Abstract
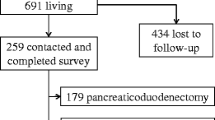
To characterize the clinical signature and etiopathogenetic factors of diabetes associated with pancreas disease [type 3 diabetes mellitus (T3cDM)]. To estimate incidence and identify predictors of both diabetes onset and remission after pancreatic surgery. A prospective observational study was conducted. From January 2008 to December 2012, patients (n = 651) with new diagnosis of pancreatic disease admitted to the Pancreatic Surgery Unit of the San Raffaele Scientific Institute were evaluated. Hospital and/or outpatient medical records were reviewed. Blood biochemical values including fasting blood glucose, insulin and/or C-peptide, glycosylated hemoglobin and anti-islet antibodies were determined. Diabetes onset was assessed after surgery and during follow-up. At baseline, the prevalence of diabetes was 38 % (age of onset 64 ± 11 years). In most cases, diabetes occurred within 48 months from pancreatic disease diagnosis. Among different pancreatic diseases, minor differences were observed in diabetes characteristics, with the exception of the prevalence. Diabetes appeared associated with classical risk factors for type 2 diabetes (i.e., age, sex, family history of diabetes and body mass index), and both beta-cell dysfunction and insulin resistance appeared relevant determinants. The prevalence of adult-onset autoimmune diabetes was as previously reported within type 2 diabetes. Within a few days after surgery, either diabetes remission or new-onset diabetes was observed. In patients with pancreatic cancer, no difference in diabetes remission was observed after palliative or resective surgery. Classical risk factors for type 2 diabetes were associated with the onset of diabetes after surgery. T3cDM appeared as a heterogeneous entity strongly overlapped with type 2 diabetes.



Similar content being viewed by others
Abbreviations
- eGFR:
-
Estimation of glomerular filtration rate
- FPG:
-
Fasting plasma glucose
- GADA:
-
Autoantibodies to glutamic acid decarboxylase
- HO:
-
Oral hypoglycemic agents
- IA-2A:
-
Autoantibodies to insulinoma-associated protein 2
- IAA:
-
Autoantibodies to insulin
- IFG:
-
Impaired fasting glucose
- NFG:
-
Normal fasting glucose
- PD:
-
Pancreaticoduodenectomy
- PET:
-
Pancreatic neuroendocrine tumor
- T3cDM:
-
Type 3 diabetes mellitus
- ZnT8A:
-
Autoantibodies to zinc transporter 8 antigen
References
American Diabetes Association (2014) Diagnosis and classification of diabetes mellitus. Diabetes Care 37(Suppl 1):S81–S90. doi:10.2337/dc14-S081
Cui Y, Andersen DK (2011) Pancreatogenic diabetes: special considerations for management. Pancreatology 11(3):279–294. doi:10.1159/000329188
Lahat G, Ben Haim M, Nachmany I, Sever R, Blachar A, Nakache R, Klausner JM (2009) Pancreatic incidentalomas: high rate of potentially malignant tumors. J Am Coll Surg 209(3):313–319. doi:10.1016/j.jamcollsurg.2009.05.009
Moran A, Becker D, Casella SJ, Gottlieb PA, Kirkman MS, Marshall BC, Slovis B (2010) Epidemiology, pathophysiology, and prognostic implications of cystic fibrosis-related diabetes: a technical review. Diabetes Care 33(12):2677–2683. doi:10.2337/dc10-1279
Jupp J, Fine D, Johnson CD (2010) The epidemiology and socioeconomic impact of chronic pancreatitis. Best Pract Res Clin Gastroenterol 24(3):219–231. doi:10.1016/j.bpg.2010.03.005
Lemmens VE, Bosscha K, van der Schelling G, Brenninkmeijer S, Coebergh JW, de Hingh IH (2011) Improving outcome for patients with pancreatic cancer through centralization. Br J Surg 98(10):1455–1462. doi:10.1002/bjs.7581
Piemonti L, Everly MJ, Maffi P, Scavini M, Poli F, Nano R, Cardillo M, Melzi R, Mercalli A, Sordi V, Lampasona V, Espadas de Arias A, Scalamogna M, Bosi E, Bonifacio E, Secchi A, Terasaki PI (2013) Alloantibody and autoantibody monitoring predicts islet transplantation outcome in human type 1 diabetes. Diabetes 62(5):1656–1664. doi:10.2337/db12-1258
