Abstract
Purposes
This study aimed to explore the effect of increased body mass index (BMI) values (overweight: BMI ≥ 25–30 kg/m2; obese: BMI ≥ 30 kg/m2) on surgical outcomes after radical resection for low rectal cancer (LRC).
Methods
Patients with LRC who underwent radical surgery from January 2009 to December 2013 were included. The patients were divided into three groups according to their BMI values (control group: BMI < 25 kg/m2; overweight group: BMI 25 to < 30 kg/m2; obese group: BMI ≥ 30 kg/m2). The patients’ clinicopathological characteristics and survival data were collected and analyzed.
Results
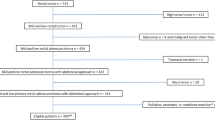
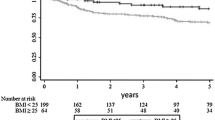
A total of 792 patients were enrolled in this study finally (control, n = 624; overweight, n = 147; obese, n = 21). The baseline characteristics of the three groups were similar. We found that an increased BMI was associated with a longer operative time (P < 0.001) and length of postoperative hospital stay (P = 0.032). Patients with increased BMI values had a significantly higher incidence of postoperative complications, including pulmonary infection (P = 0.008), anastomotic leakage (P = 0.029), allergy (P = 0.017) and incisional hernia (P = 0.045). The limited data showed that the pathological outcomes of the three groups did not differ to a statistically significant extent. A multivariate analysis showed that increased BMI was not associated with poorer OS or DFS.
Conclusion
In LRC resection, an increased BMI was associated with a longer operative time, postoperative hospital stay, and an increased number of postoperative complications. However, it did not contribute to poorer pathological or survival outcomes.

Similar content being viewed by others
References
Nicholls J. Rectal cancer seen over 30 years. Colorectal Disease. 2014;16(9):659.
Rullier E, Denost Q, Vendrely V, Rullier A, Laurent C. Low rectal cancer: classification and standardization of surgery. Dis Colon Rectum. 2013;56(5):560–7.
Dietz DW. Multidisciplinary management of rectal cancer: the OSTRICH. J Gastrointest Surg. 2013;17(10):1863–8.
Allaix ME, Fichera A. Modern rectal cancer multidisciplinary treatment: the role of radiation and surgery. Ann Surg Oncol. 2013;20(9):2921–8.
de Chaisemartin C, Penna C, Goere D, Benoist S, Beauchet A, Julie C, et al. Presentation and prognosis of local recurrence after total mesorectal excision. Colorectal Dis. 2009;11(1):60–6.
Bebenek M, Pudelko M, Cisarz K, Balcerzak A, Tupikowski W, Wojciechowski L, et al. Therapeutic results in low-rectal cancer patients treated with abdominosacral resection are similar to those obtained by means of anterior resection in mid- and upper-rectal cancer cases. Eur J Surg Oncol. 2007;33(3):320–3.
Jeong SY, Park JW, Nam BH, Kim S, Kang SB, Lim SB, et al. Open versus laparoscopic surgery for mid-rectal or low-rectal cancer after neoadjuvant chemoradiotherapy (COREAN trial): survival outcomes of an open-label, non-inferiority, randomised controlled trial. Lancet Oncol. 2014;15(7):767–74.
Bordeianou L, Maguire LH, Alavi K, Sudan R, Wise PE, Kaiser AM. Sphincter-sparing surgery in patients with low-lying rectal cancer: techniques, oncologic outcomes, and functional results. J Gastrointest Surg. 2014;18(7):1358–72.
Denost Q, Quintane L, Buscail E, Martenot M, Laurent C, Rullier E. Short- and long-term impact of body mass index on laparoscopic rectal cancer surgery. Colorectal Dis. 2013;15(4):463–9.
Shukla PJ, Makino T, Trencheva K, Milsom JW. Challenges of improving outcomes of rectal cancer surgery in obese patients. J Am Coll Surg. 2011;212(1):130 (author reply-1).
Wang K, Wang D, Pan L, Yu Y, Dong F, Li L, et al. Prevalence of obesity and related factors among Bouyei and Han peoples in Guizhou Province, Southwest China. PLoS One. 2015;10(6):e0129230.
Levi Z, Kark JD, Katz LH, Twig G, Derazne E, Tzur D, et al. Adolescent body mass index and risk of colon and rectal cancer in a cohort of 1.79 million Israeli men and women: a population-based study. Cancer. 2017;123(20):4022–30.
Fleshman J, Branda M, Sargent DJ, Boller AM, George V, Abbas M, et al. Effect of laparoscopic-assisted resection vs open resection of stage II or III rectal cancer on pathologic outcomes: the ACOSOG Z6051 randomized clinical trial. JAMA. 2015;314(13):1346–55.
Sun Y, Chi P. Impact of body mass index on surgical and oncological outcomes in laparoscopic total mesorectal excision for locally advanced rectal cancer after neoadjuvant 5-fluorouracil-based chemoradiotherapy. Gastroenterol Res Pract. 2017;2017:1509140.
