism of nutrients, the immune surveillance of the intestinal epithelial layer and regulation of the mucosal response to foreign antigens. However, the metabolism of SAA in the gut, specifically the transmethylation of methionine, will result in a net release of homocysteine, which is shown to be associated with cardiovascular disease and stroke. Furthermore, the extensive catabolism of dietary methionine by the intestine or by luminal microbes may result in a decrease in nutritional efficiency. Therefore, the regulation of SAA metabolism in the gut is not only nutritionally relevant, but also relevant to the overall health and well-being. The superiority of dl-2-hydroxy-4-methylthiobutyrate to dl-methionine in decreasing homocysteine production, alleviating stress responses, and reducing the first-pass intestinal metabolism of dietary methionine may provide a promising implication for nutritional strategies to manipulate SAA metabolism and thus to improve the nutrition and health status of animals and perhaps humans.


Similar content being viewed by others
Abbreviations
- AA:
-
Amino acids
- BCAA:
-
Branched-chain amino acids
- d-AAOX:
-
d-Amino acid oxidase
- d-HADH:
-
d-2-Hydroxy acid dehydrogenase
- dl-HMTB:
-
dl-2-Hydroxy-4-methylthiobutyrate
- dl-MET:
-
dl-Methionine
- l-HAOX:
-
l-2-Hydroxy acid oxidase
- PDV:
-
Portal drained viscera
- SAA:
-
Sulfur amino acids
References
Baker DH (2009) Advances in protein–amino acid nutrition of poultry. Amino Acids 37:29–41
Baracos VE (2004) Animal models of amino acid metabolism: a focus on the intestine. J Nutr 134:1656S–1659S
Bauchart-Thevret C, Stoll B, Chacko S, Burrin DG (2009a) Sulfur amino acid deficiency upregulates intestinal methionine cycle activity and suppresses epithelial growth in neonatal pigs. Am J Physiol Endocrinol Metab 296:E1239–E1250
Bauchart-Thevret C, Stoll B, Burrin DG (2009b) Intestinal metabolism of sulfur amino acids. Nutr Res Rev 22:175–187
Bertolo RFP, Pencharz PB, Ball RO (2005) Role of intestinal first-pass metabolism on whole-body amino acid requirements. In: Burrin DG, Mersmann HJ (eds) Biology of metabolism in growing animals. Elsevier, London, pp 127–156
Blachier F, Mariotti F, Huneau JF, Tome D (2007) Effects of amino acid-derived luminal metabolites on the colonic epithelium and physiopathological consequences. Amino Acids 33:547–562
Bos C, Stoll B, Fouillet H, Gaudichon C, Guan X, Grusak MA, Reeds PJ, Tome D, Burrin DG (2003) Intestinal lysine metabolism is driven by the enteral availability of dietary lysine in piglets fed a bolus meal. Am J Physiol Endocrinol Metab 285:E1246–E1257
Brachet P, Puigserver A (1992) Regional differences for the d-amino acid oxidase-catalysed oxidation of d-methionine in chicken small intestine. Comp Biochem Physiol B 101:509–511
Brosnan JT, Brosnan ME (2006) The sulfur-containing amino acids: an overview. J Nutr 136:1636S–1640S
Burrin D, Stoll B, Chang X, Yu H, Reeds P (1999) Total parenteral nutrition does not compromise digestion or absorption of dietary protein in neonatal pigs. FASEB J 13:A1024
Campbell KCM, Rybak LP, Meech RP, Hughes L (1996) d-Methionine provides excellent protection from cisplatin ototoxicity in the rat. Hear Res 102:90–98
Chen LX, Yin YL, Jobgen WS, Jobgen SC, Knabe DA, Hu WX, Wu G (2007) In vitro oxidation of essential amino acids by intestinal mucosal cells of growing pigs. Livest Sci 109:19–23
Chen L, Li P, Wang J, Li X, Gao H, Yin Y, Hou Y, Wu G (2009) Catabolism of nutritionally essential amino acids in developing porcine enterocytes. Amino Acids 37:143–152
Dangin M, Boirie Y, Garcia-Rodenas C, Gachon P, Fauquant J, Callier P, Ballèvre O, Beaufrère B (2001) The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am J Physiol 280:E340–E348
Di Buono M, Wykes LJ, Cole DE, Ball RO, Pencharz PB (2003) Regulation of sulfur amino acid metabolism in men in response to changes in sulfur amino acid intakes. J Nutr 133:733–739
Dibner JJ, Buttin P (2002) Use of organic acids as a model to study the impact of gut microflora on nutrition and metabolism. J Appl Poult Res 11:453–463
Dibner JJ, Ivey FJ (1992) Capacity in the liver of the broiler chick for conversion of supplemental methionine activity to l-methionine. Poult Sci 71:1695–1699