Wallace TM, Levy JC, Matthews DR (2004) Use and abuse of HOMA modeling. Diabetes Care 27(6):1487–1495. doi:10.2337/diacare.27.6.1487
Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130(6):461–470. doi:10.7326/0003-4819-130-6-199903160-00002
Chari ST, Leibson CL, Rabe KG, Timmons LJ, Ransom J, de Andrade M, Petersen GM (2008) Pancreatic cancer-associated diabetes mellitus: prevalence and temporal association with diagnosis of cancer. Gastroenterology 134(1):95–101. doi:10.1053/j.gastro.2007.10.040
Pannala R, Basu A, Petersen GM, Chari ST (2009) New-onset diabetes: a potential clue to the early diagnosis of pancreatic cancer. Lancet Oncol 10(1):88–95. doi:10.1016/S1470-2045(08)70337-1
Damiano J, Bordier L, Le Berre JP, Margery J, Dupuy O, Mayaudon H, Bauduceau B (2004) Should pancreas imaging be recommanded in patients over 50 years when diabetes is discovered because of acute symptoms? Diabetes Metab 30(2):203–207. doi:DM-04-2004-30-2-1262-3636-101019-ART16
Pelaez-Luna M, Takahashi N, Fletcher JG, Chari ST (2007) Resectability of presymptomatic pancreatic cancer and its relationship to onset of diabetes: a retrospective review of CT scans and fasting glucose values prior to diagnosis. Am J Gastroenterol 102(10):2157–2163. doi:10.1111/j.1572-0241.2007.01480.x
Garancini MP, Calori G, Ruotolo G, Manara E, Izzo A, Ebbli E, Bozzetti AM, Boari L, Lazzari P, Gallus G (1995) Prevalence of NIDDM and impaired glucose tolerance in Italy: an OGTT-based population study. Diabetologia 38(3):306–313. doi:10.1007/BF00400635
Festa A, Williams K, Hanley AJ, Haffner SM (2008) Beta-cell dysfunction in subjects with impaired glucose tolerance and early type 2 diabetes: comparison of surrogate markers with first-phase insulin secretion from an intravenous glucose tolerance test. Diabetes 57(6):1638–1644. doi:10.2337/db07-0954
Kahn SE (2003) The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 46(1):3–19. doi:10.1007/s00125-002-1009-0
Buzzetti R, Di Pietro S, Giaccari A, Petrone A, Locatelli M, Suraci C, Capizzi M, Arpi ML, Bazzigaluppi E, Dotta F, Bosi E (2007) High titer of autoantibodies to GAD identifies a specific phenotype of adult-onset autoimmune diabetes. Diabetes Care 30(4):932–938. doi:10.2337/dc06-1696
Wang F, Herrington M, Larsson J, Permert J (2003) The relationship between diabetes and pancreatic cancer. Mol Cancer 2:4
Gullo L, Pezzilli R, Morselli-Labate AM (1994) Diabetes and the risk of pancreatic cancer. N Engl J Med 331(2):81–84. doi:10.1056/NEJM199407143310203
Huxley R, Ansary-Moghaddam A, Berrington de Gonzalez A, Barzi F, Woodward M (2005) Type-II diabetes and pancreatic cancer: a meta-analysis of 36 studies. Br J Cancer 92(11):2076–2083. doi:10.1038/sj.bjc.6602619
Vigneri P, Frasca F, Sciacca L, Pandini G, Vigneri R (2009) Diabetes and cancer. Endocr Relat Cancer 16(4):1103–1123. doi:10.1677/ERC-09-0087
Ben Q, Xu M, Ning X, Liu J, Hong S, Huang W, Zhang H, Li Z (2011) Diabetes mellitus and risk of pancreatic cancer: a meta-analysis of cohort studies. Eur J Cancer 47(13):1928–1937. doi:10.1016/j.ejca.2011.03.003
Tseng CH (2013) Diabetes, insulin use, smoking, and pancreatic cancer mortality in Taiwan. Acta Diabetol 50(6):879–886. doi:10.1007/s00592-013-0471-0
Nicolucci A (2010) Epidemiological aspects of neoplasms in diabetes. Acta Diabetol 47(2):87–95. doi:10.1007/s00592-010-0187-3
Permert J, Larsson J, Westermark GT, Herrington MK, Christmanson L, Pour PM, Westermark P, Adrian TE (1994) Islet amyloid polypeptide in patients with pancreatic cancer and diabetes. N Engl J Med 330(5):313–318. doi:10.1056/NEJM199402033300503
