Abstract
Background
The aim of this retrospective study was to investigate the long-term seizure control and antiepileptic drug (AED) prescriptions, as well as identifying predictors of seizure(s) before and after surgery in a population-based cohort of operated intracranial meningioma patients.
Methods
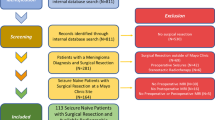
A total of 113 consecutive adult (> 18 years old) patients with newly diagnosed meningioma operated at the Karolinska University Hospital between 2006 and 2008 were included and followed up until the end of 2015. Data on seizure activity and AED prescriptions were obtained through chart review and telephone interview. Logistic regression and survival analysis were applied to identify risk factors for pre- and postoperative seizures.
Results
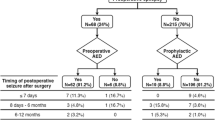
A total of 21/113 (18.6%) patients experienced seizures before surgery of which 8/21 (38.1%) went on to become seizure-free after surgery. Thirteen (14%) patients experienced new-onset seizures after surgery. The regression analysis revealed tumor diameter ≥ 3.5 cm as a risk factor for preoperative seizures (OR 3.83, 95% CI 1.14–12.87). Presence of headache (OR 0.19, 95% CI 0.05–0.76) and skull base tumor location (OR 0.14, 95% CI 0.04–0.44) decreased the risk of preoperative seizures. Postoperative seizures were associated with tumor diameter ≥ 3.5 cm (OR 2.65, 95% CI 1.06–6.62) and history of preoperative seizures (OR 3.50, 95% CI 1.55–7.90).
Conclusion
Seizures are common before and after intracranial meningioma surgery. Approximately one third of patients with preoperative seizures become seizure-free on long-term follow-up after surgery, while 14% experienced new-onset seizures after surgery. Larger tumor size, absence of headache, and non-skull base location were associated with preoperative seizures, while tumor size and preoperative seizures were associated with postoperative seizures.


Similar content being viewed by others
References
Bauer R, Ortler M, Seiz-Rosenhagen M, Maier R, Anton JV, Unterberger I (2014) Treatment of epileptic seizures in brain tumors: a critical review. Neurosurg Rev 37(3):381–388
Beghi E, Carpio A, Forsgren L, Hesdorffer DC, Malmgren K, Sander JW, Tomson T, Hauser WA (2010) Recommendation for a definition of acute symptomatic seizure. Epilepsia 51(4):671–675
Chaichana KL, Pendleton C, Zaidi H, Olivi A, Weingart JD, Gallia GL, Lim M, Brem H, Quiñones-Hinojosa A (2013) Seizure control for patients undergoing meningioma surgery. World Neurosurg 79(3–4):515–524
Chang EF, Potts MB, Keles GE, Lamborn KR, Chang SM, Barbaro NM, Berger MS (2008) Seizure characteristics and control following resection in 332 patients with low-grade gliomas. J Neurosurg 108:227–235
Chozick BS, Reinert SE, Greenblatt SH (1996) Incidence of seizures after surgery for supratentorial meningiomas: a modern analysis. J Neurosurg 84(3):382–386
De Santis A, Villani R, Sinisi M, Stocchetti N, Perucca E (2002) Add-on phenytoin fails to prevent early seizures after surgery for supratentorial brain tumors: a randomized controlled study. Epilepsia 43(2):175–182
Englot DJ, Magill ST, Han SJ, Chang EF, Berger MS, McDermott MW (2016) Seizures in supratentorial meningioma: a systematic review and meta-analysis. J Neurosurg 124(6):1552–1561
Ertürk Çetin Ö, İşler C, Uzan M, Özkara Ç (2017) Epilepsy-related brain tumors. Seizure 44:93–97
Fathi AR, Roelcke U (2013) Meningioma. Curr Neurol Neurosci Rep 13(4):337
Fisher RS, Acevedo C, Arzimanoglou A, Bogacz A, Cross JH, Elger CE, Engel J Jr, Forsgren L, French JA, Glynn M, Hesdorffer DC, Lee BI, Mathern GW, Moshé SL, Perucca E, Scheffer IE, Tomson T, Watanabe M, Wiebe S (2014) ILAE official report: a practical clinical definition of epilepsy. Epilepsia 55(4):475–482
