Abstract
Background
The optimal treatment for spinal meningioma is complete resection. The radicality of resection is the most important predictive factor for future tumor recurrence. Although clinical series dedicated to spinal meningiomas are relatively frequent, only a minimum of these defines the length of the required follow-up and difference in tumor recurrence in the context of Simpson grade I and II resection. Therefore, we propose reconsideration of surgical treatment and long-term follow-up based on a retrospective analysis of 84 patients who underwent Simpson grade II resection.
Methods
The study included 84 patients operated between 1998 and 2018. Clinical symptomatology, age, sex, risk of comorbidities, spinal level, duration of symptoms, surgical resection radicality, tumor recurrence, and complications associated with treatment were recorded and evaluated.
Results
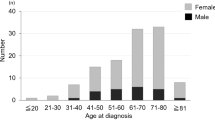
We encountered the diagnosis of spinal meningioma considerably more often in women (81%) than in men (19%). The average age of all patients was 65 years. Most meningiomas were located in the thoracic spine (82%) while the rest (18%) were located in the cervical spine. The most common symptoms were motor deficit (80%) and sensation disorders (70%). Of the symptomatic patients, 71% clinically improved, 27% showed no change and 2% worsened postoperatively. The mean follow-up was 32 months (range 1–204). During this period, there was a 5% tumor recurrence rate. However, when we analyzed a subgroup of nine patients who were monitored for more than 6 years, tumor recurrence was diagnosed in 44%, all of whom were women (mean age 51 years).
Conclusion
Our results indicate that tumor recurrence after Simpson II resection could be significantly higher than previously thought if the follow-up is long enough, especially in younger patients. This finding suggests we consider using radical Simpson grade I resection more frequently. The study also demonstrates that after spinal meningioma surgery the patients should be followed and monitored on a long-term basis.


Similar content being viewed by others
Abbreviations
- MMS:
-
Modified McCormick scale
- MRI:
-
Magnetic resonance imaging
- WHO:
-
World Health Organization
References
Duong LM, McCarthy BJ, McLendon RE, Dolecek TA, Kruchko C, Douglas LL, Ajani UA (2012) Descriptive epidemiology of malignant and nonmalignant primary spinal cord, spinal meninges, and cauda equina tumors, United States, 2004-2007. Cancer 118(17):4220–4227
Helseth A, Mørk SJ (1989) Primary intraspinal neoplasms in Norway, 1955 to 1986: a population-based survey of 467 patients. J Neurosurg 71(6):842–845
Hua L, Zhu H, Deng J, Tian M, Jiang X, Tang H, Luan S, Wakimoto H, Xie Q, Gong Y (2018) Clinical and prognostic features of spinal meningioma: a thorough analysis from a single neurosurgical center. J Neuro-Oncol 140(3):639–647
Kshettry VR, Hsieh JK, Ostrom QT, Kruchko C, Benzel EC, Barnholtz-Sloan JS (2015) Descriptive epidemiology of spinal meningiomas in the United States. Spine (Phila Pa 1976) 40(15):E886–E889
Levy WJ, Bay J, Dohn D (1982) Spinal cord meningioma. J Neurosurg 57:804–812
Solero CL, Fornari M, Giombini S, Lasio G, Oliveri G, Cimino C, Pluchino F, Gelber BR (1989) Spinal meningiomas: review of 174 operated cases. Neurosurgery 25(2):153–160
Westwick HJ, Yuh SJ, Shamji MF (2015) Complication avoidance in the resection of spinal meningiomas. World Neurosurg 83(4):627–634
Gilard V, Goia A, Ferracci FX, Marguet F, Magne N, Langlois O, Perez A, Derrey S (2018) Spinal meningioma and factors predictive of post-operative deterioration. J Neuro-Oncol 140(1):49–54
Westwick HJ, Shamji MF (2015) Effects of sex on the incidence and prognosis of spinal meningiomas: a surveillance, epidemiology, and end results study. J Neurosurg Spine 23(September):368–373