Dibner JJ, Knight CD (1984) Conversion of 2-hydroxy-4-(methylthio)butanoic acid to l-methionine in the chick: a stereospecific pathway. J Nutr 114:1716–1723
Fang ZF, Luo J, Qi ZL, Huang FR, Zhao SJ, Liu MY, Jiang SW, Peng J (2009) Effects of 2-hydroxy-4-methylthiobutyrate on portal plasma flow and net portal appearance of amino acids in piglets. Amino Acids 36:501–509
Fang ZF, Huang FR, Luo J, Wei HK, Ma LB, Jiang SW, Peng J (2010a) Effects of dl-2-hydroxy-4-methylthiobutyrate on the first-pass intestinal metabolism of dietary methionine and its extraintestinal availability. Br J Nutr. doi:10.1017/S0007114509992169
Fang ZF, Luo HF, Wei HK, Huang FR, Qi ZL, Jiang SW, Peng J (2010b) Methionine metabolism in piglets fed DL-methionine or its hydroxy analogue was affected by distribution of enzymes oxidizing these sources to keto-methionine. J Agric Food Chem. doi:10.1021/jf903317x
Finkelstein JD (2000) Pathways and regulation of homocysteine metabolism in mammals. Semin Thromb Hemost 26:219–225
Garlick PT (2006) Toxicity of methionine in humans. J Nutr 136:1722S–1725S
Hiramatsu T, Fukagawa NK, Marchini JS, Cortiella J, Yu YM, Chapman TE, Young VR (1994) Methionine and cysteine kinetics at different intakes of cystine in healthy adult men. Am J Clin Nutr 60:525–533
Hogeveen M, Blom HJ, Van Amerongen M, Boogmans B, Van Beynum IM, Van De Bor M (2002) Hyperhomocysteinemia as risk factor for ischemic and hemorrhagic stroke in newborn infants. J Pediatr 141:429–431
Libao-Mercado AJ, Zhu CL, Cant JP, Lapierre H, Thibault JN, Sève B, Fuller MF, de Lange CF (2009) Dietary and endogenous amino acids are the main contributors to microbial protein in the upper gut of normally nourished pigs. J Nutr 139:1088–1094
Lobley GE, Wester TJ, Calder AG, Parker DS, Dibner JJ, Vázquez-Añón (2006) Absorption of 2-hydroxy-4-methylthiobutyrate and conversion to methionine in lambs. J Dairy Sci 89:1072–1080
Madara JL (1991) Functional morphology of epithelium of the small intestine. In: Shultz SG (ed) Handbook of physiology: the gastrointestinal system. American Physiological Society, Bethesda, MD, pp 83–120
Martín-Venegas R, Geraert PA, Ferrer R (2006) Conversion of the methionine hydroxy analogue dl-2-hydroxy-(4-methylthio) butanoic acid to sulfur-containing amino acids in the chicken small intestine. Poult Sci 85:1932–1938
Matsushita K, Takahashi K, Akiba Y (2007) Effects of adequate or marginal excess of dietary methionine hydroxy analogue free acid on growth performance, edible meat yields and inflammatory response in female broiler chickens. Poult Sci 44:265–272
McCollum MQ, Vázquez-Añón M, Dibner JJ, Webb KE Jr (2000) Absorption of 2-hydroxy-4-(methylthio)butanoic acid by isolated sheep ruminal and omasal epithelia. J Anim Sci 78:1078–1083
Printen KJ, Brummel MC, Ei Soon Cho MS, Stegink LD (1979) Utilization of d-methionine during total parenteral nutrition in postsurgical patients. Am J Clin Nutr 32:1200–1205
Raguso CA, Ajami AM, Gleason R, Young VR (1997) Effect of cystine intake on methionine kinetics and oxidation determined with oral tracers of methionine and cysteine in healthy adults. Am J Clin Nutr 66:283–292
Richards JD, Atwell CA, Vázquez-Añón M, Dibner JJ (2005) Comparative in vitro and in vivo absorption of 2-hydroxy-4(methylthio) butanoic acid and methionine in the broiler chicken. Poult Sci 84:1397–1405
Riedijk MA, van Goudoever JB (2007) Splanchnic metabolism of ingested amino acids in neonates. Curr Opin Clin Nutr Metab Care 10:58–62
Riedijk MA, Stoll B, Chacko S, Schierbeek H, Sunehag AL, van Goudoever JB, Burrin DG (2007) Methionine transmethylation and transsulfuration in the piglet gastrointestinal tract. Proc Natl Acad Sci USA 104:3408–3413
Saunderson CL (1985) Comparative metabolism of l-methionine, dl-methionine and dl-2-hydroxy-4-methylthiobutanoic acid by broiler chicks. Br J Nutr 54:621–633
Selhub J (1999) Homocysteine metabolism. Annu Rev Nutr 19:217–246
Shoveller AK, Brunton JA, Pencharz PB, Ball RO (2003a) The methionine requirement is lower in neonatal piglets fed parenterally than in those fed enterally. J Nutr 133:1390–1397
