Abstract
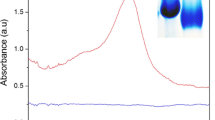
The peroxidase-like activity of hollow Prussian Blue nanocubes (hPBNCs) is used, in combination with the enzyme alcohol oxidase (AOx), in a colorimetric ethanol assay. Different from other nanozymes, the large cavity structure of the hPBNCs provides a larger surface and more binding sites for AOx to be bound on their surface or in the pores. This extremely enhances the sensitivity of the assay system. In the presence of ethanol, AOx is capable of catalyzing the oxidation of alcohols to aldehydes, accompanied by the generation of hydrogen peroxide (H2O2). The hPBNCs act as peroxidase mimics and then can catalyze the oxidation of 3,3′5,5′-tetramethylbenzidine (TMB) by H2O2, resulting in a color change of the solution from colorless to blue with a strong absorption at 652 nm. The lower detection limit for ethanol is 1.41 μg∙mL−1. Due to the high catalytic activity of hPBNCs in weakly acidic and neutral solutions, the system was successfully applied to the determination of ethanol in mice blood. This is critically important for studying the alcohol consumption and monitoring the ethanol toxicokinetics.

Schematic representation of hollow Prussian Blue nanocubes (hPBNCs) used as both a peroxidase mimetic and as a carrier for alcohol oxidase. Utilizing hPBNCs along with the ethanol conversion enzyme, a sensitive colorimetric assay for ethanol was developed and applied to blood samples with satisfactory results.






Similar content being viewed by others
References
Voas RB, Fell JC (2010) Preventing alcohol-related problems through health policy research. Alcohol Res Health 33:18–28
Chen X, Cai F, Guo S, Ding F, He Y, Wu J, Liu C (2014) Protective effect of Flos puerariae extract following acute alcohol intoxication in mice. Alcohol Clin Exp Res 38:1839–1846
Liu Y, Du J, Yan M, Lau MY, Hu J, Han H, Yang OO, Liang S, Wei W, Wang H, Li J, Zhu X, Shi L, Chen W, Ji C, Lu Y (2013) Biomimetic enzyme nanocomplexes and their use as antidotes and preventive measures for alcohol intoxication. Nat Nanotechnol 8:187–192
Rocchitta G, Secchi O, Alvau MD, Migheli R, Calia G, Bazzu G, Farina D, Desole MS, O'Neill RD, Serra PA (2012) Development and characterization of an implantable biosensor for telemetric monitoring of ethanol in the brain of freely moving rats. Anal Chem 84:7072–7079
Kim J, Jeerapan I, Imani S, Cho TN, Bandodkar A, Cinti S, Mercier PP, Wang J (2016) Noninvasive alcohol monitoring using a wearable tattoo-based iontophoretic-biosensing system. ACS Sens 1:1011–1019
Selvam AP, Muthukumar S, Kamakoti V, Prasad S (2016) A wearable biochemical sensor for monitoring alcohol consumption lifestyle through ethyl glucuronide (EtG) detection in human sweat. Sci Rep 6:23111
Mohan AMV, Windmiller JR, Mishra RK, Wang J (2017) Continuous minimally-invasive alcohol monitoring using microneedle sensor arrays. Biosens Bioelectron 91:574–579
Wasfi IA, Al-Awadhi AH, Al-Hatali ZN, Al-Rayami FJ, Al Katheeri NA (2004) Rapid and sensitive static headspace gas chromatography-mass spectrometry method for the analysis of ethanol and abused inhalants in blood. J Chromatogr B Anal Technol Biomed Life Sci 799:331–336
Lidén H, Vijayakumar AR, Gorton L, Marko-Varga G (1998) Rapid alcohol determination in plasma and urine by column liquid chromatography with biosensor detection. J Pharm Biomed Anal 17:1111–1128
Sharma K, Sharma SP, Lahiri SC (2010) Estimation of blood alcohol concentration by horizontal attenuated total reflectance-Fourier transform infrared spectroscopy. Alcohol 44:351–357
El Bakkari M, Luguya R, Correa R, Vincent J (2008) A copper (II)-based multiphasic fluorous colorimetric ethanol assay. New J Chem 32:193–196
Manivannan S, Saranya AM, Renganathan B, Sastikumar D, Gobi G, Park KC (2012) Single-walled carbon nanotubes wrapped poly-methyl methacrylate fiber optic sensor for ammonia, ethanol and methanol vapors at room temperature. Sensors Actuators B Chem 171–172:634–638
Girei SH, Shabaneh AA, Mohd HN, Hamidon MN, Mahdi MA, Yaacob MH (2015) Tapered optical fiber coated with graphene based nanomaterials for measurement of ethanol concentrations in water. Opt Rev 22:385–392
Zhu S, Lei C, Sun J, Zhao X-E, Wang X, Yan X, Liu W, Wang H (2019) Probing NAD+/NADH-dependent biocatalytic transformations based on oxidase mimics of MnO2. Sensors Actuators B Chem 282:896–903
Zhang Y, Li J, An G, He X (2010) Highly porous SnO2 fibers by electrospinning and oxygen plasma etching and its ethanol-sensing properties. Sensors Actuators B Chem 144:43–48
