Abstract
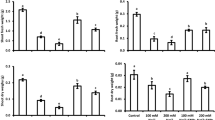
Materials with a particle size less than 100 nm are classified as nano-materials. The physical and chemical properties of nano-materials can vary considerably from those of bulk materials of the same composition. Silicon (Si) still fails to get recognized as an essential nutrient for plant growth and development, however the beneficial effects in terms of growth, biotic and abiotic stress resistance have been indicated in a variety of plant species for their growth. The aim of this study was to investigate the effects of different nano-silicon rates on the growth and antioxidant activities of soybean (Glycine max L. cv. M7) under salt stress. The results showed that salinity decreased shoot and root dry weight, potassium (K+) concentration in the root and leaf; however, increased sodium (Na+) concentration, catalase, peroxidase, ascorbate peroxidase and superoxide dismutase activities, phenolic components, ascorbic acid and α-tocopherol contents, lipid peroxidation, hydrogen peroxide, and oxygen radical’s concentration. Between the treatments, 0.5 and 1 mM of nanosilicon oxide (nano-SiO2) improved shoot and root growth of seedlings. In contrast, a foliar application of SiO2 at 2 mM reduced the soybean growth. Overall, exogenous nano-silicon alleviated the salt stress by increase in K+ concentration, antioxidant activities, non-enzymatic compounds and decreasing of Na+ concentration, lipid peroxidation, and reactive oxygen species production.



Similar content being viewed by others
References
Adrees M, Ali S, Rizwan M, Zia-ur-Rehman M, Ibrahim M, Abbas F, Farid M, Qayyum MF, Irshad MK (2015) Mechanisms of silicon-mediated alleviation of heavy metal toxicity in plants: a review. Ecotoxicol Environ Saf 119:186–197. https://doi.org/10.1016/j.ecoenv.2015.05.011
Ainsworth EA, Gillespie KM (2007) Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin–Ciocalteu reagent. Nat Protoc 2(4):875–877. https://doi.org/10.1038/nprot.2007.102
Almutairi ZM (2016) Effect of nano-silicon application on the expression of salt tolerance genes in germinating tomato (Solanum lycopersicum L.) seedlings under salt stress. Plant OMICS 9:106–114
Ashraf M (2009) Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol Adv 27(1):84–93. https://doi.org/10.1016/j.biotechadv.2008.09.003
Deshmukh RK, Vivancos J, Guérin V, Sonah H, Labbé C, Belzile F, Bélanger RR (2013) Identification and functional characterization of silicon transporters in soybean using comparative genomics of major intrinsic proteins in Arabidopsis and rice. Plant Mol Biol 83(4-5):303–315. https://doi.org/10.1007/s11103-013-0087-3
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32(1):93–101. https://doi.org/10.1093/jxb/32.1.93
Epstein E (1999) Silicon. Annu Rev Plant Biol 50(1):641–664. https://doi.org/10.1146/annurev.arplant.50.1.641
Farhangi-Abriz S, Torabian S (2017) Antioxidant enzyme and osmotic adjustment changes in bean seedlings as affected by biochar under salt stress. Ecotoxicol Environ Saf 137:64–70. https://doi.org/10.1016/j.ecoenv.2016.11.029
Fathi A, Zahedi M, Torabian S, Khoshgoftar A (2017) Response of wheat genotypes to foliar spray of ZnO and Fe2O3 nanoparticles under salt stress. J Plant Nutr 40(10):1376–1385. https://doi.org/10.1080/01904167.2016.1262418
Foyer CH, Noctor G (2003) Redox sensing and signaling associated with reactive oxygen in chloroplasts, peroxisomes and mitochondria. Physiol Plant 119(3):355–364. https://doi.org/10.1034/j.1399-3054.2003.00223.x
Gajewska E, Sklodowska M (2007) Effect of nickel on ROS content and antioxidative enzyme activities in wheat leaves. Biometals 20(1):27–36. https://doi.org/10.1007/s10534-006-9011-5
Garratt LC, Janagoudar BS, Lowe KC, Anthony P, Power JB, Davey MR (2002) Salinity tolerance and antioxidant status in cotton cultures. Free Radic Biol Med 33(4):502–511. https://doi.org/10.1016/S0891-5849(02)00838-9
