Abstract
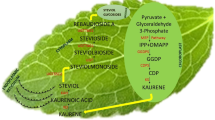
Dual metabolite, i.e., ginsenoside and anthocyanin, co-accumulating cell suspensions of Panax sikkimensis were subjected to elicitation with culture filtrates of Serratia marcescens (SD 21), Bacillus subtilis (FL11), Trichoderma atroviridae (TA), and T. harzianum (TH) at 1.25% and 2.5% v/v for 1- and 3-week duration. The fungal-derived elicitors (TA and TH) did not significantly affect biomass accumulation; however, bacterial elicitors (SD 21 and FL11), especially SD 21, led to comparable loss in biomass growth. In terms of ginsenoside content, differential responses were observed. A maximum of 3.2-fold increase (222.2 mg/L) in total ginsenoside content was observed with the use of 2.5% v/v TH culture filtrate for 1 week. Similar ginsenoside accumulation was observed with the use of 1-week treatment with 2.5% v/v SD 21 culture filtrate (189.3 mg/L) with a 10-fold increase in intracellular Rg2 biosynthesis (31 mg/L). Real-time PCR analysis of key ginsenoside biosynthesis genes, i.e., FPS, SQS, DDS, PPDS, and PPTS, revealed prominent upregulation of particularly PPTS expression (20–23-fold), accounting for the observed enhancement in protopanaxatriol ginsenosides. However, none of the elicitors led to successful enhancement in in vitro anthocyanin accumulation as compared to control values.






Similar content being viewed by others
References
Awad V, Kuvalekar A, Harsulkar A (2014) Microbial elicitation in root cultures of Taverniera cuneifolia (Roth) Arn. for elevated glycyrrhizic acid production. Ind Crop Prod 54:13–16. https://doi.org/10.1016/j.indcrop.2013.12.036
Baeg IH, So SH (2013) The world ginseng market and the ginseng (Korea). J Ginseng Res 37(1):1–7. https://doi.org/10.5142/jgr.2013.37.1
Biswas T (2016) Elicitation of in vitro secondary metabolite production and its transcript expression profiling in Panax species. PhD Thesis, Jawarharlal Nehru University, Delhi, India
Biswas T, Ajayakumar PV, Mathur AK, Mathur A (2015a) Solvent-based extraction optimization for efficient ultrasonication-assisted ginsenoside recovery from Panax quinquefolius and P. sikkimensis cell suspension lines. Natural Product Res 29(13):1256–1263. https://doi.org/10.1080/14786419.2015.1024119
Biswas T, Singh M, Mathur AK, Mathur A (2015b) A dual purpose cell line of an Indian congener of ginseng—Panax sikkimensis with distinct ginsenoside and anthocyanin production profiles. Protoplasma 252(2):697–703. https://doi.org/10.1007/s00709-014-0695-z
Biswas T, Kalra A, Mathur AK, Lal RK, Singh M, Mathur A (2016) Elicitors’ influenced differential ginsenoside production and exudation into medium with concurrent Rg3/Rh2 panaxadiol induction in Panax quinquefolius cell suspensions. Appl Microbiol Biotechnol 100(11):4909–4922. https://doi.org/10.1007/s00253-015-7264-z
Cai Z, Kastell A, Speiser C, Smetanska I (2013) Enhanced resveratrol production in Vitis vinifera cell suspension cultures by heavy metals without loss of cell viability. Appl Biochem Biotechnol 171(2):330–340. https://doi.org/10.1007/s12010-013-0354-4
Chodisetti B, Rao K, Gandi S, Giri A (2013) Improved gymnemic acid production in the suspension cultures of Gymnema sylvestre through biotic elicitation. Plant Biotechnol Rep 7(4):519–525. https://doi.org/10.1007/s11816-013-0290-3
Christensen LP (2009) Ginsenosides chemistry, biosynthesis, analysis and potential health effects. Adv Food Nutr Res 55:1–99. https://doi.org/10.1016/S1043-4526(08)00401-4
