Abstract
Background
High-sensitivity C-reactive protein (hs-CRP) is a well-recognized biomarker of neurologic complications and clinical outcome of stroke patients. However, whether hs-CRP can predict the occurrence of acute kidney injury (AKI) in aneurysmal subarachnoid hemorrhage (aSAH) patients is still unclear. The objective of this study was to assess the feasibility of using serum hs-CRP level to predict the occurrence of AKI in aSAH patients.
Methods
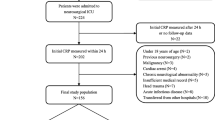
One hundred sixty-four aSAH patients were enrolled into a prospective observational study. AKI was diagnosed using the modified Kidney Disease Improving Global Outcomes (KDIGO) standard. The relationship between serum hs-CRP level at admission and occurrence of AKI was analyzed.
Results
AKI occurred in 17 patients (10.4%) in this cohort. Patients with AKI had significantly higher hs-CRP levels than those without. The mortality of the AKI group tends to be higher than that of the non-AKI group, but the difference was not statistically significant (4/17 (23.5%) vs. 13/147 (8.8%), P = 0.081). After adjusting for possible confounding factors including World Federation of Neurosurgical Societies grade, diabetes, and serum creatinine, multivariate analysis revealed that serum hs-CRP level and antibiotic therapy were both significant factors independently associated with AKI following aSAH (serum hs-CRP: OR = 1.2, 95% confidence interval (CI) = 1.1–1.3, P = 0.003; antibiotic therapy: OR = 5.8, 95%CI = 1.6–20.7, P = 0.007). Receiver operating characteristic curve analysis showed that hs-CRP had a sensitivity of 76.5% and a specificity of 64.6% for predicting the development of AKI on the basis of the best thresholds. The post hoc log-rank test revealed that patients having serum hs-CRP level > 6.6 mg/L had a significantly higher AKI rate than patients having serum hs-CRP level ≤ 6.6 mg/L (P = 0.001).
Conclusions
Serum hs-CRP level might be helpful as a predictor for the development of AKI in aSAH patients. Delayed cerebral ischemia occurrence rate and mortality of patients with AKI tend to be higher than those of patients without in this cohort; however, they were not significantly different.


Similar content being viewed by others
Change history
19 February 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00701-021-04734-1
Abbreviations
- hs-CRP:
-
High-sensitivity C-reactive protein
- AKI:
-
Acute kidney injury
- aSAH:
-
Aneurysmal subarachnoid hemorrhage
- CTA:
-
Computed tomography angiography
- DSA:
-
Digital subtraction angiography
- WFNS:
-
World Federation of Neurosurgical Societies
- MRA:
-
Magnetic resonance angiography
- CSF:
-
Cerebrospinal fluid
- KDIGO:
-
Kidney Disease Improving Global Outcomes
- ROC:
-
Receiver operating characteristic
- AUC:
-
Area under the curve
References
Ahmad A, Dempsey SK, Daneva Z, Azam M, Li N, Li P-L, Ritter JK et al (2018) Role of nitric oxide in the cardiovascular and renal systems. Int J Mol Sci 19:E2605
Al-Mufti F, Amuluru K, Damodara N, El-Ghanem M, Nuoman R et al (2018) Novel management strategies for medically-refractory vasospasm following aneurysmal subarachnoid hemorrhage. J Neurol Sci 390:44–51
Al-Mufti F, Misiolek KA, Roh D, Alawi A, Bauerschmidt A, Park S et al (2019) White blood cell count improves prediction of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. Neurosurgery 84:397–403
Badjatia N, Cremers S, Claassen J, Connolly ES, Mayer SA, Karmally W, Seres D (2018) Serum glutamine and hospital-acquired infections after aneurysmal subarachnoid hemorrhage. Neurology 91:e421–e426
Bercker S, Winkelmann T, Busch T, Laudi S, Lindner D et al (2018) Hydroxyethyl starch for volume expansion after subarachnoid haemorrhage and renal function: results of a retrospective analysis. PLoS One 13:e0192832
Chang CZ, Wu SC, Kwan AL, Hwang SL, Howng SL (2011) Magnesium lithospermate B alleviates the production of endothelin-1 through an NO-dependent mechanism and reduces experimental vasospasm in rats. Acta Neurochir 153:2211–2217
Chavakula V, Gross BA, Frerichs KU, Du R (2013) Contrast-induced nephropathy in patients with aneurysmal subarachnoid hemorrhage. Neurocrit Care 19:157–160
Chen S, Li Q, Wu H, Krafft PR, Wang Z, Zhang JH (2014) The harmful effects of subarachnoid hemorrhage on extracerebral organs. Biomed Res Int 2014:858496
