Abstract
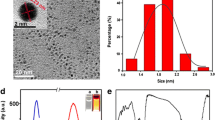
A method is described for rapid (1 h) synthesis of gold nanoclusters (AuNCs) co-functionalized with serine and 11-mercaptoundecanoic acid. The co-functionalized AuNCs exhibit good stability towards temperature, pH values, and over time. They were characterized by atomic force microscopy, high-resolution transmission electron microscopy, dynamic light scattering, and by fluorescence, IR and X-ray photoelectron spectroscopies. The fluorescence of the AuNCs is quenched by Hg(II) and restored on subsequent addition of aureomycin (CTC) or oxytetracycline (OTC). A fluorescent turn-on assay was worked out for simultaneous detection of CTC and OTC based on recording the change of the restored fluorescence measured at 420 and 500 nm under 340 nm photoexcitation. The detection limits are 20 and 9 nM for CTC and OTC, respectively. The concentrations of CTC and OTC can also be visualized by UV illumination. The nanoprobe was successfully applied to the simultaneous determination of CTC and OTC in spiked human urine.

Schematic of a two-wavelength fluorescence recovery method for the simultaneous determination of aureomycin and oxytetracycline. It is based on the use of gold nanocrystals modified with serine and 11-mercaptoundecanoic acid, and on recording the change of the restored fluorescence measured at 420 and 500 nm.





Similar content being viewed by others
References
Hou H, Bai XJ, Xing CY, Gu NY, Zhang BL, Tang JL (2013) Aptamer-based cantilever array sensors for oxytetracycline detection. Anal Chem 85:2010–2014
Lai C, Liu X, Qin L, Zhang C, Zeng G, Huang D, Cheng M, Xu P, Yi H, Huang D (2017) Chitosan-wrapped gold nanoparticles for hydrogen-bonding recognition and colorimetric determination of the antibiotic kanamycin. Microchim Acta 184:2097–2105
Qin L, Zeng G, Lai C, Huang D, Zhang C, Xu P, Hu T, Liu X, Cheng M, Liu Y (2017) A visual application of gold nanoparticles: simple, reliable and sensitive detection of kanamycin based on hydrogen-bonding recognition. Sensors Actuators B Chem 243:946–954
Yan ZD, Gan N, Li TH, Cao YT, Chen YJ (2016) A sensitive electrochemical aptasensor for multiplex antibiotics detection based on high-capacity magnetic hollow porous nanotracers coupling exonuclease-assisted cascade target recycling. Biosens Bioelectron 78:51–57
Liu CB, Lu CX, Tang ZG, Chen X, Wang GH, Sun FX (2015) Aptamer-functionalized magnetic nanoparticles for simultaneous fluorometric determination of oxytetracycline and kanamycin. Microchim Acta 182:2567–2575
Wang Y, Gan N, Zhou Y, Li TH, Hu FT, Cao YT, Chen YJ (2017) Novel label-free and high-throughput microchip electrophoresis platform for multiplex antibiotic residues detection based on aptamer probes and target catalyzed hairpin assembly for signal amplification. Biosens Bioelectron 97:100–106
Li J, Jiang F, Wei X (2010) Molecularly imprinted sensor based on an enzyme amplifier for ultratrace oxytetracycline determination. Anal Chem 82:6074–6078
Li N, Chang CY, Pan W, Tang B (2012) A multicolor Nanoprobe for detection and imaging of tumor-RelatedmRNAs in living cells. Angew Chem Int Ed 51:7426–7430
Pan W, Zhang TT, Yang HJ, Diao W, Li N, Tang B (2013) Multiplexed detection and imaging of intracellular mRNAs using a four-color Nanoprobe. Anal Chem 85:10581–10588
Yang LM, Chen YY, Pan W, Wang HY, Li N, Tang B (2017) Visualizing the conversion process of alcohol-induced fatty liver to steatohepatitis in vivo with a fluorescent Nanoprobe. Anal Chem 89:6196–6201
Yang LM, Ren YF, Pan W, Yu ZZ, Tong LL, Li N, Tang B (2016) Fluorescent nanocomposite for visualizing cross-talk between MicroRNA-21 and hydrogen peroxide in ischemia-reperfusion injury in live cells and in vivo. Anal Chem 88:11886–11891
Chen LY, Wang CW, Yuan Z, Chang HT (2015) Fluorescent gold nanoclusters: recent advances in sensing and imaging. Anal Chem 87:216–229
Xu SH, Gao T, Feng XY, Mao YN, Liu PP, Yu XJ, Luo XL (2016) Dual ligand co-functionalized fluorescent gold nanoclusters for the “turn on” sensing of glutathione in tumor cells. J Mater Chem B 4:1270–1275
Xu SH, Feng XY, Gao T, Liu GF, Mao YN, Lin JH, Yu XJ, Luo XL (2017) Aptamer induced multicoloured AuNCs-MoS2 “switch on” fluorescence resonance energy transfer biosensor for dual color simultaneous detection of multiple tumor markers by single wavelength excitation. Anal Chim Acta 983:173–180