Condori J, Sivakumar G, Hubstenberger J, Dolan MC, Sobolev VS, Medina-Bolivar F (2010) Induced biosynthesis of resveratrol and the prenylated stilbenoids arachidin-1 and arachidin-3 in hairy root cultures of peanut: effects of culture medium and growth stage. Plant Physiol Biochem 48(5):310–318. https://doi.org/10.1016/j.plaphy.2010.01.008
Court WE (2000) Introduction to ginseng. In: Hardman R (ed) Ginseng—the genus Panax Hardwood. Academic Publishers, Netherlands, pp 1–11
Dörnenburg H, Knorr D (1995) Strategies for the improvement of secondary metabolite production in plant cell cultures. Enzyme Microbial Technol 17(8):674–684. https://doi.org/10.1016/0141-0229(94)00108-4
Gurung B, Bhardwaj PK, Rai AK, Sahoo D (2018) Major ginsenoside contents in rhizomes of Panax sokpayensis and Panax bipinnatifidus. Nat Prod Res 32(2):234–238. https://doi.org/10.1080/14786419.2017.1343322
Hu FX, Zhong JJ (2008) Jasmonic acid mediates gene transcription of ginsenoside biosynthesis in cell cultures of Panax notoginseng treated with chemically synthesized 2-hydroxyethyl jasmonate. Process Biochem 43(1):113–118. https://doi.org/10.1016/j.procbio.2007.10.010
Hu X, Neill S, Cai W, Tang Z (2003) Hydrogen peroxide and jasmonic acid mediate oligogalacturonic acid-induced saponin accumulation in suspension-cultured cells of Panax ginseng. Physiol Plant 118(3):414–421. https://doi.org/10.1034/j.1399-3054.2003.00124.x
Huang C, Zhong JJ (2013) Elicitation of ginsenoside biosynthesis in cell cultures of Panax ginseng by vanadate. Process Biochem 48(8):1227–1234. https://doi.org/10.1016/j.procbio.2013.05.019
Huang C, Qian ZG, Zhong JJ (2013) Enhancement of ginsenoside biosynthesis in cell cultures of Panax ginseng by N, N′-dicyclohexylcarbodiimide elicitation. J Biotechnol 165(1):30–36. https://doi.org/10.1016/j.jbiotec.2013.02.012
Kim C, Im H, Kim H, Huh H (2001) Accumulation of 2, 5-dimethoxy-1, 4-benzoquinone in suspension cultures of Panax ginseng by a fungal elicitor preparation and a yeast elicitor preparation. Appl Microbiol Biotechnol 56(1-2):239–242. https://doi.org/10.1007/s002530000557
Kim YS, Hahn EJ, Murthy HN, Paek KY (2004) Adventitious root growth and Ginsenoside accumulation in Panax ginseng cultures as affected by methyl jasmonate. Biotechnol Lett 26(21):1619–1622. https://doi.org/10.1007/s10529-004-3183-2
Kim YJ, Zhang D, Yang DC (2015) Biosynthesis and biotechnological production of ginsenosides. Biotechnol Adv 33(6):717–735. https://doi.org/10.1016/j.biotechadv.2015.03.001
Lee MH, Jeong JH, Seo JW, Shin CG, Kim YS, In JG, Yang DC, Yi JS, Choi YE (2004) Enhanced triterpene and phytosterol biosynthesis in Panax ginseng overexpressing squalene synthase gene. Plant Cell Physiol 45(8):976–984. https://doi.org/10.1093/pcp/pch126
Li J, Liu S, Wang J, Li J, Liu D, Li J, Gao W (2016) Fungal elicitors enhance ginsenosides biosynthesis, expression of functional genes as well as signal molecules accumulation in adventitious roots of Panax ginseng CA Mey. J Biotechnol 239:106–114. https://doi.org/10.1016/j.jbiotec.2016.10.011
Mañero F, Gutiérrezv J, Algar E, Martín Gómez MS, Saco Sierra MD, Solano BR (2012) Elicitation of secondary metabolism in Hypericum perforatum by rhizosphere bacteria and derived elicitors in seedlings and shoot cultures. Pharm Biol 50(10):1201–1209. https://doi.org/10.3109/13880209.2012.664150
Martinoia E, Klein M, Geisler M, Bovet L, Forestier C, Kolukisaoglu U, Muller-Rober B, Schulz B (2002) Multifunctionality of plant ABC transporters—more than just detoxifiers. Planta 214(3):345–355. https://doi.org/10.1007/s004250100661
Mathur A, Gangwar A, Mathur AK, Sangwan RS, Jain DC (2002) A procedure for the development of an anthocyanin producing callus line of Panax sikkimensis (Indian species of ginseng) US PATENT 6368860.
