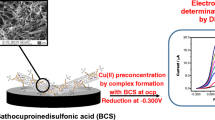
Abstract
The authors describe a novel electrochemical determination method for bisphenol A (BPA) based on the electrosynthesised Cu-BTC (H3BTC: trimesic acid) films. Using H3BTC as the ligand, Cu(NO3)2 as the precursor of copper ions, and triethylamine hydrochloride (Et3NHCl) as the probase source, Cu-BTC films were directly deposited on glassy carbon electrode (GCE) surface via cathodic electrochemical reduction under −1.30 V. Considering the electrocatalytic activity of metal center Cu2+, Cu-BTC films were applied to construct the electrochemical determination platform for BPA. Chronocoulometry and electrochemical impedance spectroscopy were used to study the signal enhancement mechanism. The determination conditions were optimized. As a result, a sensitive electrochemical method was constructed for BPA. The peak currents, best measured at voltage of 0.496 V vs. SCE (KCl saturated calomel reference electrode), increase linearly in the range from 5.0 to 2000 nM. The value of determination limit is 0.72 nM. The proposed method was successfully applied to determine BPA in spiked urine, spiked waste water samples and plastic products. The results were in good agreement with those obtained for the same samples by high-performance liquid chromatography (HPLC).

Schematics for the construction of electrochemical determination for bisphenol A.





Similar content being viewed by others
References
Almeida S, Raposo A, Almeid-Gonzalez M, Carrascosa C (2018) Bisphenol A: food exposure and impact on human health. Compr Rev Food Sci Food Saf 17:1503–1517
Ahmed RG, Walaa GH, Asmaa FS (2018) Suppressive effects of neonatal bisphenol A on the neuroendocrine system. Toxicol Ind Health 34:397–407
Balistrieri A, Hobohm L, Srivastava T, Meier A, Corriden R (2018) Alterations in human neutrophil function caused by bisphenol A. Am J Physiol-Cell Physiol 315:C636–C642
Nevoral J, Kolinko Y, Moravec J, Zalmanova T, Hoskova K, Prokesova S, Klein P, Ghaibour K, Hosek P, Stiavnicka M, Rimnacova H, Tonar Z, Petr J, Milena Kralickova M (2018) Long-term exposure to very low doses of bisphenol S affects female reproduction. Reproduction 156:47–57
Hakova M, Havlikova LC, Chvojka J, Solich P, Satínsky D (2018) An on-line coupling of nanofibrous extraction with column-switching high performance liquid chromatography-a case study on the determination of bisphenol A in environmental water samples. Talanta 178:141–146
Tang Z, Kang T, Kang X (2019) Determination of five phthalate esters and bisphenol A in fruit juice by packed-nanofiber solid phase extraction coupled with gas chromatography. J Food Eng Technol 8:50–54
Cao W, Chao YJ, Liu L, Liu QL, Pei MS (2014) Flow injection chemiluminescence sensor based on magnetic oil-based surface molecularly imprinted nanoparticles for determination of bisphenol A. Sens Actuator B-Chem 204:704–709
Ren S, Li Q, Li Y, Li S, Han T, Wang J, Peng Y, Bai J, Ning B, Gao Z (2019) Upconversion fluorescent aptasensor for bisphenol A and 17β-estradiol based on a nanohybrid composed of black phosphorus and gold, and making use of signal amplification via DNA tetrahedrons. Microchim Acta 186(3):1–8
Uzek R, Sari E, Senel S, Denizli A, Merkoci A (2019) A nitrocellulose paper strip for fluorometric determination of bisphenol A using molecularly imprinted nanoparticles. Microchim Acta 186(4):1–10
Wang L, Cao H-X, Pan C-G, He Y-S, Liu H-F, Zhou L-H, Li C-Q, Liang G-X (2019) A fluorometric aptasensor for bisphenol A based on the inner filter effect of gold nanoparticles on the fluorescence of nitrogen-doped carbon dots. Microchim Acta 186(1):1–9