Shoveller AK, Brunton JA, House JD, Pencharz PB, Ball RO (2003b) Dietary cysteine reduces the methionine requirement by an equal proportion in both parenterally and enterally fed piglets. J Nutr 133:4215–4224
Shoveller AK, House JD, Brunton JA, Pencharz PB, Ball RO (2004) The balance of dietary sulfur amino acids and the route of feeding affect plasma homocysteine concentrations in neonatal piglets. J Nutr 134:609–612
Shoveller AK, Stoll B, Ball RO, Burrin DG (2005) Nutritional and functional importance of intestinal sulfur amino acid metabolism. J Nutr 135:1609–1612
Stegink LD, Besten LD (1972) Synthesis of cysteine from methionine in normal adult subjects: effect of route of alimentation. Science 178:514–516
Stipanuk MH (2004) Sulfur amino acid metabolism: pathways for production and removal of homocysteine and cysteine. Annu Rev Nutr 24:539–577
Stoll B, Burrin DG (2006) Measuring splanchnic amino acid metabolism in vivo using stable isotope tracers. J Anim Sci 84:E60–E72
Stoll B, Henry J, Reeds PJ, Yu H, Jahoor F, Burrin DG (1998) Catabolism dominates the first-pass intestinal metabolism of dietary essential amino acids in milk protein-fed piglets. J Nutr 128:606–614
Van Beynum IM, Smeitink JA, den Heijer M, te Poele Pothoff MT, Blom HJ (1999) Hyperhomocysteinemia: a risk factor for ischemic stroke in children. Circulation 99:2070–2072
van der Schoor SR, van Goudoever JB, Stoll B, Henry JF, Rosenberger JR, Burrin DG, Reeds PJ (2001) The pattern of intestinal substrate oxidation is altered by protein restriction in pigs. Gastroenterology 121:1167–1175
Van Goudoever JB, Stoll B, Henry JF, Burrin DG, Reeds PJ (2000) Adaptive regulation of intestinal lysine metabolism. Proc Natl Acad Sci USA 97:11620–11625
van Goudoever JB, van der Schoor SR, Stoll B, Burrin DG, Wattimena D, Schierbeek H, Schaart MW, Riedijk MA, van der Lugt J (2006) Intestinal amino acid metabolism in neonates. Nestle Nutr Workshop Ser Pediatr Program 58:95–102, discussion 102–108
van Goudoever JB, Corpeleijn W, Riedijk M, Schaart M, Renes I, van der Schoor S (2008) The impact of enteral insulin-like growth factor 1 and nutrition on gut permeability and amino acid utilization. J Nutr 138:1829S–1833S
Van Klinken BJ, Einerhand AW, Buller HA, Dekker J (1998) Strategic biochemical analysis of mucins. Anal Biochem 265:103–116
Velez-Carrasco W, Merkel M, Twiss CO, Smith JD (2008) Dietary methionine effects on plasma homocysteine and HDL metabolism in mice. J Nutr Biochem 19:362–370
Walter A, Gutknecht J (1984) Monocarboxylic acid permeation through lipid bilayer membranes. J Membr Biol 77:255–264
Wang WW, Qiao SY, Li DF (2009) Amino acids and gut function. Amino Acids 37:105–110
Wilson RH, Leibholz J (1981) Digestion in the pig between 7 and 35 d of age. 2. The digestion of dry matter and the pH of digesta in pigs given milk and soya-bean proteins. Br J Nutr 45:321–336
Windmueller HG (1982) Glutamine utilization by the small intestine. Adv Enzymol 53:201–237
Wu G (1998) Intestinal mucosa amino acid metabolism. J Nutr 128:1249–1252
Wu G (2009) Amino acids: metabolism, functions, and nutrition. Amino Acids 37:1–17
Wu G, Knabe DA, Flynn NE (2005) Amino acid metabolism in the small intestine: biochemical bases and nutritional significance. In: Burrin DG, Mersmann HJ (eds) Biology of metabolism in growing animals. Elsevier, London, pp 107–126
Xie M, Hou SS, Huang W, Fan HP (2007) Effect of excess methionine and methionine hydroxy analogue on growth performance and plasma homocysteine of growing Pekin ducks. Poult Sci 86:1995–1999
Acknowledgments
This work was supported by the National Natural Science Foundation of China (30901042), the Program for Changjiang Scholars and Innovative Research Team in the University (IRTO555), and the Key Project of Sichuan Provincial Education Department (00924100). The authors thank graduate students, technicians, and colleagues for their important contributions to the work described in this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fang, Z., Yao, K., Zhang, X. et al. Nutrition and health relevant regulation of intestinal sulfur amino acid metabolism. Amino Acids 39, 633–640 (2010). https://doi.org/10.1007/s00726-010-0502-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-010-0502-x