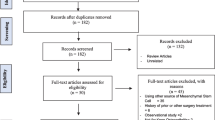
Abstract
Background
Despite being a common cause of quality-of-life impairment, there are no efficacious therapies that could prevent the progression of knee osteoarthritis (KOA). We conducted an open-label trial of human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) and hyaluronic acid (HA) for treating KOA.
Methods
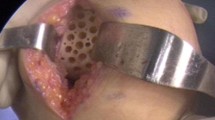
This open-label study was conducted from July 2015 to December 2018 at Cipto Mangunkusumo Hospital, Jakarta, Indonesia. Patients diagnosed with KOA were injected three times, comprising of 10 × 106 units of hUC-MSCs in 2-ml secretome implantation and 2-ml hyaluronic acid (HA) injection in the first week, followed with 2-ml HA injection twice in the second and third week.
Results
Twenty-nine subjects (57 knees) were recruited. Seventeen (58.6%) subjects were male, and the mean age was 58.3 ± 9.6 years. Thirty-three (57.9%) knees were classified into Kellgren–Lawrence grade I–II KOA (mild OA). hUC-MSCs significantly decreased pain measured by visual analogue scale in severe KOA from initial to 6th month follow-up [5 ± 2.97 to 3.38 ± 2.44 (p = 0.035)]. The International Knee Documentation Committee score significantly increased at 6th month follow-up (53.26 ± 16.66 to 65.49 ± 13.01, p < 0.001, in subjects with grade I–II and 48.84 ± 18.41 to 61.83 ± 18.83, p = 0.008, in subjects with severe KOA). The Western Ontario and McMaster Universities Osteoarthritis decreased significantly in both groups from initial to 6th month follow-up (from 22.55 ± 15.94 to 13.23 ± 10.29, p = 0.003, and from 27.57 ± 15.99 to 17.92 ± 19.1, p = 0.003, in those with mild and severe KOA, respectively).
Conclusions
hUC-MSCs could be a potentially new regenerative treatment for KOA. The maximum effect of hUC-MSCs was achieved after 6 months of injection.
Level of evidence
Therapeutic level II.
Similar content being viewed by others
References
Morille M, Toupet K, Montero-Menei CN et al (2016) PLGA-based microcarriers induce mesenchymal stem cell chondrogenesis and stimulate cartilage repair in osteoarthritis. Biomaterials. https://doi.org/10.1016/j.biomaterials.2016.02.022
Zhen G, Wen C, Jia X et al (2013) Inhibition of TGF-β signaling in mesenchymal stem cells of subchondral bone attenuates osteoarthritis. Nat Med 19:704–712. https://doi.org/10.1038/nm.3143
Wang Y, Yuan M, Guo Q-Y et al (2015) Mesenchymal stem cells for treating articular cartilage defects and osteoarthritis. Cell Transplant 24:1661–1678. https://doi.org/10.3727/096368914X683485
Li G, Yin J, Gao J et al (2013) Subchondral bone in osteoarthritis: insight into risk factors and microstructural changes. Arthritis Res Ther 15:223. https://doi.org/10.1186/ar4405
Dulay GS, Cooper C, Dennison EM (2015) Knee pain, knee injury, knee osteoarthritis and work. Best Pract Res Clin Rheumatol 29:454–461. https://doi.org/10.1016/j.berh.2015.05.005
Mamidi MK, Das AK, Zakaria Z, Bhonde R (2016) Mesenchymal stromal cells for cartilage repair in osteoarthritis. Osteoarthr Cartil 24:1307–1361. https://doi.org/10.1016/j.joca.2016.03.003
Hussain SM, Neilly DW, Baliga S et al (2016) Knee osteoarthritis: a review of management options. Scott Med J 61:7–16. https://doi.org/10.1177/0036933015619588
Barry F, Murphy M (2013) Mesenchymal stem cells in joint disease and repair. Nat Rev Rheumatol 9:584–594