Quan Y, Yao J, Yang S, Chen L, Li J, Liu Y, Lang J, Shen H, Wang Y, Wang Y, Yang J, Gao M (2019) ZnO nanoparticles on MoS2 microflowers for ultrasensitive SERS detection of bisphenol A. Microchim Acta 186(8):1–8
Ren X, Cheshari EC, Qi J, Li X (2018) Silver microspheres coated with a molecularly imprinted polymer as a SERS substrate for sensitive detection of bisphenol A. Microchim Acta 185(4):1–8
Joiakim A, Kaplan D, Putt DA, Santos JM, Friedrich K, Kim SH, Kim H (2019) Bisphenol A (BPA) in liquid portions of canned foods obtained from domestic and Asian markets in the United States. Environ Dis 4:6–11
Mo FY, Xie JW, Wu TT, Liu ML, Zhang YY, Yao SZ (2019) A sensitive electrochemical sensor for bisphenol A on the basis of the AuPd incorporated carboxylic multi-walled carbon nanotubes. Food Chem 292:253–259
Vieira Jodar L, Orzari LO, Storti Ortolani T, Assumpçao MH, Vicentini FC, Janegitz BC (2019) Electrochemical sensor based on casein and carbon black for bisphenol A detection. Electroanalysis. https://doi.org/10.1002/elan.201900176
Butmee P, Tumcharern G, Saejueng P, Stankovic D, Ortner A, Jitcharoen J, Kalcher K, Samphao A (2019) A direct and sensitive electrochemical sensing platform based on ionic liquid functionalized graphene nanoplatelets for the detection of bisphenol A. J Electroanal Chem 833:370–379
Messaoud NB, Lahcen AA, Dridi C, Amine A (2018) Ultrasound assisted magnetic imprinted polymer combined sensor based on carbon black and gold nanoparticles for selective and sensitive electrochemical detection of bisphenol A. Sensor Actuat B-chem 276:304–312
Zhang J, Xu X, Chen L (2018) An ultrasensitive electrochemical bisphenol A sensor based on hierarchical Ce-metal-organic framework modified with cetyltrimethylammonium bromide. Sensor Actuat B-chem 261:425–433
Li XY, Li CL, Wu C, Wu KB (2019) Strategy for highly sensitive electrochemical sensing: in situ coupling of a metal-organic framework with ball-mill-exfoliated Graphene. Anal Chem 91:6043–6050
Qin JY, Shen J, Xu XY, Yuan Y, He GY, Chen HQ (2018) A glassy carbon electrode modified with nitrogen-doped reduced graphene oxide and melamine for ultra-sensitive voltammetric determination of bisphenol A. Microchim Acta 185:459–467
Ashraf G, Asif M, Aziz A, Wang Z, Qiu X, Huang Q, Xiao F, Liu H (2019) Nanocomposites consisting of copper and copper oxide incorporated into MoS4 nanostructures for sensitive voltammetric determination of bisphenol A. Microchim Acta 186(6):1–9
Baghayeri M, Ansari R, Nodehi M, Razavipanah I, Veisi H (2018) Voltammetric aptasensor for bisphenol A based on the use of a MWCNT/Fe3O4@gold nanocomposite. Microchim Acta 185(7):1–9
Zhou H-C, Long JR, Yaghi OM (2012) Introduction to metal-organic frameworks. Chem Rev 112:673–674
Xue DX, Wang Q, Bai J (2019) Amide-functionalized metal-organic frameworks: syntheses, structures and improved gas storage and separation properties. Coord Chem Rev 378:2–16
Wang L, Zheng M, Xie ZG (2018) Nanoscale metal-organic frameworks for drug delivery: a conventional platform with new promise. J Mater Chem B 6:707–717
Jiao L, Wang Y, Jiang HL, Xu Q (2018) Metal-organic frameworks as platforms for catalytic applications. Adv Mater 30:1703663
Liang ZB, Qu C, Guo WH, Zou RQ, Xu Q (2018) Pristine metal-organic frameworks and their composites for energy storage and conversion. Adv Mater 30:1702891
Yuan BQ, Zhang RC, Jiao XX, Li J, Shi HZ, Zhang DJ (2014) Amperometric determination of reduced glutathione with a new co-based metal-organic coordination polymer modified electrode. Electrochem Commun 40:92–95
