Abstract
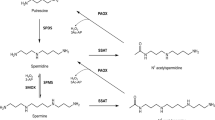
Ionic interactions are essential for the biological functions of the polyamines spermidine and spermine in mammalian physiology. Here, we describe a simple gram scale method to prepare 1,12-diamino-3,6,9-triazadodecane (SpmTrien), an isosteric charge-deficient spermine analogue. The protonation sites of SpmTrien were determined at pH range of 2.2–11.0 using two-dimensional 1H-15N NMR spectroscopy, which proved to be more feasible than conventional methods. The macroscopic pK a values of SpmTrien (3.3, 6.3, 8.5, 9.5 and 10.3) are significantly lower than those of 1,12-diamino-4,9-diazadodecane (spermine). The effects of SpmTrien and its parent molecule, 1,8-diamino-3,6-diazaoctane (Trien), on cell growth and polyamine metabolism were investigated in DU145 prostate carcinoma cells. SpmTrien downregulated the biosynthetic enzymes ornithine decarboxylase (ODC) and S-adenosyl-l-methionine decarboxylase and decreased intracellular polyamine levels, whereas the effects of Trien alone were minor. Interestingly, both SpmTrien and Trien were able to partially overcome growth arrest induced by an ODC inhibitor, α-difluoromethylornithine, indicating that they are able to mimic some functions of the natural polyamines. Thus, SpmTrien is a novel tool to influence polyamine interaction sites at the molecular level and offers a new means to study the contribution of the protonation of spermine amino group(s) in the regulation of polyamine-dependent biological processes.




Similar content being viewed by others
Abbreviations
- AdoMetDC:
-
S-adenosyl-L-methionine decarboxylase
- AG:
-
Aminoguanidine
- DENSpm:
-
N1,N11-diethylnorspermine
- DFMO:
-
α-Difluoromethylornithine (2,5-diamino-2-(difluoromethyl)pentanoic acid)
- HMBC:
-
Heteronuclear multiple bond correlation
- HSQC:
-
Heteronuclear single quantum correlation
- ODC:
-
Ornithine decarboxylase
- Pu:
-
Putrescine (1,4-diaminobutane)
- Spm:
-
Spermine (1,12-diamino-4,9-diazadodecane)
- SpmTrien:
-
(1,12-Diamino-3,6,9-triazadodecane)
- Spd:
-
Spermidine (1,8-diamino-4-azaoctane)
- SSAT:
-
Spermidine/spermine N 1-acetyltransferase
- Trien:
-
Triethylenetetramine (1,8-diamino-3,6-diazaoctane)
- TSP:
-
Sodium 3-trimethylsilyl[2,2,3,3-d 4]propionate
References
Agostinelli E, De Matteis G, Mondovì B, Morpurgo L (1998) Reconstitution of Cu2+-depleted bovine serum amine oxidase with Co2+. Biochem J 330:383–387
Baillon J, Mamont PS, Wagner J, Gerhart F, Lux P (1988) Fluorinated analogues of spermidine as substrates of spermine synthase. Eur J Biochem 176:237–242
Basu HS, Schwietert HC, Feuerstein BG, Marton LJ (1990) Effects of variation in the structure of spermine on the association with DNA and the induction of DNA conformational changes. Biochem J 269:329–334
Bencini A, Bianchi A, Garcia-España E, Micheloni M, Ramirez JA (1999) Proton coordination by polyamine compounds in aqueous solution. Coord Chem Rev 188:97–156
Bergeron RJ, Neims AH, McManis JS, Hawthorne TR, Vinson JR, Bortell R, Ingeno MJ (1988) Synthetic polyamine analogues as antineoplastics. J Med Chem 31:1183–1190
Bergeron RJ, McManis JS, Weimar WR, Schreier KM, Gao F, Wu Q, Ortiz-Ocasio J, Luchetta GR, Porter C, Vinson JR (1995) The role of charge in polyamine analogue recognition. J Med Chem 38:2278–2285
Bernacki RJ, Bergeron RJ, Porter CW (1992) Antitumor activity of N,N′-bis(ethyl)spermine homologues against human MALME-3 melanoma xenografts. Cancer Res 52:2424–2430
Casero RA Jr, Marton LJ (2007) Targeting polyamine metabolism and function in cancer and other hyperproliferative diseases. Nat Rev Drug Discov 6:373–390
Casero RA, Woster PM (2001) Terminally alkylated polyamine analogues as chemotherapeutic agents. J Med Chem 44:1–26
Cooper GJS, Phillips ARJ, Choong SY et al (2004) Regeneration of the heart in diabetes by selective copper chelation. Diabetes 53:2501–2508
Dixon HBF, Gibbs K, Walshe JM (1972) Preparation of triethylenetetramine dihydrochloride for the treatment of Wilson’s disease. Lancet 1:853
Eloranta TO, Khomutov AR, Khomutov RM, Hyvönen T (1990) Aminooxy analogues of spermidine as inhibitors of spermine synthase and substrates of hepatic polyamine acetylating activity. J Biochem (Tokyo) 108:593–598
Frassineti C, Ghelli S, Gans P, Sabatini A, Moruzzi MS, Vacca A (1995) Nuclear magnetic resonance as a tool for determining protonation constants of natural polyprotic bases in solution. Anal Biochem 231:374–382