Davatchi F, Sadeghi Abdollahi B, Mohyeddin M, Nikbin B (2016) Mesenchymal stem cell therapy for knee osteoarthritis: 5 years follow-up of three patients. Int J Rheum Dis 19:219–225. https://doi.org/10.1111/1756-185X.12670
Buckwalter JA (2000) Advancing the science and art of orthopaedics: lessons from history. J Bone Jt Surg Ser A 82-A:1782–1803. https://doi.org/10.2106/00004623-200012000-00012
Kurtz S, Ong K, Lau E et al (2007) Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Jt Surg Ser A 89:780–785. https://doi.org/10.2106/JBJS.F.00222
Derar H, Shahinpoor M (2015) Recent patents and designs on hip replacement prostheses. Open Biomed Eng J. https://doi.org/10.2174/1874120701509010092
Gallo J, Kamínek P, Tichá V et al (2002) Particle disease. A comprehensive theory of periprosthetic osteolysis: a review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 146:21–28. https://doi.org/10.5507/bp.2002.004
Sargeant A, Goswami T (2006) Hip implants: paper V. Mater Des, Physiological Eff. https://doi.org/10.1016/j.matdes.2004.10.028
Srimongkol S (2012) A review of mathematical modeling in total hip replacement. Int Math Forum 7:2561–2569
Lo GH, LaValley M, McAlindon T, Felson DT (2003) Intra-articular hyaluronic acid in treatment of knee osteoarthritis: a meta-analysis. J Am Med Assoc 290:3115–3321. https://doi.org/10.1001/jama.290.23.3115
Gallagher B, Tjoumakaris FP, Harwood MI et al (2015) Chondroprotection and the prevention of osteoarthritis progression of the knee: a systematic review of treatment agents. Am J Sports Med 43:734–744. https://doi.org/10.1177/0363546514533777
Deng M-W, Wei S-J, Yew T-L et al (2015) Cell therapy with G-CSF-mobilized stem cells in a rat osteoarthritis model. Cell Transplant 24:1085–1096. https://doi.org/10.3727/096368914X680091
Saw KY, Anz A, Merican S et al (2011) Articular cartilage regeneration with autologous peripheral blood progenitor cells and hyaluronic acid after arthroscopic subchondral drilling: a report of 5 cases with histology. Arthrosc J Arthrosc Relat Surg 27:493–506. https://doi.org/10.1016/j.arthro.2010.11.054
Chiang ER, Ma HL, Wang JP et al (2016) Allogeneic mesenchymal stem cells in combination with hyaluronic acid for the treatment of osteoarthritis in rabbits. PLoS ONE 11:e0149835. https://doi.org/10.1371/journal.pone.0149835
Liu Q, Niu J, Huang J et al (2015) Knee osteoarthritis and all-cause mortality: the Wuchuan Osteoarthritis Study. Osteoarthr Cartil 23:1154–1157. https://doi.org/10.1016/j.joca.2015.03.021
Gore M, Tai K-S, Sadosky A et al (2011) Clinical comorbidities, treatment patterns, and direct medical costs of patients with osteoarthritis in usual care: a retrospective claims database analysis. J Med Econ 14:497–507. https://doi.org/10.3111/13696998.2011.594347
Liu TM, Martina M, Hutmacher DW et al (2006) Identification of common pathways mediating differentiation of bone marrow- and adipose tissue-derived human mesenchymal stem cells into three mesenchymal lineages. Stem Cells 25:750–760. https://doi.org/10.1634/stemcells.2006-0394
Ding D-C, Chang Y-H, Shyu W-C, Lin S-Z (2015) Human umbilical cord mesenchymal stem cells: a new era for stem cell therapy. Cell Transplant 24:339–347. https://doi.org/10.3727/096368915X686841
Ding DC, Chou HL, Chang YH et al (2016) Characterization of HLA-G and related immunosuppressive effects in human umbilical cord stroma-derived stem cells. Cell Transplant 25:217–228. https://doi.org/10.3727/096368915X688182