Mathur A, Mathur AK, Gangwar A, Verma P, Sangwan RS (2010) Anthocyanin production in a callus line of Panax sikkimensis Ban. In Vitro Cell Develop Biol-Plant 46(1):13–21. https://doi.org/10.1007/s11627-009-9253-3
Matkowski A (2008) Plant in vitro culture for the production of antioxidants—a review. Biotechnol Adv 26(6):548–560. https://doi.org/10.1016/j.biotechadv.2008.07.001
Mehta JK, Haridasan K (1992) The ginsengs in Arunachal Pradesh. Arunachal Forest news 10:56–58
Paek KY, Murthy HN, Hahn EJ, Zhong JJ (2009) Large scale culture of ginseng adventitious roots for production of ginsenosides. Adv Biochem Engg Biotechnol 113:151–176
Qianliang M, Su C, Zheng C, Jia M, Zhang Q, Zhang H, Rahman K, Han T, Qin L (2013) Elicitors from the endophytic fungus Trichoderma atroviride promote Salvia miltiorrhiza hairy root growth and tanshinone biosynthesis. J Exptl Bot 64:5687–5694
Radman R, Saez T, Bucke C, Keshavarz T (2003) Elicitation of plants and microbial cell systems. Biotechnol Appl Biochem 37(1):91–102. https://doi.org/10.1042/BA20020118
Rahimi S, Kim YJ, Devi BS, Oh JY, Kim SY, Kwon WS, Yang DC (2016) Sodium nitroprusside enhances the elicitation power of methyl jasmonate for ginsenoside production in Panax ginseng roots. Res Chem Intermediates 42(4):2937–2951. https://doi.org/10.1007/s11164-015-2188-x
Rao SR, Ravishankar GA (2002) Plant cell cultures: chemical factories of secondary metabolites. Biotechnol Adv 20(2):101–153
Robinson GW, Tsay YH, Kienzle BK, Smith-Montroy CA, Bishop RW (1993) Conservation between human and fungal squalene synthetases: similarities in structure, function, and regulation. Mol Cell Biol 13(5):2706–2717. https://doi.org/10.1128/MCB.13.5.2706
Ryu CM, Choi HK, Lee CH, Murphy JF, Lee JK, Kloepper JW (2013) Modulation of quorum sensing in acyl-homoserine lactone-producing or -degrading tobacco plants leads to alteration of induced systemic resistance elicited by the rhizobacterium Serratia marcescens 90-166. Plant Pathol J 29(2):182–192. https://doi.org/10.5423/PPJ.SI.11.2012.0173
Sharma SK, Pandit MK (2011) A morphometric analysis and taxonomic study of Panax bipinnatifidus species complex from Sikkim, Himalaya, India. Plant Syst Evol 297(1-2):87–98. https://doi.org/10.1007/s00606-011-0501-8
Sharma SK, Bisht MS, Pandit MK (2010) Synaptic mutation-driven male sterility in Panax sikkimensis (Ban) from Eastern Himalaya, India. Plant Syst Evol 287(1-2):29–36. https://doi.org/10.1007/s00606-010-0286-1
Silva FAS, Azevedo CAV (2016) The Assistant Software Version 7.7 and its use in the analysis of experimental data. Afr J Agric Res 11:3733–3740
Singh RK, Chaudhary BD (1979) Biometrical methods in quantitative genetic analysis. Kalyani Publishers, New Delhi
Smolenskaya IN, Reshetnyak OV, Nosov AV, Zoriniants SE, Chaiko AL, Smirnova YN, Nosov AM (2007) Ginsenoside production, growth and cytogenetic characteristics of sustained Panax japonicus var. repens cell suspension culture. Biol Plant 51(2):235–241. https://doi.org/10.1007/s10535-007-0047-3
Tansakul P, Shibuya M, Kushiro T, Ebizuka Y (2006) Dammarenediol-II synthase, the first dedicated enzyme for ginsenoside biosynthesis, in Panax ginseng. FEBS Lett 580(22):5143–5149. https://doi.org/10.1016/j.febslet.2006.08.044
Thanh NT, Murthy HN, Yu KW, Hahn EJ, Paek KY (2005) Methyl jasmonate elicitation enhanced synthesis of ginsenoside by cell suspension cultures of Panax ginseng in 5-l balloon type bubble bioreactors. Appl Microbiol Biotechnol 67(2):197–201. https://doi.org/10.1007/s00253-004-1759-3
Thanh NT, Ket NV, Yoeup PK (2007) Effect of medium composition on biomass and ginsenoside production in cell suspension culture of Panax vietnamensis Ha et Grushv. VNU J Sci Nat Sci Technol 23:269–274