Hyvönen T, Keinänen TA, Khomutov AR, Khomutov RM, Eloranta TO (1992) Monitoring of the uptake and metabolism of aminooxy analogues of polyamines in cultured cells by high-performance liquid chromatography. J Chromatogr 574:17–21
Hyvönen T, Keinänen TA, Khomutov AR, Khomutov RM, Eloranta TO (1995) Aminooxy analogues of spermidine evidence the divergent roles of the charged amino nitrogens in the cellular physiology of spermidine. Life Sci 56:349–360
Hyvönen MT, Howard M, Grigorenko N, Khomutov AR, Vepsäläinen J, Alhonen L, Jänne J, Keinänen TA (2009) Divergent regulation of the key metabolic enzymes of polyamine metabolism by chiral α-methylated polyamine analogs. Biochem J 422:321–328
Jänne J, Williams-Ashman HG (1971) On the purification of l-ornithine decarboxylase from rat prostate and effects of thiol compounds on the enzyme. J Biol Chem 246:1725–1732
Jones M, Singh P, Zimmerman L, Gomez M, Albina L, Domingo J (1995) Effects of some chelating agents on urinary copper excretion by the rat. Chem Res Toxicol 8:942–948
Khomutov AR, Vepsäläinen J, Shvetsov AS, Hyvonen T, Keinanen TA, Pustobaev VN, Eloranta TO, Khomutov RM (1996) Synthesis of hydroxylamine analogues of polyamines. Tetrahedron 52:13751–13766
Khomutov AR, Simonian AR, Vepsäläinen J, Keinänen TA, Alhonen L, Jänne J (2005) New oxaanalogues of spermine. Russ J Bioorganic Chem 31:206–212
Khomutov AR, Grigorenko NA, Skuridin SG (2007) Novel approach to design isosteric charge-deficient analogue of spermine and its biochemically important derivatives. Biochem Soc Trans 35:369–373
Kobayashi M, Fujisaki H, Sugawara M, Iseki K, Miyazaki K (1999) The presence of an Na+/spermine antiporter in the rat renal brush-border membrane. J Pharm Pharmacol 51:279–284
Laatikainen R, Niemitz M, Weber U, Sundelin J, Hassinen T, Vepsäläinen J (1996) General strategies for total-lineshape-type spectral analysis of NMR spectra using integral-transform iterator. J Magn Reson Ser A 120:1–10
Lin PKT, Maguire NM, Brown DM (1994) Synthesis of novel oxa-isosteres of spermidine and spermine. Tetrahedron Lett 35:3605–3608
Lu J, Chan YK, Gamble GD, Poppitt SD, Othman AA, Cooper GJ (2007) Triethylenetetramine and metabolites: levels in relation to copper and zinc excretion in urine of healthy volunteers and type 2 diabetic patients. Drug Metab Dispos 35:221–227
Nocera S, Marcocci L, Pietrangeli P, Mondovì B (2003) New perspectives on the role of amine oxidases in physiopathology. Amino Acids 24:13–17
Onufriev A, Case DA, Ullmann GM (2001) A novel view of pH titration in biomolecules. Biochemistry 40:3413–3419
Ramirez FJ, Thomas TJ, Antony T, Ruiz-Chica J, Thomas T (2002) Effects of aminooxy analogues of biogenic polyamines on aggregation and stability of calf thymus DNA. Biopolymers 65:148–157
Schilsky ML (2001) Treatment of Wilson’s disease: what are the relative roles of penicillamine, trientine, and zinc supplementation? Curr Gastroenterol Rep 3:54–59
Schwarzenbach G (1950) Metallkomplexe mit Polyaminen III: Mit Triäthylen-tetramin = “trien”. Helv Chem Acta 33:974–985
Seiler N (2004) Catabolism of polyamines. Amino Acids 26:217–233
Szakacs Z, Kraszni M, Noszal B (2004) Determination of microscopic acid-base parameters from NMR-pH titration. Anal Bioanal Chem 378:1428–1448
Turchanowa L, Shvetsov AS, Demin AV, Khomutov AR, Wallace HM, Stein J, Milovic V (2002) Insufficiently changed isosteric analogue of spermine: interaction with polyamine uptake, and effect on Caco-2 cell growth. Biochem Pharmacol 64:649–655
Ullmann GM (2003) Relations between protonation constants and titration curves in polyprotic acids: a critical view. J Phys Chem B 107:1263–1271
Wallace HM, Fraser AV (2004) Inhibitors of polyamine metabolism: review article. Amino Acids 26:353–365
Wallace HM, Niiranen K (2007) Polyamine analogues–an update. Amino Acids 33:261–265
Acknowledgments
We would like to thank Ms. Maritta Salminkoski, Ms. Tuula Reponen, Ms. Anne Karppinen, Ms. Arja Korhonen for their skilful technical assistance and Ph.D. Elina Jarho for her assistance with the automatic titrator. This work was supported by Academy of Finland (project nos. 124185 and 128702), Russian Foundation for Basic Research (project no. 09-04-01272), and the program Molecular and Cell Biology of the Presidium of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weisell, J., Hyvönen, M.T., Vepsäläinen, J. et al. Novel isosteric charge-deficient spermine analogue—1,12-diamino-3,6,9-triazadodecane: synthesis, pK a measurement and biological activity. Amino Acids 38, 501–507 (2010). https://doi.org/10.1007/s00726-009-0409-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-009-0409-6