Summary.
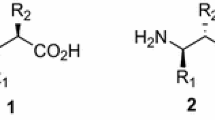
A procedure for the synthesis of enantiopure β3-amino acids from proteinogenic α-amino acids, developed by our group a few years ago, has been modified to enable the production of C-2 fully deuterated, C-protected β3-amino acids and, even more important, the synthesis of valuable deuterium labelled N(Boc)-protected chiral synthons, such as 2-aminoalcohols, 2-aminoiodides, and β3-amino nitriles.
Similar content being viewed by others
References
A Basak (1997) Enantioselective preparation of deuterated β-alanines E Juaristi (Eds) Enantioselective synthesis of β-amino acids Wiley VCH New York 465–480
Bolognese A, Fierro O, Guarino D, Longobardo L, Caputo R (2006) One-pot synthesis of orthogonally protected enantiopure S-(aminoalkyl)-cysteine derivatives. Eur J Org Chem, 169–173
Brandl M, Kozhushkov SI, Zlatopolskiy BD, Alvermann P, Geers B, Zeeck A, de Meijere A (2005) The biosynthesis of 3-(trans-2-nitrocyclopropyl)alanine, a constituent of the signal metabolite hormaomycin. Eur J Org Chem, 123–135
R Caputo E Cassano L Longobardo G Palumbo (1995a) ArticleTitleChiral N-protected β-iodoamines from α-aminoacids: a general synthesis Tetrahedron Lett 36 167–168 Occurrence Handle10.1016/0040-4039(94)02202-M Occurrence Handle1:CAS:528:DyaK2MXjtFClsbs%3D
R Caputo E Cassano L Longobardo G Palumbo (1995b) ArticleTitleSynthesis of enantiopure N- and C-protected homo-β-amino acids by direct homologation of α-amino acids Tetrahedron 51 12337–12550 Occurrence Handle10.1016/0040-4020(95)00778-7 Occurrence Handle1:CAS:528:DyaK2MXpt1OqsLo%3D
RP Cheng SH Gellman WF DeGrado (2001) ArticleTitleβ-Peptides: from structure to function Chem Rev 101 3219–3232 Occurrence Handle11710070 Occurrence Handle10.1021/cr000045i Occurrence Handle1:CAS:528:DC%2BD3MXntlGntbg%3D
NJ Church DW Young (1998) ArticleTitleSynthesis of the suicide substrate d-propargylglycine stereospecifically labelled with deuterium and investigation of its oxidation by d-amino acid oxidase J Chem Soc Perkin Trans 1 1475–1482 Occurrence Handle10.1039/a800579f
KH Gardner LE Kay (1997) ArticleTitleProduction and incorporation of 15N, 13C, 2H (1H-δ1 methyl) isoleucine into proteins for multidimensional NMR studies J Am Chem Soc 119 7599–7600 Occurrence Handle10.1021/ja9706514 Occurrence Handle1:CAS:528:DyaK2sXkvVems7g%3D
SP Gygi B Rist SA Gerber F Turecek MH Gelb R Aebersold (1999) ArticleTitleQuantitative analysis of complex protein mixtures using isotope-coded affinity tags Nat Biotechnol 17 994–996 Occurrence Handle10504701 Occurrence Handle10.1038/13690 Occurrence Handle1:CAS:528:DyaK1MXmvVelsLs%3D
B Lygo LD Humphreys (2002) ArticleTitleEnantioselective synthesis of α-carbon deuterium-labelled l-α-amino acids Tetrahedron Lett 43 6677–6679 Occurrence Handle10.1016/S0040-4039(02)01251-0 Occurrence Handle1:CAS:528:DC%2BD38XmsVWjsbk%3D
JA Rufián-Henares FJ Morales (2006) ArticleTitleDetermination of acrylamide in potato chips by a reversed-phase LC-MS method based on a stable isotope dilution assay Food Chem 97 555–562 Occurrence Handle10.1016/j.foodchem.2005.06.007
I Sack YS Balazs S Rahimipour S Vega (2000) ArticleTitleSolid-state NMR determination of peptide torsion angles: applications of 2H-dephased REDOR J Am Chem Soc 122 12263–12269 Occurrence Handle10.1021/ja000489w Occurrence Handle1:CAS:528:DC%2BD3cXotFynur0%3D
SP Schrimpf V Meskenaite E Brunner D Rutishauser P Walther J Eng R Aebersold P Sonderegger (2005) ArticleTitleProteomic analysis of synaptosomes using isotope-coded affinity tags and mass spectrometry Proteomics 5 2531–2541 Occurrence Handle15984043 Occurrence Handle10.1002/pmic.200401198 Occurrence Handle1:CAS:528:DC%2BD2MXmtF2gt7s%3D
D Seebach A Beck DJ Bierbaum (2004) ArticleTitleThe world of β- and γ-peptides comprised of homologated proteinogenic amino acids and other components Chem Biodiv 1 1111–1239 Occurrence Handle10.1002/cbdv.200490087 Occurrence Handle1:CAS:528:DC%2BD2cXnslCktb0%3D
Sewald N, Hiller KD, Helmreich B (1995) Asymmetric synthesis of β-amino acids and α-deuterated β-amino acids via conjugate addition of homochiral amidocuprates. Liebigs Ann, 925–928
T Stark H Justus T Hofmann (2006) ArticleTitleQuantitative analysis of N-phenylpropenoyl-l-amino acids in roasted coffee and cocoa powder by means of a stable isotope dilution assay J Agric Food Chem 54 2859–2867 Occurrence Handle16608201 Occurrence Handle10.1021/jf053207q Occurrence Handle1:CAS:528:DC%2BD28XhvF2isbg%3D
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Caputo, R., Longobardo, L. Enantiopure β3-amino acids-2,2-d2 via homologation of proteinogenic α-amino acids. Amino Acids 32, 401–404 (2007). https://doi.org/10.1007/s00726-006-0384-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-006-0384-0