Abstract
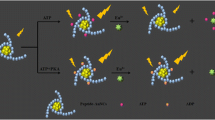
The authors describe a fluorometric method for the determination of the activity and inhibition of protein kinase A (PKA). In the presence of ATP, PKA catalyzes the transfer of phosphate groups from ATP to a peptide, and the generated phosphorylated peptide quenches the fluorescence (measured at excitation/emission peaks of 340/440 nm) of the hydroxyapatite nanoparticles (HAP-NPs). A linear logarithmic relationship of PKA concentrations with fluorescence intensity in the range from 1 to 50 U·L−1 was obtained, and the lower limit of detection (LOD) is 0.5 U·L−1. This is much lower than LODs reported in the literature. The PKA inhibitor H-89 was studied, and the inhibition plot has a sigmoidal shape with a half-maximal inhibitory concentration of around 750 nM of H-89. At a 4.5 nM level of H-89, fluorescence of HAP-NPs fell to levels of no PKA controls, demonstrating that the assay is a viable tool to screen for kinase inhibitors. An assay with Hela cell lysates in combination with forskolin (an activator of adenylyl cyclase) and IBMX (a phosphodiesterase inhibitor used to activate the cellular activity of PKA) resulted in decreased fluorescence of HAP-NPs. This suggests that the assay can be applied for testing in vitro cell kinase activity. In our perception, this method will enable high-throughput screening for kinase-related drugs and fluorometric enzymatic detection in various areas.

Fluorescence assay based on hydroxyapatite nanoparticles (HAP) fluorescence quenching was developed for analysis of the activity and inhibition of protein kinase A (PKA).







Similar content being viewed by others
References
Wang Y, Zhang L, Liang R-P, Bai J-M, Qiu J-D (2013) Using graphene quantum dots as photoluminescent probes for protein kinase sensing. Anal Chem 85(19):9148–9155
Wang L-j, Yang Y, C-y Z (2015) Phosphorylation-directed assembly of a single quantum dot based nanosensor for protein kinase assay. Anal Chem 87(9):4696–4703
Munoz L (2017) Non-kinase targets of protein kinase inhibitors. Nat Rev Drug Discov 16(6):424–U156
Shen C, Xia X, Hu S, Yang M, Wang J (2015) Silver nanoclusters-based fluorescence assay of protein kinase activity and inhibition. Anal Chem 87(1):693–698
Shen C, Li X, Rasooly A, Guo L, Zhang K, Yang M (2016) A single electrochemical biosensor for detecting the activity and inhibition of both protein kinase and alkaline phosphatase based on phosphate ions induced deposition of redox precipitates. Biosens Bioelectron 85:220–225
Sapio L, Di Maiolo F, Illiano M, Esposito A, Chiosi E, Spina A, Naviglio S (2014) Targeting protein kinase A in cancer therapy: an update. EXCLI J 13:843–855
Melnikova I, Golden J (2004) Targeting protein kinases. Nat Rev Drug Discov 3(12):993–994
Li T, Liu X, Liu D, Wang Z (2013) Sensitive detection of protein kinase A activity in cell lysates by peptide microarray-based assay. Anal Chem 85(15):7033–7037
Schachterle C, Christian F, Fernandes JM, Klussmann E (2015) Screening for small molecule disruptors of AKAP-PKA interactions. Methods Mol Biol 1294:151–166
Leytus SP, Patterson WL, Mangel WF (1983) New class of sensitive and selective fluorogenic substrates for serine proteinases. Amino acid and dipeptide derivatives of rhodamine. Biochem J 215(2):253–260
Leytus SP, Melhado LL, Mangel WF (1983) Rhodamine-based compounds as fluorogenic substrates for serine proteinases. Biochem J 209(2):299–307
Haider A, Haider S, Han SS, Kang I-K (2017) Recent advances in the synthesis, functionalization and biomedical applications of hydroxyapatite: a review. RSC Adv 7(13):7442–7458
Syamchand SS, Sony G (2015) Multifunctional hydroxyapatite nanoparticles for drug delivery and multimodal molecular imaging. Microchim Acta 182(9–10):1567–1589
Huang Y, Tang C, Liu J, Cheng J, Si Z, Li T, Yang M (2017) Signal amplification strategy for electrochemical immunosensing based on a molybdophosphate induced enhanced redox current on the surface of hydroxyapatite nanoparticles. Microchim Acta 184(3):855–861
Qu F, Yang M, Rasooly A (2016) Dual signal amplification electrochemical biosensor for monitoring the activity and inhibition of the Alzheimer's related protease beta-Secretase. Anal Chem 88(21):10559–10565