Yan X, Li HX, Cao BC, Ding ZY, Su XG (2015) A highly sensitive dual-readout assay based on gold nanoclusters for folic acid detection. Microchim Acta 182:1281–1288
Bian PP, Zhou J, Liu YY, Ma ZF (2013) One-step fabrication of intense red fluorescent gold nanoclusters and their application in cancer cell imaging. Nano 5:6161–6166
Sun J, Yang F, Zhao D, Yang XR (2014) Highly sensitive real-time assay of inorganic pyrophosphatase activity based on the fluorescent gold nanoclusters. Anal Chem 86:7883–7889
Xu SH, Li XL, Mao YN, Gao T, Feng XY, Luo XL (2016) Novel dual ligand co-functionalized fluorescent gold nanoclusters as a versatile probe for sensitive analysis of Hg2+ and oxytetracycline. Anal Bioanal Chem 408:2955–2962
Santosh A, Remant BKC, Dharmaraj N, Bhattarai N, Kim CH, Kim HY (2006) Spectroscopic identification of S-au interaction in cysteine capped gold nanoparticles, Spectrochim. Acta Part A 63:160–163
Wang Y, Chen JT, Yan XP (2013) Fabrication of transferrin functionalized gold nanoclusters/graphene oxide nanocomposite for turn-on near-infrared fluorescent bioimaging of Cancer cells and small animals. Anal Chem 85:2529–2535
Shang L, Dörlich RM, Brandholt S, Schneider R, Trouillet V, Bruns M, Gerthsen D, Nienhaus GU (2011) Facile preparation of water-soluble fluorescent gold nanoclusters for cellular imaging applications. Nano 3:2009–2014
Whetten RL, Price RC (2007) Nano-Golden Order. Science 318:407–408
Yu Y, Luo ZT, Chevrier DM, Leong DT, Zhang P, Jiang DE, Xie JP (2014) Identification of a highly luminescent Au22(SG)18 nanocluster. J Am Chem Soc 136:1246–1249
Wu ZK, Jin RC (2010) On the ligand's role in the fluorescence of gold nanoclusters. Nano Lett 10:2568–2573
Zhou C, Sun C, Yu M, Qin Y, Wang J, Kim M, Zheng J (2010) Luminescent gold nanoparticles with mixed valence states generated from dissociation of polymeric au (I) thiolates. J Phys Chem C 114:7727–7732
Xu SH, Feng XY, Gao T, Wang RZ, Mao YN, Lin JH, Yu XJ, Luo XL (2017) A novel dual-functional biosensor for fluorometric detection of inorganic pyrophosphate and pyrophosphatase activity based on globulin stabilized gold nanoclusters. Anal Chim Acta 958:22–29
Pyo K, Thanthirige VD, Kwak K, Pandurangan P, Ramakrishna G, Lee D (2015) Ultrabright luminescence from gold nanoclusters: rigidifying the au(I)-thiolate Shell. J Am Chem Soc 137:8244–8250
Huang CC, Chang HT (2007) Parameters for selective colorimetric sensing of mercury (II) in aqueous solutions using mercaptopropionic acid-modified gold nanoparticles. Chem Commun (12):1215–1217
Huang CC, Yang Z, Lee KH, Chang HT (2007) Synthesis of highly fluorescent gold nanoparticles for sensing mercury (II). Angew Chem Int Ed 46:6824–6828
Chen W, Tu X, Guo X (2009) Fluorescent gold nanoparticles-based fluorescence sensor for Cu2+ ions. Chem Commun (13):1736–1738
Zheng J, Zhang C, Dickson RM (2004) Highly fluorescent, watersoluble, size-tunable gold quantum dots. Phys Rev Lett 93:0774021–0774024
Ross PD, Subramanian S (1981) Thermodynamics of protein association reactions:forces contributing to stability. Biochemistry 20:3096–3102
Kawasaki H, Hamaguchi K, Osaka I, Arakawa R (2011) Ph-dependent synthesis of pepsin-mediated gold nanoclusters with blue green and red fluorescent emission. Adv Funct Mater 21:3508–3515
Cheng ZQ, Wang ZY, Gillespie DE, Lausted C, Zheng Z, Yang M, Zhu JS (2015) Plain silver surface Plasmon resonance for microarray application. Anal Chem 87:1466–1469
Gaudin V (2017) Advances in biosensor development for the screening of antibiotic residues in food products of animal origin-A comprehensive review. 90: 363–377
Acknowledgements
S. Xu, Y. Mao, Y. Nie and J. Wang are grateful for the financialsupport provided by NSFC (21505081) and the Doctoral Found of QUST (010022832). X. Luo acknowledges the financial support by NSFC (21422504 and 21675093), the Natural Science Foundation of Shandong Province of China (JQ201406) and the Taishan Scholar Program of Shandong Province, China(ts20110829).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOC 1326 kb)
Rights and permissions
About this article
Cite this article
Mao, Y., Wu, Y., Nie, Y. et al. A two-wavelength fluorescence recovery method for the simultaneous determination of aureomycin and oxytetracycline by using gold nanocrystals modified with serine and 11-mercaptoundecanoic acid. Microchim Acta 185, 222 (2018). https://doi.org/10.1007/s00604-018-2762-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2762-1