Zhang DJ, Zhang JC, Shi HZ, Guo XL, Guo YY, Zhang RC, Yuan BQ (2015) Redox-active microsized metal-organic framework for efficient nonenzymatic H2O2 sensing. Sensor Actuat B-chem 221:224–229
Dong SY, Suo GC, Li N, Chen Z, Peng L, Fu YL, Yang Q, Huang TL (2016) A simple strategy to fabricate high sensitive 2,4-dichlorophenol electrochemical sensor based on metal organic framework Cu3(BTC)2. Sensor Actuat B-chem 222:972–979
Li W-J, Tu M, Cao R, Fischer RA (2016) Metal-organic framework thin films: electrochemical fabrication techniques and corresponding applications and perspectives. J Mater Chem A 4:12356–12369
Gurrappa I, Binder L (2008) Electrodeposition of nanostructured coatings and their characterization-a review. Sci Technol Adv Mater 9:043001
Joaristi AM, Juan-Alcaniz J, Serra-Crespo P, Kapteijn F, Gascon J (2012) Electrochemical synthesis of some archetypical Zn2+, Cu2+, and Al3+ metal organic frameworks. Cryst Growth Des 12:3489–3498
Li MY, Dinca M (2011) Reductive electrosynthesis of crystalline metal-organic frameworks. J Am Chem Soc 133:12926–12929
Li MY, Dinca M (2014) Selective formation of biphasic thin films of metal-organic frameworks by potential-controlled cathodic electrodeposition. Chem Sci 5:107–111
Alizadeh S, Nematollahi D (2017) Electrochemically assisted self-assembly technique for the fabrication of mesoporous metal-organic framework thin films: composition of 3D hexagonally packed crystals with 2D honeycomb-like mesopores. J Am Chem Soc 139:4753–4761
Wang X, Lu XB, Wu LD, Chen JP (2015) 3D metal-organic framework as highly efficient biosensing platform for ultrasensitive and rapid detection of bisphenol A. Biosens Bioelectron 65:295–301
Liu LT, Zhou YL, Liu S, Xu MT (2018) The applications of metal-organic frameworks in electrochemical sensors. ChemElectroChem 5(1):6–19
Ji LD, Cheng Q, Wu KB, Yang XF (2016) Cu-BTC frameworks-based electrochemical sensing platform for rapid and simple determination of sunset yellow and Tartrazine. Sensor Actuat B-chem 231:12–17
Ji LD, Wang J, Wu KB, Yang NJ (2018) Tunable electrochemistry of Electrosynthesized copper metal-organic frameworks. Adv Funct Mater 28(2018):1706961
Bard AJ, Faulkner LR (1980) Electrochemical methods: fundamentals and applications, 2nd edn. Wiley, New York
Wu C, Cheng Q, Wu KB (2015) Electrochemical functionalization of N-methyl-2-pyrrolidone-exfoliated graphene nanosheets as highly sensitive analytical platform for phenols. Anal Chem 87(6):3294–3299
Herr T, Fischer P, Tübke J, Pinkwart K, Elsneret P (2014) Increasing the energy density of the non-aqueous vanadium redox flow battery with the acetonitrile-1, 3-dioxolane-dimethyl sulfoxide solvent mixture. J Power Sources 265:317–324
Kurzweil P, Fischle HJ (2004) A new monitoring method for electrochemical aggregates by impedance spectroscopy. J Power Sources 127(1–2):331–340
Zhang YY, Yan P, Wan QJ, Wu KB, Yang NJ (2016) Morphology-dependent electrochemistry of FeOOH nanostructures. Electrochem Commun 68:10–14
Acknowledgements
This work was supported by the Natural Science Foundation of Hubei Province (No. 2019CFB248) and the Scientific Research Project of Education Department of Hubei Province (Nos. Q20192808, Q20192809).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 609 kb)
Rights and permissions
About this article
Cite this article
Hu, P., Zhu, X., Luo, X. et al. Cathodic electrodeposited Cu-BTC MOFs assembled from Cu(II) and trimesic acid for electrochemical determination of bisphenol A. Microchim Acta 187, 145 (2020). https://doi.org/10.1007/s00604-020-4124-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-020-4124-z