Gottschalk F, Sun T, Nowak B (2013) Environmental concentrations of engineered nanomaterials: review of modeling and analytical studies. Environ Pollut 181:287–300. https://doi.org/10.1016/j.envpol.2013.06.003
Grieve CM, Grattan SR, Maas EV (2012) Plant salt tolerance. In: Wallender WW, Tanji KK (eds) ASCE manual and reports on engineering practice no. 17 agricultural salinity assessment and management, 2nd edn. ASCE, Reston. Chapter 13, pp 405–459
Gueta-Dahan Y, Yaniv Z, Zilinskas BA, Ben-Hayyim G (1997) Salt and oxidative stress: similar and specific responses and their relation to salt tolerance in citrus. Planta 203(4):460–469. https://doi.org/10.1007/s004250050215
Haghighi M, Pessarakli M (2013) Influence of silicon and nano-silicon on salinity tolerance of cherry tomatoes (Solanum lycopersicum L.) at early growth stage. Sci Hortic 161:111–117. https://doi.org/10.1016/j.scienta.2013.06.034
Haghighi M, Afifipour Z, Mozafarian M (2012) The effect of N-Si on tomato seed germination under salinity levels. J Biol Environ Sci 6:87–90
Hamayun M, Khan SA, Khan AL, Shinwari ZK, Hussain J, Sohn EY, Kang SM, Kim YH, Khan MA, Lee IJ (2010) Effect of salt stress on growth attributes and endogenous growth hormones of soybean cultivar Hwangkeumkong. Pak J Bot 42:3103–3112
Hemeda HM, Klein BP (1990) Effects of naturally occurring antioxidants on peroxidase activity of vegetable extracts. J Food Sci 55(1):184–185. https://doi.org/10.1111/j.1365-2621.1990.tb06048.x
Hossain MS, El Sayed AI, Moore M, Dietz KJ (2017) Redox and reactive oxygen species network in acclimation for salinity tolerance in sugar beet. J Exp Bot 68(5):1283–1298. https://doi.org/10.1093/jxb/erx019
Hsu YT, Kao CH (2007) Toxicity in leaves of rice exposed to cadmium is due to hydrogen peroxide accumulation. Plant Soil 298(1-2):231–241. https://doi.org/10.1007/s11104-007-9357-7
Ibrahim W, Ahmed IM, Chen X, Cao F, Zhu S, Wu F (2015) Genotypic differences in photosynthetic performance, antioxidant capacity, ultrastructure and nutrients in response to combined stress of salinity and Cd in cotton. Biometals 28(6):1063–1078. https://doi.org/10.1007/s10534-015-9890-4
Ismail H, Maksimović JD, Maksimović V, Shabala L, Živanović BD, Tian Y, Jacobsen SE, Shabala S (2016) Rutin, a flavonoid with antioxidant activity, improves plant salinity tolerance by regulating K+ retention and Na+ exclusion from leaf mesophyll in quinoa and broad beans. Funct Plant Biol 43:75–86
Karimi J, Mohsenzadeh S (2016) Effects of silicon oxide nanoparticles on growth and physiology of wheat seedlings. Russ J Plant Physiol 63(1):119–123. https://doi.org/10.1134/S1021443716010106
Konyalιoglu S, Sağlam H, Kιvçak B (2005) α-tocopherol, flavonoid, and phenol contents and antioxidant activity of Ficus carica. leaves. Pharm Biol 43(8):683–686. https://doi.org/10.1080/13880200500383538
Lee SK, Sohn EY, Hamayun M, Yoon JY, Lee IJ (2010) Effect of silicon on growth and salinity stress of soybean plant grown under hydroponic system. Agrofor Syst 80(3):333–340. https://doi.org/10.1007/s10457-010-9299-6
Liang Y, Chen QIN, Liu Q, Zhang W, Ding R (2003) Exogenous silicon (Si) increases antioxidant enzyme activity and reduces lipid peroxidation in roots of salt-stressed barley (Hordeum vulgare L.) J Plant Physiol 160(10):1157–1164. https://doi.org/10.1078/0176-1617-01065
Liang Y, Zhang W, Chen Q, Liu Y, Ding R (2006) Effect of exogenous silicon (Si) on H+-ATPase activity, phospholipids and fluidity of plasma membrane in leaves of salt-stressed barley (Hordeum vulgare L.) Environ Exp Bot 57(3):212–219. https://doi.org/10.1016/j.envexpbot.2005.05.012
Liu Y, Zhang Z, Zhang Q, Baker GL, Worden RM (2014) Biomembrane disruption by silica-core nanoparticles: effect of surface functional group measured using a tethered bilayer lipid membrane. Biochim Biophys Acta 1838(1):429–437. https://doi.org/10.1016/j.bbamem.2013.09.007