Wiemels J, Wrensch M, Claus EB (2010) Epidemiology and etiology of meningioma. J Neuro-Oncol 99:307–314
Wigertz A, Lonn S, Mathiesen T, Ahlbom A, Hall P, Feychting M (2006) Original contribution risk of brain tumors associated with exposure to exogenous female sex hormones. Am J Epidemiol 164(7):629–636
Bayoumi AB, Laviv Y, Yokus B, Efe IE, Toktas ZO, Kilic T, Demir MK, Konya D, Kasper EM (2017) Proposal of a new radiological classification system for spinal meningiomas as a descriptive tool and surgical guide. Clin Neurol Neurosurg 162(September):118–126
Noh SH, Kim KH, Shin DA et al (2019) Treatment outcomes of 17 patients with atypical spinal meningioma, including 4 with metastases: a retrospective observational study. Spine J 19(2):276–284
Nakamura M, Toyama Y, Chiba K, Fujiyoshi K, Tsuji O, Tsuji T, Matsumoto M, Watanabe K, Ishii K, Hosogane N (2011) Long-term surgical outcomes of spinal meningiomas. Spine (Phila Pa 1976) 37(10):E617–E623
Yamamuro K, Seichi A, Kimura A, Kikkawa I, Kojima M (2012) Histological investigation of resected dura mater. Spine (Phila Pa 1976) 37(22):13–16
McCormick P, Roland T, Post K, Stein BM (1990) Intramedullary ependymoma of the spinal cord. J Neurosurg 72:523–532
Simpson D (1957) The recurrence of intracranial meningiomas after surgical treatment. J Neurol Neurosurg Psychiatry 20(1):22–39
Namer IJ, Pamir MN, Benli K, Saglam S, Erbengi E (1987) Spinal meningiomas. Neurochirurgia (Stuttg) 30:11–15
Maiti TK, Guthikonda B, Patra DP, Nanda A, Bir SC, Kalakoti P (2016) Spinal meningiomas: clinicoradiological factors predicting recurrence and functional outcome. Neurosurg Focus 41(August):E6
Cushing H, Eisenhardt L (1938) Meningiomas. Their classification, regional behaviour, life history, and surgical end results. Springfield, I11 Charles C.:735
Gezen F, Kahraman S, Çanakci Z, Bedük A (2000) Review of 36 cases of spinal cord meningioma. Spine (Phila Pa 1976) 25(6):727–731
Sandalcioglu IE, Bassiouni H, Müller O et al (2015) Surgical outcome of spinal canal meningiomas. J Neuro-Oncol 25(2):4025–4032
Arima H, Takami T, Yamagata T, Naito K, Abe J, Shimokawa N (2014) Surgical management of spinal meningiomas: a retrospective case analysis based on preoperative surgical grade. Surg Neurol Int 5:333–339
Boström A, Bürgel U, Reinacher P, Krings T, Rohde V, Gilsbach JM, Hans FJ (2008) A less invasive surgical concept for the resection of spinal meningiomas. Acta Neurochir 150(6):551–556
Cohen-Gadol AA, Zikel OM, Koch CA, Scheithauer BW, Krauss WE (2003) Spinal meningiomas in patients younger than 50 years of age: a 21-year experience. J Neurosurg 98(3 SUPPL):258–263
Gottfried ON, Gluf W, Quinones-Hinojosa A, Kan P, Schmidt MH (2008) Spinal meningiomas: surgical management and outcome. Neurosurg Focus 14(6):1–7
Iacob G (2014) Spinal meningiomas. Personal experience and review of literature. Rom Neurosurg 21:146–160
Maiuri F, Del Basso ML, Caro D, De Divitiis O, Vergara P, Mariniello G (2011) Spinal meningiomas : age-related features. Clin Neurol Neurosurg 113(1):34–38
Peker S, Cerci A, Ozgen S, Isik N, Kalelioglu M, Pamir MN (2005) Spinal meningiomas: evaluation of 41 patients. J Neurosurg Sci 49(1):7–11
Postalci L, Tugcu B, Gungor A, Guclu G (2011) Spinal meningiomas: recurrence in ventrally located individuals on long-term follow-up: a review of 46 operated cases. Turk Neurosurg 21:449–453
Raco A, Pesce A, Toccaceli G, Domenicucci M (2018) Factors leading to a poor functional outcome in spinal meningioma surgery : remarks on 173 cases. Neurosurgery 80(4):602–609
Riad H, Knafo S, Segnarbieux F, Lonjon N (2013) Spinal meningiomas: surgical outcome and literature review. Neurochirurgie 59(1):30–34
Sandalcioglu IE, Bassiouni H, Müller O, Hunold A, Asgari S, Stolke D (2008) Spinal meningiomas: critical review of 131 surgically treated patients. Eur Spine J 17(8):1035–1041
Santos RC, de Amoreira Gepp R (2018) Benefits of spinal meningioma resection. Surg Neurol Int (9):16. https://doi.org/10.4103/sni.sni_409_17