Vasconsuelo A, Boland R (2007) Molecular aspects of the early stages of elicitation of secondary metabolites in plants. Plant Sci 172(5):861–875. https://doi.org/10.1016/j.plantsci.2007.01.006
Verma P, Khan SA, Mathur AK, Shanker K, Kalra A (2014) Fungal endophytes enhanced the growth and production kinetics of Vinca minor hairy roots and cell suspensions grown in bioreactor. Plant Cell Tissue Organ Cult 118(2):257–268. https://doi.org/10.1007/s11240-014-0478-4
Wang W, Zhang ZY, Zhong JJ (2005) Enhancement of ginsenoside biosynthesis in high-density cultivation of Panax notoginseng cells by various strategies of methyl jasmonate elicitation. Appl Microbiol Biotechnol 67(6):752–758. https://doi.org/10.1007/s00253-004-1831-z
Wang J, Gao W, Zhang J, Huang T, Wen T, Zhang L, Huang L (2011) Production of saponins and polysaccharides in the presence of lactalbumin hydrolysate in Panax quinquefolium L. cell cultures. Plant Growth Regul 63(3):217–223. https://doi.org/10.1007/s10725-010-9518-1
Wang J, Gao W, Zuo B, Zhang L, Huang L (2013) Effect of methyl jasmonate on the ginsenoside content of Panax ginseng adventitious root cultures and on the genes involved in triterpene biosynthesis. Res Chem Intermed 39(5):1973–1980. https://doi.org/10.1007/s11164-012-0730-7
Wu J, Zhong JJ (1999) Production of ginseng and its bioactive components plant cell culture: current technological and applied aspects. J Biotechnol 68(2-3):89–99. https://doi.org/10.1016/S0168-1656(98)00195-3
Xu MJ, Dong JF, Zhu MY (2005) Nitric oxide mediates the fungal elicitor-induced hypericin production of Hypericum perforatum cell suspension cultures through a jasmonic-acid-dependent signal pathway. Plant Physiol 139(2):991–998. https://doi.org/10.1104/pp.105.066407
Xu M, Yang B, Dong J, Lu D, Jin H, Sun L, Zhu Y, Xu X (2011) Enhancing hypericin production of Hypericum perforatum cell suspension culture by ozone exposure. Biotechnol Prog 27(4):1101–1106. https://doi.org/10.1002/btpr.614
Yu KW, Murthy HN, Jeong CS, Hahn EJ, Paek KY (2005) Organic germanium stimulates the growth of ginseng adventitious roots and ginsenoside production. Process Biochem 40(9):2959–2961. https://doi.org/10.1016/j.procbio.2005.01.015
Yu Y, Zhang WB, Li XY, Piao XC, Jiang J, Lian ML (2016) Pathogenic fungal elicitors enhance ginsenoside biosynthesis of adventitious roots in Panax quinquefolius during bioreactor culture. Ind Crop Prod 94:729–735. https://doi.org/10.1016/j.indcrop.2016.09.058
Zabala M, Angarita M, Restrepo J, Caicedo L, Perea M (2010) Elicitation with methyl-jasmonate stimulates peruvoside production in cell suspension cultures of Thevetia peruviana. In Vitro Cell Dev Biol-Plant 46(3):233–238. https://doi.org/10.1007/s11627-009-9249-z
Zhao J, Lawrence CD, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23(4):283–333. https://doi.org/10.1016/j.biotechadv.2005.01.003
Acknowledgements
The authors are grateful to the Director, CSIR-CIMAP, for the infrastructure and the lab facilities provided for the studies. TB also acknowledges the award of a Senior Research Fellowship (SRF) granted by the University Grants Commission, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical compliance
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Biswas, T., Pandey, S.S., Maji, D. et al. Enhanced expression of ginsenoside biosynthetic genes and in vitro ginsenoside production in elicited Panax sikkimensis (Ban) cell suspensions. Protoplasma 255, 1147–1160 (2018). https://doi.org/10.1007/s00709-018-1219-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-018-1219-z