Gazzeri R, Galarza M, Gazzeri G (2008) Giant olfactory groove meningioma: ophthalmological and cognitive outcome after bifrontal microsurgical approach. Acta Neurochir 150:1117–1126
Hamasaki T, Yamada K, Kuratsu J (2013) Seizures as a presenting symptom in neurosurgical patients: a retrospective single-institution analysis. Clin Neurol Neurosurg 115(11):2336–2340
Hamasaki T, Yamada K, Yano S, Nakamura H, Makino K, Hide T, Hasegawa Y, Kuroda J, Hirai T, Kuratsu J (2012) Higher incidence of epilepsy in meningiomas located on the premotor cortex: a voxel-wise statistical analysis. Acta Neurochir 154(12):2241–2249
LieuAS HSL (2000) Intracranial meningiomas and epilepsy: incidence, prognosis and influencing factors. Epilepsy Res 38(1):45–52
Kerrigan S, Grant R (2011) Antiepileptic drugs for treating seizures in adults with brain tumours. Cochrane Database Syst Rev 8:CD008586
Komotar RJ, Raper DM, Starke RM, Iorgulescu JB, Gutin PH (2011) Prophylactic antiepileptic drug therapy in patients undergoing supratentorial meningioma resection: a systematic analysis of efficacy. J Neurosurg 115(3):483–490
Musluman AM, Yilmaz ARTC, Cavusoglu H, Kahyaoglu O, Aydin Y (2012) Unilateral frontal interhemispheric transfalcial approaches for the removal of olfactory groove meningiomas. Turk Neurosurg 22:174–182
Ostrom QT, Gittleman H, Liao P, Rouse C, Chen Y, Dowling J, Wolinsky Y, Kruchko C, Barnholtz-Sloan J (2014) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro-Oncology 16(Suppl 4):1–63
Skardelly M, Rother C, Noell S, Behling F, Wuttke TV, Schittenhelm J, Bisdas S, Meisner C, Rona S, Tabatabai G, Roser F, Tatagiba MS (2017) Rsk factors of preoperative and early postoperative seizures in patients with meningioma: a retrospective single-center cohort study. World Neurosurg 97:538–546
Sughrue ME, Rutkowski MJ, Chang EF, Shangari G, Kane AJ, McDermott MW, Berger MS, Parsa AT (2011) Postoperative seizures following the resection of convexity meningiomas: are prophylactic anticonvulsants indicated? Clinical article. J Neurosurg 114(3):705–709
Tanti MJ, Marson AG, Chavredakis E, Jenkinson MD (2016) The impact of epilepsy on the quality of life of patients with meningioma: a systematic review. Br J Neurosurg 30(1):23–28
van Breemen MS, Wilms EB, Vecht CJ (2007) Epilepsy in patients with brain tumours: epidemiology, mechanisms, and management. Lancet Neurol 6(5):421–430
van Breemen MS, Wilms EB, Vecht CJ (2012) Seizure control in brain tumors. Handb Clin Neurol 104:381–389
Wirsching HG, Morel C, Gmür C, Neidert MC, Baumann CR, Valavanis A, Rushing EJ, Krayenbühl N, Weller M (2016) Predicting outcome of epilepsy after meningioma resection. Neuro-Oncology 18(7):1002–1010
Xue H, Sveinsson O, Tomson T, Mathiesen T (2015) Intracranial meningiomas and seizures: a review of the literature. Acta Neurochir 157(9):1541–1548
Zhang B, Zhao G, Yang HF, Wang D, Yu JL, Huang HY (2011) Assessment of risk factors for early seizures following surgery for meningiomas using logistic regression analysis. J Int Med Res 39(5):1728–1735
Zheng Z, Chen P, Fu W, Zhu J, Zhang H, Shi J, Zhang J (2013) Early and late postoperative seizure outcome in 97 patients with supratentorial meningioma and preoperative seizures: a retrospective study. J Neuro-Oncol 114(1):101–109
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the ethics committee of Karolinska Institutet.
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Xue, H., Sveinsson, O., Bartek, J. et al. Long-term control and predictors of seizures in intracranial meningioma surgery: a population-based study. Acta Neurochir 160, 589–596 (2018). https://doi.org/10.1007/s00701-017-3434-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-017-3434-3