Basso D, Greco E, Fogar P, Pucci P, Flagiello A, Baldo G, Giunco S, Valerio A, Navaglia F, Zambon CF, Falda A, Pedrazzoli S, Plebani M (2006) Pancreatic cancer-derived S-100A8N-terminal peptide: a diabetes cause? Clin Chim Acta 372(1–2):120–128. doi:10.1016/j.cca.2006.03.027
Aggarwal G, Ramachandran V, Javeed N, Arumugam T, Dutta S, Klee GG, Klee EW, Smyrk TC, Bamlet W, Han JJ, Rumie Vittar NB, de Andrade M, Mukhopadhyay D, Petersen GM, Fernandez-Zapico ME, Logsdon CD, Chari ST (2012) Adrenomedullin is up-regulated in patients with pancreatic cancer and causes insulin resistance in beta cells and mice. Gastroenterology 143(6):1510–1517 e1511. doi:10.1053/j.gastro.2012.08.044
Pannala R, Leirness JB, Bamlet WR, Basu A, Petersen GM, Chari ST (2008) Prevalence and clinical profile of pancreatic cancer-associated diabetes mellitus. Gastroenterology 134(4):981–987. doi:10.1053/j.gastro.2008.01.039
Pannala R, Leibson CL, Rabe KG, Timmons LJ, Ransom J, de Andrade M, Petersen GM, Chari ST (2009) Temporal association of changes in fasting blood glucose and body mass index with diagnosis of pancreatic cancer. Am J Gastroenterol 104(9):2318–2325. doi:10.1038/ajg.2009.253
Chari ST, Leibson CL, Rabe KG, Ransom J, de Andrade M, Petersen GM (2005) Probability of pancreatic cancer following diabetes: a population-based study. Gastroenterology 129(2):504–511. doi:10.1016/j.gastro.2005.05.007
Permert J, Ihse I, Jorfeldt L, von Schenck H, Arnquist HJ, Larsson J (1993) Improved glucose metabolism after subtotal pancreatectomy for pancreatic cancer. Br J Surg 80(8):1047–1050
Fogar P, Pasquali C, Basso D, Sperti C, Panozzo MP, Tessari G, D’Angeli F, Del Favero G, Plebani M (1994) Diabetes mellitus in pancreatic cancer follow-up. Anticancer Res 14(6B):2827–2830
Sala PC, Torrinhas RS, Heymsfield SB, Waitzberg DL (2012) Type 2 diabetes mellitus: a possible surgically reversible intestinal dysfunction. Obes Surg 22(1):167–176. doi:10.1007/s11695-011-0563-2
Saad MF, Knowler WC, Pettitt DJ, Nelson RG, Mott DM, Bennett PH (1989) Sequential changes in serum insulin concentration during development of non-insulin-dependent diabetes. Lancet 1(8651):1356–1359. doi:10.1016/S0140-6736(89)92804-3
Vigneri R (2009) Diabetes: diabetes therapy and cancer risk. Nat Rev Endocrinol 5(12):651–652. doi:10.1038/nrendo.2009.219
Perseghin G, Calori G, Lattuada G, Ragogna F, Dugnani E, Garancini MP, Crosignani P, Villa M, Bosi E, Ruotolo G, Piemonti L (2012) Insulin resistance/hyperinsulinemia and cancer mortality: the Cremona study at the 15th year of follow-up. Acta Diabetol 49(6):421–428. doi:10.1007/s00592-011-0361-2
Acknowledgments
This study was supported by the Associazione Italiana per la Ricerca sul Cancro (AIRC, bando 5 × 1,000 N_12182 and Progetto IGN_11783).
Conflict of interest
Gianpaolo Balzano, Erica Dugnani, Valentina Pasquale, Giovanni Capretti, Maria Grazia Radaelli, Tania Garito, Gregorio Stratta, Alessandro Nini, Raffaele Di Fenza, Renato Castoldi, Carlo Staudacher, Michele Reni, Marina Scavini, Claudio Doglioni and Lorenzo Piemonti declare that they have no conflict of interest.
Human and Animal Rights disclosure
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent disclosure
Informed consent was obtained from all patients for being included in the study.
Author information
Authors and Affiliations
Corresponding author
Additional information
Managed by Antonio Secchi.
Gianpaolo Balzano and Erica Dugnani have equally contributed to the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Balzano, G., Dugnani, E., Pasquale, V. et al. Clinical signature and pathogenetic factors of diabetes associated with pancreas disease (T3cDM): a prospective observational study in surgical patients. Acta Diabetol 51, 801–811 (2014). https://doi.org/10.1007/s00592-014-0614-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-014-0614-y