Chen K, Wu Y, Wang Q, Wang J, Li X, Zhao Z, Zhou J (2015) The methodology and pharmacokinetics study of intraventricular administration of vancomycin in patients with intracranial infections after craniotomy. J Crit Care 30(218):e211–e215
Connolly ES, Rabinstein AA, Carhuapoma JR, Derdeyn CP, Dion J et al (2012) Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 43:1711–1737
Csajbok LZ, Nylen K, Ost M, Sonander H, Nellgard B (2015) In-hospital C-reactive protein predicts outcome after aneurysmal subarachnoid haemorrhage treated by endovascular coiling. Acta Anaesthesiol Scand 59:255–264
Dhar R, Diringer MN (2008) The burden of the systemic inflammatory response predicts vasospasm and outcome after subarachnoid hemorrhage. Neurocrit Care 8:404–412
Diringer MN, Bleck TP, Claude Hemphill J, Menon D, Shutter L et al (2011) Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. J Neurocrit Care 15:211–240
Drenckhahn C, Brabetz C, Major S, Wiesenthal D, Woitzik J, Dreier JP et al (2013) Criteria for the diagnosis of noninfectious and infectious complications after aneurysmal subarachnoid hemorrhage in DISCHARGE-1. Acta Neurochir Suppl 115:153–159
Flechet M, Guiza F, Schetz M, Wouters P, Vanhorebeek I et al (2017) AKIpredictor, an online prognostic calculator for acute kidney injury in adult critically ill patients: development, validation and comparison to serum neutrophil gelatinase-associated lipocalin. Intensive Care Med 43:764–773
Frijns CJ, Fijnheer R, Algra A, van Mourik JA, van Gijn J, Rinkel GJ (2006) Early circulating levels of endothelial cell activation markers in aneurysmal subarachnoid haemorrhage: associations with cerebral ischaemic events and outcome. J Neurol Neurosurg Psychiatry 77:77–83
Frontera JA, Fernandez A, Schmidt JM, Claassen J, Wartenberg KE et al (2009) Defining vasospasm after subarachnoid hemorrhage: what is the most clinically relevant definition? Stroke 40:1963–1968
Hall A, O'Kane R (2018) The extracranial consequences of subarachnoid hemorrhage. World Neurosurg 109:381–392
Hauer EM, Stark D, Staykov D, Steigleder T, Schwab S, Bardutzky J (2011) Early continuous hypertonic saline infusion in patients with severe cerebrovascular disease. Crit Care Med 39:1766–1772
Jeon YT, Lee JH, Lee H, Lee HK, Hwang JW, Lim YJ, Park HP et al (2012) The postoperative C-reactive protein level can be a useful prognostic factor for poor outcome and symptomatic vasospasm in patients with aneurysmal subarachnoid hemorrhage. J Neurosurg Anesthesiol 24:317–324
Kumar AB, Shi Y, Shotwell MS, Richards J, Ehrenfeld JM (2015) Hypernatremia is a significant risk factor for acute kidney injury after subarachnoid hemorrhage: a retrospective analysis. Neurocrit Care 22:184–191
Lazaridis C, Naval N (2010) Risk factors and medical management of vasospasm after subarachnoid hemorrhage. Neurosurg Clin N Am 21:353–364
Lee HG, Kim WK, Yeon JY, Kim JS, Kim KH, Jeon P, Hong SC (2018) Contrast-induced acute kidney injury after coil embolization for aneurysmal subarachnoid hemorrhage. Yonsei Med J 59:107–112
Luther MK, Timbrook TT, Caffrey AR, Dosa D, Lodise TP, LaPlante KL (2018) Vancomycin plus piperacillin-tazobactam and acute kidney injury in adults: a systematic review and meta-analysis. Crit Care Med 46:12–20
Mackey J, Khoury JC, Alwell K, Moomaw CJ, Kissela BM, Flaherty ML et al (2016) Stable incidence but declining case-fatality rates of subarachnoid hemorrhage in a population. Neurology 87:2192–2197
McWilliam SJ, Antoine DJ, Jorgensen AL, Smyth RL, Pirmohamed M (2018) Urinary biomarkers of aminoglycoside-induced nephrotoxicity in cystic fibrosis: kidney injury molecule-1 and neutrophil gelatinase-associated lipocalin. Sci Rep 8:5094
Mizuno T, Sato W, Ishikawa K, Shinjo H, Miyagawa Y, Noda Y et al (2012) KDIGO (Kidney Disease: Improving Global Outcomes) criteria could be a useful outcome predictor of cisplatin-induced acute kidney injury. Oncology 82:354–359
Mukerji SS, Buchbinder BR, Singhal AB (2015) Reversible cerebral vasoconstriction syndrome with reversible renal artery stenosis. Neurology 85:201–202
Prowle JR (2015) Measurement of AKI biomarkers in the ICU: still striving for appropriate clinical indications. Intensive Care Med 41:541–543
Rampoldi B, Tessarolo S, Giubbilini P, Gaia P, Corino SD et al (2018) Neutrophil gelatinase-associated lipocalin and acute kidney injury in endovascular aneurysm repair or open aortic repair: a pilot study. Biochem Med (Zagreb) 28:010904