Jiang W, Tian D, Zhang L, Guo Q, Cui Y, Yang M (2017) Dual signal amplification strategy for amperometric aptasensing using hydroxyapatite nanoparticles. Application to the sensitive detection of the cancer biomarker platelet-derived growth factor BB. Microchim Acta 184:4375–4381
Kawasaki K (1991) Hydroxyapatite as a liquid chromatographic packing. J Chromatogr 544:147–184
Krenkova J, Lacher NA, Svec F (2010) Control of selectivity via nanochemistry: monolithic capillary column containing hydroxyapatite nanoparticles for separation of proteins and enrichment of phosphopeptides. Anal Chem 82(19):8335–8341
Kim EJ, Choi SW, Hong SH (2007) Synthesis and photoluminescence properties of Eu3+−doped calcium phosphates. J Am Ceram Soc 90(9):2795–2798
Al-Kattan A, Dufour P, Dexpert-Ghys J, Drouet C (2010) Preparation and physicochemical characteristics of luminescent apatite-based colloids. J Phys Chem C 114(7):2918–2924
Chen F, Huang P, Zhu YJ, Wu J, Zhang CL, Cui DX (2011) The photoluminescence, drug delivery and imaging properties of multifunctional Eu3+/Gd3+ dual-doped hydroxyapatite nanorods. Biomaterials 32(34):9031–9039
Zhang CM, Li CX, Huang SS, Hou ZY, Cheng ZY, Yang PP, Peng C, Lin J (2010) Self-activated luminescent and mesoporous strontium hydroxyapatite nanorods for drug delivery. Biomaterials 31(12):3374–3383
Lopez-Macipe A, Gomez-Morales J, Rodriguez-Clemente R (1998) Nanosized hydroxyapatite precipitation from homogeneous calcium/citrate/phosphate solutions using microwave and conventional heating. Adv Mater 10(1):49–54
Singh RK, Kim TH, Patel KD, Kim JJ, Kim HW (2014) Development of biocompatible apatite nanorod-based drug-delivery system with in situ fluorescence imaging capacity. J Mater Chem B 2(14):2039–2050
Zhang C, Yang J, Quan Z, Yang P, Li C, Hou Z, Lin J (2009) Hydroxyapatite nano- and microcrystals with multiform morphologies: controllable synthesis and luminescence properties. Cryst Growth Des 9(6):2725–2733
Vdovenko SI, Kolycheva MT, Gerus II, Kukhar VP (1993) Influence of phosphate ion on the fluorescence of 3-fluorotyrosine. Amino Acids 4(3):303–306
Bakkialakshmi S, Shanthi B, Chandrakala D (2011) Interaction of potassium mono and di phosphates with bovine serum albumin studied by fluorescence quenching method. J Fluoresc 21(2):687–692
Xu X, Liu X, Nie Z, Pan Y, Guo M, Yao S (2011) Label-free fluorescent detection of protein kinase activity based on the aggregation behavior of unmodified quantum dots. Anal Chem 83:52–59
Yin C, Wang M, Lei C, Wang Z, Li P, Li Y, Li W, Huang Y, Nie Z, Yao S (2015) Phosphorylation-mediated assembly of a semisynthetic fuorescent protein for label-free detection of protein kinase activity. Anal Chem 87(12):6311–6318
Liu Q, Na W, Wang L, Su X (2017) Gold nanocluster-based fluorescent assay for label-free detection of protein kinase and its inhibitors. Microchim Acta 184:3381–3387
Wang M, Wang L, Liu Q, Su X (2018) A fluorescence sensor for protein kinase activity detection based on gold nanoparticles/copper nanoclusters system. Sensors Actuators B Chem 256:691–698
Lu G, Tan P, Lei C, Nie Z, Huang Y, Yao S (2014) Fluorescent detection of protein kinase based on positively charged gold nanoparticles. Talanta 128:360–365
Yin H, Wang M, Li B, Yang Z, Zhou Y, Ai S (2015) A sensitive electrochemical biosensor for detection of protein kinase A activity and inhibitors based on Phos-tag and enzymatic signal amplification. Biosens Bioelectron 63:26–32
Ren W, Liu C, Lian S, Li Z (2013) Flow cytometry-assisted mix-and-read assay for ultrasensitive detection of protein kinase activity by use of Zr4+-functionalized mesoporous SiO2 microspheres. Anal Chem 85(22):10956–10961
Acknowledgments
The authors thank the support of this work by the National Natural Science Foundation of China (No. 21575165) and the Hunan Provincial Science and Technology Plan Project, China (No. 2016TP1007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Zhang, K., Zeng, K., Shen, C. et al. Determination of protein kinase A activity and inhibition by using hydroxyapatite nanoparticles as a fluorescent probe. Microchim Acta 185, 225 (2018). https://doi.org/10.1007/s00604-018-2754-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2754-1