Ma JF (2004) Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci Plant Nutr 50(1):11–18. https://doi.org/10.1080/00380768.2004.10408447
Ma JF, Miyake Y, Takahashi E (2001) Silicon as a beneficial element for crop plants. Stud Plant Sci 31:17–39
Mahajan P, Dhoke SK, Khanna AS (2011) Effect of nano-ZnO particle suspension on growth of mung (Vigna radiata) and gram (Cicer arietinum) seedlings using plant agar method. J Nanotechnol 696535:1–7
Miller G, Shulaev V, Mittler R (2008) Reactive oxygen signaling and abiotic stress. Physiol Plant 133(3):481–489. https://doi.org/10.1111/j.1399-3054.2008.01090.x
Mishra P, Bhoomika K, Dubey RS (2013) Differential responses of antioxidative defense system to prolonged salinity stress in salt-tolerant and salt-sensitive Indica rice (Oryza sativa L.) seedlings. Protoplasma 250(1):3–19. https://doi.org/10.1007/s00709-011-0365-3
Mozafariyan M, Kamelmanesh MM, Hawrylak-Nowak B (2016) Ameliorative effect of selenium on tomato plants grown under salinity stress. Arch Agron Soil Sci 62(10):1368–1380. https://doi.org/10.1080/03650340.2016.1149816
Mukherjee SP, Choudhuri MA (1983) Implications of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant 58(2):166–170. https://doi.org/10.1111/j.1399-3054.1983.tb04162.x
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Nasir Khan M, Mobin M, Abbas ZK, AlMutairi KA, Siddiqui ZH (2017) Role of nanomaterials in plants under challenging environments. Plant Physiol Biochem 110:194–209
Nikolic M, Nikolic N, Liang Y, Kirkby EA, Romheld V (2007) Germanium-68 as an adequate tracer for silicon transport in plants. Characterization of silicon uptake in different crop species. Plant Physiol 143(1):495–503. https://doi.org/10.1104/pp.106.090845
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Biol 49(1):249–279. https://doi.org/10.1146/annurev.arplant.49.1.249
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60(3):324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Parveen N, Ashraf M (2010) Role of silicon in mitigating the adverse effects of salt stress on growth and photosynthetic attributes of two maize (Zea Mays L.) cultivars grown hydroponically. Pak J Bot 42:1675–1684
Pedersen P, Kumudini S, Board J, Conley S (2014) Soybean growth and development. Iowa State University, University Extension, United States, Ames
Pignocchi C, Foyer CH (2003) Apoplastic ascorbate metabolism and its role in the regulation of cell signalling. Curr Opin Plant Biol 6(4):379–389. https://doi.org/10.1016/S1369-5266(03)00069-4
Qi M, Liu Y, Li T (2013) Nano-TiO2 improve the photosynthesis of tomato leaves under mild heat stress. Biol Trace Elem Res 156(1-3):323–328. https://doi.org/10.1007/s12011-013-9833-2
Ranjit SL, Manish P, Penna S (2016) Early osmotic, antioxidant, ionic, and redox responses to salinity in leaves and roots of Indian mustard (Brassica juncea L.) Protoplasma 253(1):101–110. https://doi.org/10.1007/s00709-015-0792-7
Rasool S, Ahmad A, Siddiqi TO, Ahmad P (2013) Changes in growth, lipid peroxidation and some key antioxidant enzymes in chickpea genotypes under salt stress. Acta Physiol Plant 35(4):1039–1050. https://doi.org/10.1007/s11738-012-1142-4
Rico CM, Morales MI, McCreary R, Castillo-Michel H, Barrios AC, Hong J, Tafoya A, Lee WY, Varela-Ramirez A, Peralta-Videa JR, Gardea-Torresdey JL (2013) Cerium oxide nanoparticles modify the antioxidative stress enzyme activities and macromolecule composition in rice seedlings. Environ Sci Technol 47(24):14110–14118. https://doi.org/10.1021/es4033887
Sharma SS, Dietz KJ (2009) The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci 14(1):43–50. https://doi.org/10.1016/j.tplants.2008.10.007
Silva EN, Silveira JAG, Rodrigues CRF, Viegas RA (2015) Physiological adjustment to salt stress in Jatropha curcas is associated with accumulation of salt ions, transport and selectivity of K+, osmotic adjustment and K+/Na+ homeostasis. Plant Biol 17(5):1023–1029. https://doi.org/10.1111/plb.12337