Rennie TJW, De Souza N, Donnan PT et al (2018) Risk of acute kidney injury following community prescription of antibiotics: self-controlled case series. Nephrol Dial Transplant. https://doi.org/10.1093/ndt/gfy187
Romero FR, Cataneo DC, Cataneo AJ (2014) C-reactive protein and vasospasm after aneurysmal subarachnoid hemorrhage. Acta Cir Bras 29:340–345
Rumalla K, Mittal MK (2016) Acute renal failure in aneurysmal subarachnoid hemorrhage: nationwide analysis of hospitalizations in the United States. World Neurosurg 91:542–547
Salem S, Jankowski V, Asare Y, Liehn E, Welker P, Raya-Bermudez A et al (2015) Identification of the vasoconstriction-inhibiting factor (VIF), a potent endogenous cofactor of angiotensin II acting on the angiotensin II type 2 receptor. Circulation 131:1426–1434
Sam R, Hart P, Haghighat R, Ing TS (2012) Hypervolemic hypernatremia in patients recovering from acute kidney injury in the intensive care unit. Clin Exp Nephrol 16:136–146
Sawada A, Kawanishi K, Morikawa SA, Davey P, Dreischulte T, Bell S (2018) Biopsy-proven vancomycin-induced acute kidney injury: a case report and literature review. BMC Nephrol 19:72
Schuiling WJ, Dennesen PJ, Rinkel GJ (2005) Extracerebral organ dysfunction in the acute stage after aneurysmal subarachnoid hemorrhage. Neurocrit Care 3:1–10
Shah SR, Tunio SA, Arshad MH, Moazzam Z, Noorani K, Feroze AM et al (2015) Acute kidney injury recognition and management: a review of the literature and current evidence. Glob J Health Sci 8:120–124
Shen J, Karki M, Jiang T, Zhao B (2018) Complications associated with diagnostic cerebral angiography: a retrospective analysis of 644 consecutive cerebral angiographic cases. Neurol India 66:1154–1158
Srinivasan A, Aggarwal A, Gaudihalli S, Mohanty M, Dhandapani M, Singh H et al (2016) Impact of early leukocytosis and elevated high-sensitivity C-reactive protein on delayed cerebral ischemia and neurologic outcome after subarachnoid hemorrhage. World Neurosurg 90:91–95
Tam AK, Ilodigwe D, Mocco J, Mayer S, Kassell N, Ruefenacht D, Schmiedek P et al (2010) Impact of systemic inflammatory response syndrome on vasospasm, cerebral infarction, and outcome after subarachnoid hemorrhage: exploratory analysis of CONSCIOUS-1 database. Neurocrit Care 13:182–189
Tujjar O, Belloni I, Hougardy JM, Scolletta S, Vincent JL, Creteur J et al (2017) Acute kidney injury after subarachnoid hemorrhage. J Neurosurg Anesthesiol 29:140–149
Zacharia BE, Ducruet AF, Hickman ZL, Grobelny B, Fernandez L et al (2009) Renal dysfunction as an independent predictor of outcome after aneurysmal subarachnoid hemorrhage: a single-center cohort study. Stroke 40:2375–2381
Acknowledgments
We are grateful to all colleagues who participated in providing cases (Dr. Jiawei Cai and Dr. Fuxin Lin).
Funding
This study was supported by grants from the Key Clinical Specialty Discipline Construction Program of Fujian, P.R.C., and the major project of Fujian Provincial Department of Science and Technology (No. 2014YZ0003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
De-Zhi Kang received grants from the Key Clinical Specialty Discipline Construction Program of Fujian, P.R.C., and the major project of Fujian Provincial Department of Science and Technology (No. 2014YZ0003). All other authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All procedures in the study were approved by the Ethics Committee of the First Affiliated Hospital of Fujian Medical University (Fujian, China).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Comments
This paper reports an association between admission plasma levels of high-sensitivity C-reactive protein and development within 14 days of acute kidney injury in patients with aneurysmal subarachnoid haemorrhage. This is an interesting observation, which warrants consideration regarding the mechanism of association. However, as a clinician I probably would not add this relatively expensive test to my prognostic toolbox at this time.
Kirsten Moeller
Copenhagen, Denmark
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Vascular Neurosurgery - Aneurysm
Rights and permissions
About this article
Cite this article
Yang, BH., He, Q., Ding, CY. et al. High-sensitivity C-reactive protein as a predictive factor of acute kidney injury following aneurysmal subarachnoid hemorrhage: a prospective observational study. Acta Neurochir 161, 1783–1791 (2019). https://doi.org/10.1007/s00701-019-04006-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-019-04006-z