Tian J, Deng S, Li D, Shan D, He W, Zhang X, Shi Y (2013) Bioinspired polydopamine as the scaffold for the active AuNPs anchoring and the chemical simultaneously reduced graphene oxide: characterization and the enhanced biosensing application. Biosens Bioelectron 49:466–471
Thungon PD, Kakoti A, Ngashangva L, Goswami P (2017) Advances in developing rapid, reliable and portable detection systems for alcohol. Biosens Bioelectron 97:83–99
Chinnadayyala SR, Santhosh M, Singh NK, Goswami P (2015) Alcohol oxidase protein mediated in-situ synthesized and stabilized gold nanoparticles for developing amperometric alcohol biosensor. Biosens Bioelectron 69:155–161
Das M, Goswami P (2013) Direct electrochemistry of alcohol oxidase using multiwalled carbon nanotube as electroactive matrix for biosensor application. Bioelectrochemistry 89:19–25
Alhadeff EM, Salgado AM, Cós O, Pereira N, Valero F, Valdman B (2008) Integrated biosensor systems for ethanol analysis. Appl Biochem Biotechnol 146:129–136
Il Kim M, Shim J, Parab HJ, Shin SC, Lee J, Park HG (2012) A convenient alcohol sensor using one-pot nanocomposite entrapping alcohol oxidase and magnetic nanoparticles as peroxidase mimetics. J Nanosci Nanotechnol 12:5914–5919
Fu X, Zhang H, Xiao J, Liu S (2012) Enzymatic detection of ethanol based on H2O2 sensitive quantum dots. J Cent South Univ 19:3040–3045
Nasir M, Nawaz MH, Latif U, Yaqub M, Hayat A, Rahim A (2017) An overview on enzyme-mimicking nanomaterials for use in electrochemical and optical assays. Microchim Acta 184:323–342
Gao L, Zhuang J, Nie L, Zhang J, Zhang Y, Gu N, Wang T, Feng J, Yang D, Perrett S, Yan X (2007) Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat Nanotechnol 2:577–583
Song Y, Qu K, Zhao C, Ren J, Qu X (2010) Graphene oxide: intrinsic peroxidase catalytic activity and its application to glucose detection. Adv Mater 22:2206–2210
Cheng H, Liu Y, Hu Y, Ding Y, Lin S, Cao W, Wang Q, Wu J, Muhammad F, Zhao X, Zhao D, Li Z, Xing H, Wei H (2017) Monitoring of heparin activity in live rats using metal–organic framework nanosheets as peroxidase mimics. Anal Chem 89:11552–11559
Niu X, Xu X, Li X, Pan J, Qiu F, Zhao H, Lan M (2018) Surface charge engineering of nanosized CuS via acidic amino acid modification enables high peroxidase-mimicking activity at neutral pH for one-pot detection of glucose. Chem Commun 54:13443–13446
Niu X, He Y, Zhang W, Li X, Qiu F, Pan J (2018) Elimination of background color interference by immobilizing Prussian blue on carbon cloth: a monolithic peroxidase mimic for on-demand photometric sensing. Sensors Actuators B Chem 256:151–159
Zhou D, Zeng K, Yang M (2019) Gold nanoparticle-loaded hollow Prussian blue nanoparticles with peroxidase-like activity for colorimetric determination of L-lactic acid. Mikrochim Acta 186:121
Wu T, Hou W, Ma Z, Liu M, Liu X, Zhang Y, Yao S (2019) Colorimetric determination of ascorbic acid and the activity of alkaline phosphatase based on the inhibition of the peroxidase-like activity of citric acid-capped Prussian blue nanocubes. Mikrochim Acta 186:123
Zhu W, Liu K, Sun X, Wang X, Li Y, Cheng L, Liu Z (2015) Mn2+-doped prussian blue nanocubes for bimodal imaging and photothermal therapy with enhanced performance. ACS Appl Mater Interfaces 7:11575–11582
Chen Z, Chen C, Huang H, Luo F, Guo L, Zhang L, Lin Z, Chen G (2018) Target-induced horseradish peroxidase deactivation for multicolor colorimetric assay of hydrogen sulfide in rat brain microdialysis. Anal Chem 90:6222–6228
Savola O, Niemelä O, Hillbom M (2004) Blood alcohol is the best indicator of hazardous alcohol drinking in young adults and working-age patients with trauma. Alcohol Alcohol 39:340–345
Chen WJ, Parnell SE, West JR (2001) Nicotine decreases blood alcohol concentration in neonatal rats. Alcohol Clin Exp Res 25:1072–1077
Gonchar MV, Maidan MM, Pavlishko HM, Sibirny AA (2001) A new oxidase-peroxidase kit for ethanol assays in alcoholic beverages. Food Technol Biotechnol 39:37–42
Acknowledgements
Financial support was provided by the National Natural Science Foundation of China (Grant No.21807024), the Youth Top-notch Talents Supporting Plan of Hebei Province (BJ2018007), the Hundred Persons Plan of Hebei Province (E2018050012), and the Natural Science Foundation of Hebei Province (Grant Nos. H2016206280 and H2017206281).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 2446 kb)
Rights and permissions
About this article
Cite this article
Wang, S., Yan, H., Wang, Y. et al. Hollow Prussian Blue nanocubes as peroxidase mimetic and enzyme carriers for colorimetric determination of ethanol. Microchim Acta 186, 738 (2019). https://doi.org/10.1007/s00604-019-3826-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3826-6