Schaller B (2005) Spinal meningioma: relationship between histological subtypes and surgical outcome? J Neuro-Oncol 75(2):157–161
Setzer M, Vatter H, Vrionis FD, Marquardt G, Seifert V (2007) Management of spinal meningiomas: surgical results and a review of the literature. Neurosurg Focus 23(4):E14
Yoon SH, Chung CK, Jahng TA (2008) Surgical outcome of spinal canal meningiomas. J Korean Neurosurg Soc 42(4):300
Dodd RL, Ryu MR, Kamnerdsupaphon P, Gibbs IC, Chang SD, Adler JR (2006) CyberKnife radiosurgery for benign intradural extramedullary spinal tumors. Neurosurgery 58:674–685
Sachdev S, Soltys SG, Tupper L, Luxton G, Gibbs IC, Dodd RL, Adler JR, Choi CYH, Chang SD (2011) Stereotactic radiosurgery yields long-term control for benign intradural, extramedullary spinal tumors. Neurosurgery 69(3):533–539
Barresi V, Caffo M, Tuccari G (2016) Classification of human meningiomas: lights, shadows, and future perspectives. J Neurosci Res 94(12):1604–1612
Mirimanoff RO, Dosoretz DE, Linggood RM, Ojemann RG, Martuza RL (1985) Meningioma: analysis of recurrence and progression following neurosurgical resection. J Neurosurg 62:18–24
Kim CH, Eoh W, Choi Y et al (2015) Long-term recurrence rates after the removal of spinal meningiomas in relation to Simpson grades. Eur Spine J 25(12):4025–4032
King AT, Sharr MM, Gullan RW, Bartlett JR (1998) Spinal meningiomas: a 20-year review, Br J Neurosurg 12(6):521–526
Funding
Our study was supported by the following grants: Q28/LF1, MO IP 1012, and NV19-04-00272.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Additional information
Comments
Voldrich et al. present an interesting case series of 84 patients who were diagnosed with an intraspinal meningioma and subsequently resected according to Simpson grade II. The patients were collected over the last two decades from 1998 to 2018 and all treated at one institution. Twenty years is a long inclusion period, but 13 surgeons for 84 cases in a single center may suggest that intraspinal surgery should be kept on fewer hands in the future. During a mean follow-up of 32 months, 4 patients were diagnosed with a recurrence. Only 2 patients had a second surgery, but these patients had a significantly longer follow-up of more than 96 months.
Interestingly, 24% of all the enrolled patients experienced a prolonged hospital stay due to various minor and major complications. Twelve patients out of 19 patients with prolonged postoperative stay in hospital underwent revision surgery. This proportion represents 14% of all enrolled patients in the study, which is a rather high revision rate. The majority of patients had an improved or similar MMS score post-op, and only 2 patients experienced worsening in symptoms after treatment according to the MMS score. Around 840 MRIs must be performed according to the proposed follow-up program to find all reported recurrences in the study. This means 420 MRIs per surgery for a tumor recurrence. A more pragmatic approach could be patient education that emphasizes on sensibility disturbances, gait difficulties, or problems with balance. After all, this is a slow-growing benign disease.
Case series like this are not only valuable for the daily practice of a neurosurgeon but also for the planning of future prospective studies and follow-up programs in outpatient clinics. In addition, the authors provide a nice summarized overview over similar published studies.
Jesper Kelsen
Denmark
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Tumor - Meningioma
Rights and permissions
About this article
Cite this article
Voldřich, R., Netuka, D. & Beneš, V. Spinal meningiomas: is Simpson grade II resection radical enough?. Acta Neurochir 162, 1401–1408 (2020). https://doi.org/10.1007/s00701-020-04280-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-020-04280-2