Singh BK, Sharma SR, Singh B (2010) Antioxidant enzymes in cabbage: variability and inheritance of superoxide dismutase, peroxidase and catalase. Sci Hortic 124(1):9–13. https://doi.org/10.1016/j.scienta.2009.12.011
Singh R, Parihar P, Singh S, Mishra RK, Singh VP, Prasad SM (2017) Reactive oxygen species signaling and stomatal movement: current updates and future perspectives. Redox Biol 11:213–218. https://doi.org/10.1016/j.redox.2016.11.006
Smirnoff N, Wheeler GL (2000) Ascorbic acid in plants: biosynthesis and function. Crit Rev Biochem Mol Biol 35(4):291–314. https://doi.org/10.1080/10409230008984166
Soliman AS, El-feky SA, Darwish E (2015) Alleviation of salt stress on Moringa peregrina using foliar application of nanofertilizers. J Hortic For 7:36–47
Soundararajan P, Manivannan A, Ko CH, Jeong BR (2017) Silicon enhanced redox homeostasis and protein expression to mitigate the salinity stress in Rosa hybrida ‘Rock Fire’. J Plant Growth Regul:1–19. https://doi.org/10.1007/s00344-017-9705-7
Suriyaprabha R, Karunakaran G, Yuvakkumar R, Prabu P, Rajendran V, Kannan N (2012) Growth and physiological responses of maize (Zea mays L.) to porous silica nanoparticles in soil. J Nanopart Res 14(12):1294–1296. https://doi.org/10.1007/s11051-012-1294-6
Torabian S, Zahedi M, Khoshgoftar AH (2016) Effects of foliar spray of two kinds of zinc oxide on the growth and ion concentration of sunflower cultivars under salt stress. J Plant Nutr 39(2):172–180. https://doi.org/10.1080/01904167.2015.1009107
Torabian S, Zahedi M, Khoshgoftar AH (2017) Effects of foliar spray of nano-particles of FeSO4 on the growth and ion content of sunflower under saline condition. J Plant Nutr 40(5):615–623. https://doi.org/10.1080/01904167.2016.1240187
Wang SY, Jiao H (2000) Scavenging capacity of berry crops on superoxide radicals, hydrogen peroxide, hydroxyl radicals, and singlet oxygen. J Agric Food Chem 48(11):5677–5684. https://doi.org/10.1021/jf000766i
Wang S, Liu P, Chen D, Yin L, Li H, Deng X (2015) Silicon enhanced salt tolerance by improving the root water uptake and decreasing the ion toxicity in cucumber. Front Plant Sci 6:1–10
Worms IA, Boltzman J, Garcia M, Slaveykova VI (2012) Cell-wall-dependent effect of carboxyl-CdSe/ZnS quantum dots on lead and copper availability to green microalgae. Environ Pollut 167:27–33. https://doi.org/10.1016/j.envpol.2012.03.030
Xie Y, Li B, Zhang Q, Zhang C (2012) Effects of nano-silicon dioxide on photosynthetic fluorescence characteristics of Indocalamus barbatus McClure. J Nanjing Univ Nat Sci 2:59–63
Yamane K, Taniguchi M, Miyake H (2012) Salinity-induced subcellular accumulation of H2O2 in leaves of rice. Protoplasma 249(2):301–308. https://doi.org/10.1007/s00709-011-0280-7
Yeo AR, Flowers SA, Rao G, Welfare K, Senanayake N, Flowers TJ (1999) Silicon reduces sodium uptake in rice (Oryza sativa L.) in saline conditions and this is accounted for by a reduction in the transpirational bypass flow. Plant Cell Environ 22(5):559–565. https://doi.org/10.1046/j.1365-3040.1999.00418.x
Yuvakkumar R, Elango V, Rajendran V, Kannan NS, Prabu P (2011) Influence of nanosilica powder on the growth of maize crop (Zea mays L.) Int J Green Nanotechnol 3:180–190
Zhu Y, Gong H (2014) Beneficial effects of silicon on salt and drought tolerance in plants. Agron Sustain Dev 34(2):455–472. https://doi.org/10.1007/s13593-013-0194-1
Acknowledgements
The authors are thankful to Dr. Judith Rathjen for the help in improving the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Handling Editor: Néstor Carrillo
Rights and permissions
About this article
Cite this article
Farhangi-Abriz, S., Torabian, S. Nano-silicon alters antioxidant activities of soybean seedlings under salt toxicity. Protoplasma 255, 953–962 (2018). https://doi.org/10.1007/s00709-017-1202-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-017-1202-0