Abstract
Background
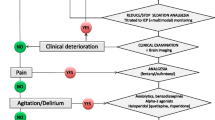
The Wessex Modified Richmond Sedation Scale (WMRSS) has been developed with the aim of improving the early identification of patients requiring decompressive hemicraniectomy for malignant middle cerebral artery syndrome (MMS). The objective of this study was to evaluate the WMRSS against the Glasgow Coma Scale (GCS).
Methods
A retrospective study was conducted of patients admitted to our unit for observation of MMS. Data were obtained on WMRSS and GCS recordings from admission up to 120-h post-ictus. Patients’ meeting inclusion criteria were recommended for theatre based on subsequent deteriorations in consciousness on either WMRSS or GCS from a 6-h post-stroke baseline, after ruling out non-neurological causes.
Results
Approximately, 60% of those eligible for monitoring were not recommended for theatre, and none died; however, these patients continued to demonstrate some variability in recorded conscious level. Patients requiring surgical intervention showed earlier drops in WMRSS compared to GCS. Neither the GCS nor the WMRSS on admission predicted the subsequent need for decompressive surgery. There was no increase in mortality with the introduction of WMRSS.
Conclusions
WMRSS adds value to monitoring MMS by indicating need for surgery prior to GCS. Early reduction in consciousness may not be sufficient for proceeding to surgical intervention, but subsequent reduction in consciousness may be a more appropriate criterion for surgery.

Similar content being viewed by others
References
Beck C, Kruetzelmann A, Forkert ND et al (2014) A simple brain atrophy measure improves the prediction of malignant middle cerebral artery infarction by acute DWI lesion volume. J Neurol 261(6):1097–1103
Ely EW, Truman B, Shintani A et al (2003) Monitoring sedation status over time in ICU patients: reliability and validity of the Richmond Agitation-Sedation Scale (RASS). JAMA 289(22):2983–2991
Foerch C, Otto B, Singer OC, Neumann-Haefelin T, Yan B, Berkefeld J, Steinmetz H, Sitzer M (2004) Serum S100B predicts a malignant course of infarction in patients with acute middle cerebral artery occlusion. Stroke 35(9):2160–2164
Frank JI (1995) Large hemispheric infarction, deterioration, and intracranial pressure. Neurology 45(7):1286–1290
Hacke W, Schwab S, Horn M, Spranger M, De Georgia M, von Kummer R (1996) “Malignant” middle cerebral artery territory infarction: clinical course and prognostic signs. Arch Neurol 53(4):309–315
Hanley JA, McNeil BJ (1983) A method of comparing the areas under receiver operating characteristic curves derived from the same cases. Radiology 148(3):839–843
Heinsius T, Bogousslavsky J, Van Melle G (1998) Large infarcts in the middle cerebral artery territory. Etiology and outcome patterns. Neurology 50(2):341–350
Hofmeijer J, Kappelle LJ, Algra A, Amelink GJ, van Gijn J, van der Worp HB, investigators HAMLET (2009) Surgical decompression for space-occupying cerebral infarction (the Hemicraniectomy After Middle Cerebral Artery infarction with Life-threatening Edema Trial [HAMLET]): a multicentre, open, randomised trial. Lancet Neurol 8(4):326–333
Jüttler E, Schwab S, Schmiedek P, Unterberg A, Hennerici M, Woitzik J, Witte S, Jenetzky E, Hacke W, DESTINY Study Group (2007) Decompressive Surgery for the Treatment of Malignant Infarction of the Middle Cerebral Artery (DESTINY): a randomized, controlled trial. Stroke 38(9):2518–2525
Kamran S, Akhtar N, Salam A, Alboudi A, Kamran K, Ahmed A, Khan RA, Mirza MK, Inshasi J, Shuaib A (2017) Revisiting hemicraniectomy: late decompressive hemicraniectomy for malignant middle cerebral artery stroke and the role of infarct growth rate. Stroke Res Treat 2017:2507834
Lammy S, Al-Romhain B, Osborne L, St George EJ (2016) 10-year institutional retrospective case series of decompressive craniectomy for malignant middle cerebral artery infarction (mMCAI). World Neurosurg 96:383–389
Maciel CB, Sheth KN (2015) Malignant MCA stroke: an update on surgical decompression and future directions. Curr Atheroscler Rep 17(7):40
Oppenheim C, Samson Y, Manaï R et al (2000) Prediction of malignant middle cerebral artery infarction by diffusion-weighted imaging. Stroke 31(9):2175–2181
Poca MA, Benejam B, Sahuquillo J, Riveiro M, Frascheri L, Merino MA, Delgado P, Alvarez-Sabin J (2010) Monitoring intracranial pressure in patients with malignant middle cerebral artery infarction: is it useful? J Neurosurg 112(3):648–657
Ronchetti G, Panciani PP, Stefini R, Spena G, Fontanella MM (2014) Acute supratentorial ischemic stroke: when surgery is mandatory. Biomed Res Int 2014:624126
Vahedi K, Vicaut E, Mateo J et al (2007) Sequential-design, multicenter, randomized, controlled trial of early decompressive craniectomy in malignant middle cerebral artery infarction (DECIMAL Trial). Stroke 38(9):2506–2517
Varndell W, Elliott D, Fry M (2015) The validity, reliability, responsiveness and applicability of observation sedation-scoring instruments for use with adult patients in the emergency department: a systematic literature review. Australas Emerg Nurs J 18(1):1–23
Wartenberg KE (2012) Malignant middle cerebral artery infarction. Curr Opin Crit Care 18(2):152–163
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Approval for the study was obtained from Southampton University Hospitals NHS Trust Clinical Effectiveness Committee (Ref: ZUAD3316) and University of Southampton Ethics and Research Governance Committee (Ref: 14786). For this type of study formal consent is not required.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Comments:
Grading scales are useful tools to harmonize patient care across different medical centers in different countries and to facilitate intelligent and reproducible scientific study of stroke outcomes. The clinical community ultimately adjudicates which grading scales come into common use. The universal adoption of the GCS is one example of this. NIHSS for decision-making in endovascular interventions is another. mRS for stroke outcomes is a third.
This will become an increasing area of interest as the potential for endovascular salvage grows. For example, just last week in the USA, the FDA approved the on-label use of one stent-retriever device, previously limited to 6 h, now up to 24 h after stroke onset, based on the clinical results from the DAWN trial.
In the present report the authors elaborate on their retrospective study of the novel WMRSS scale as a potentially more sensitive predictor than GCS, to identify which MMS patients should proceed to decompressive craniectomy, and also to perhaps decrease the necessity of frequent HCT evaluations. It is a worthwhile effort. We await the judgment of the surgical community regarding the adoption of this method or other similar paradigms.
Christopher Miranda Loftus
Philadelphia, USA
Conference Presentations
Myers M, et al. A comparison of the Wessex modified Richmond Agitation and Sedation Score and Glasgow Coma Scale for monitoring patients at risk of malignant Middle Cerebral Artery syndrome. Br J Neurosurg 2016; 30 (2):130-186; FM2-3.
Hayat TTA et al. Introduction of the Wessex modified Richmond Agitation and Sedation Scale for monitoring patients at risk of malignant MCA syndrome. Br J Neurosurg, 2015; Abstract No: 1140.
Rights and permissions
About this article
Cite this article
Hayat, T.T.A., Myers, M.A., Hell, J. et al. The Wessex modified Richmond Sedation Scale as a novel tool for monitoring patients at risk of malignant MCA syndrome. Acta Neurochir 160, 1115–1119 (2018). https://doi.org/10.1007/s00701-018-3531-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-018-3531-y