Doleman B, Mills KT, Lim S, Zelhart MD, Gagliardi G. Body mass index and colorectal cancer prognosis: a systematic review and meta-analysis. Tech Coloproctol. 2016;20(8):517–35.
Kwak HD, Ju JK, Kang DW, Baek SJ, Kwak JM, Kim J, et al. Outcomes according to body mass index following laparoscopic surgery in patients with colorectal cancer. Gastroenterol Res Pract. 2018;14(2):134–9.
Ogiso S, Yamaguchi T, Hata H, Fukuda M, Ikai I, Yamato T, et al. Evaluation of factors affecting the difficulty of laparoscopic anterior resection for rectal cancer: “narrow pelvis” is not a contraindication. Surg Endosc. 2011;25(6):1907–12.
Keller DS, Park KJ, Augestad KM, Delaney CP. Integration of open and laparoscopic approaches for rectal cancer resection: oncologic and short-term outcomes. Surg Endosc. 2014;28(7):2129–36.
Wu QB, Deng XB, Zhang XB, Kong LH, Zhou ZG, Wang ZQ. Short-term and long-term outcomes of laparoscopic versus open surgery for low rectal cancer. J Laparoendosc Adv Surg Tech Part A. 2018;28(6):637–44.
Cakir H, Heus C, Verduin WM, Lak A, Doodeman HJ, Bemelman WA, et al. Visceral obesity, body mass index and risk of complications after colon cancer resection: a retrospective cohort study. Surgery. 2015;157(5):909–15.
Kawamata F, Homma S, Minagawa N, Kawamura H, Takahashi N, Taketomi A. Comparison of single-incision plus one additional port laparoscopy-assisted anterior resection with conventional laparoscopy-assisted anterior resection for rectal cancer. World J Surg. 2014;38(10):2716–23.
Jeong SH, Kim P, Yi SW, Kim YJ, Baeg MK, Yi JJ. Body mass index and gastrointestinal cancer mortality in Korean adults: a prospective cohort study. J Gastroenterol Hepatol. 2018;33(9):1582–9.
Bell S, Kong JC, Wale R, Staples M, Oliva K, Wilkins S, et al. The effect of increasing body mass index on laparoscopic surgery for colon and rectal cancer. Colorectal Dis. 2018;20(9):778–88.
Kuritzkes BA, Pappou EP, Kiran RP, Baser O, Fan L, Guo X, et al. Visceral fat area, not body mass index, predicts postoperative 30-day morbidity in patients undergoing colon resection for cancer. Int J Colorectal Dis. 2018;33(8):1019–28.
Hovaguimian F, Lysakowski C, Elia N, Tramer MR. Effect of intraoperative high inspired oxygen fraction on surgical site infection, postoperative nausea and vomiting, and pulmonary function: systematic review and meta-analysis of randomized controlled trials. Anesthesiology. 2013;119(2):303–16.
Nakayama M, Yoshimatsu K, Yokomizo H, Yano Y, Okayama S, Satake M, et al. Incidence and risk factors for incisional hernia after open surgery for colorectal cancer. Hepato-gastroenterology. 2014;61(133):1220–3.
Kroenke CH, Neugebauer R, Meyerhardt J, Prado CM, Weltzien E, Kwan ML, et al. Analysis of body mass index and mortality in patients with colorectal cancer using causal diagrams. JAMA Oncol. 2016;2(9):1137–45.
Greco M, Capretti G, Beretta L, Gemma M, Pecorelli N, Braga M. Enhanced recovery program in colorectal surgery: a meta-analysis of randomized controlled trials. World J Surg. 2014;38(6):1531–41.
Sun Y, Xu Z, Lin H, Lu X, Huang Y, Huang S, et al. Impact of body mass index on treatment outcome of neoadjuvant chemoradiotherapy in locally advanced rectal cancer. Eur J Surg Oncol. 2017;43(10):1828–34.
Nir S, Greenberg R, Shacham-Shmueli E, White I, Schneebaum S, Avital S. Number of retrieved lymph nodes and survival in node-negative patients undergoing laparoscopic colorectal surgery for cancer. Tech Coloproctol. 2010;14(2):147–52.
Kuo YH, Lee KF, Chin CC, Huang WS, Yeh CH, Wang JY. Does body mass index impact the number of LNs harvested and influence long-term survival rate in patients with stage III colon cancer? Int J Colorectal Dis. 2012;27(12):1625–35.
Akagi Y, Adachi Y, Kinugasa T, Oka Y, Mizobe T, Shirouzu K. Lymph node evaluation and survival in colorectal cancer: review of population-based, prospective studies. Anticancer Res. 2013;33(7):2839–47.
Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014;383(9927):1490–502.
Acknowledgements
This work was supported by the Science and Technology Support Program of the Science and Technology Department of Sichuan Province (Grant numbers: 2016SZ0043).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest in association with the present study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Wu, Q., Gu, C. et al. The effect of increased body mass index values on surgical outcomes after radical resection for low rectal cancer. Surg Today 49, 401–409 (2019). https://doi.org/10.1007/s00595-019-01778-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-019-01778-w