Ding DC, Shyu WC, Lin SZ et al (2012) Human umbilical cord mesenchymal stem cells support nontumorigenic expansion of human embryonic stem cells. Cell Transplant 21:1515–1527. https://doi.org/10.3727/096368912X647199
Wang H, Yan X, Jiang Y et al (2018) The human umbilical cord stem cells improve the viability of OA degenerated chondrocytes. Mol Med Rep 17:4474–4482. https://doi.org/10.3892/mmr.2018.8413
Zhu Y, Guan YM, Huang HL, Wang QS (2014) Human umbilical cord blood mesenchymal stem cell transplantation suppresses inflammatory responses and neuronal apoptosis during early stage of focal cerebral ischemia in rabbits. Acta Pharmacol Sin 35:585–591. https://doi.org/10.1038/aps.2014.9
Min F, Gao F, Li Q, Liu Z (2015) Therapeutic effect of human umbilical cord mesenchymal stem cells modified by angiotensin-converting enzyme 2 gene on bleomycin-induced lung fibrosis injury. Mol Med Rep 11:2387–2396. https://doi.org/10.3892/mmr.2014.3025
Amable PR, Teixeira MVT, Carias RBV et al (2014) Protein synthesis and secretion in human mesenchymal cells derived from bone marrow, adipose tissue and Wharton’s jelly. Stem Cell Res Ther 5:53. https://doi.org/10.1186/scrt442
Chen J, Liu Z, Hong MM et al (2014) Proangiogenic compositions of microvesicles derived from human umbilical cord mesenchymal stem cells. PLoS ONE 16:e115316. https://doi.org/10.1371/journal.pone.0115316
Centeno CJ, Busse D, Kisiday J et al (2008) Increased knee cartilage volume in degenerative joint disease using percutaneously implanted, autologous mesenchymal stem cells, platelet lysate and dexamethasone. Am J Case Rep 11:343–353
Centeno CJ, Busse D, Kisiday J et al (2008) Regeneration of meniscus cartilage in a knee treated with percutaneously implanted autologous mesenchymal stem cells. Med Hypotheses 71:900–908. https://doi.org/10.1016/j.mehy.2008.06.042
Emadedin M, Aghdami N, Taghiyar L et al (2012) Intra-articular injection of autologous mesenchymal stem cells in six patients with knee Osteoarthritis. Arch Iran Med 15:422–428
Kellgren JH, Lawrence JS (1957) Radiological assessment of osteo-arthrosis. Ann Rheum Dis 16:494–502
Murphy JM, Fink DJ, Hunziker EB, Barry FP (2003) Stem cell therapy in a caprine model of osteoarthritis. Arthritis Rheum 48:3464–3474. https://doi.org/10.1002/art.11365
Pawitan JA, Liem IK, Budiyanti E et al (2014) Umbilical cord derived stem cell culture: multiple-harvest explant method. Int J PharmTech Res 6:1202–1208
Dilogo IH, Primaputra MRA, Pawitan JA, Liem IK (2017) Modified Masquelet technique using allogeneic umbilical cord-derived mesenchymal stem cells for infected non-union femoral shaft fracture with a 12 cm bone defect: a case report. Int J Surg Case Rep 34:11–16. https://doi.org/10.1016/j.ijscr.2017.03.002
Litwic A, Edwards MH, Dennison EM, Cooper C (2013) Epidemiology and burden of osteoarthritis. Br Med Bull 105:185–199. https://doi.org/10.1093/bmb/lds038
Kong L, Zheng LZ, Qin L, Ho KKW (2017) Role of mesenchymal stem cells in osteoarthritis treatment. J Orthop Transl 9:89–103. https://doi.org/10.1016/j.jot.2017.03.006
Brittberg M, Lindahl A, Nilsson A et al (1994) Treatment of deep cartilage defects in the knee with autologous chondrocyte transplantation. N Engl J Med 331:889–895. https://doi.org/10.1056/NEJM199410063311401
Martel-Pelletier J, Wildi LM, Pelletier JP (2012) Future therapeutics for osteoarthritis. Bone 51:297–311. https://doi.org/10.1016/j.bone.2011.10.008
Alhadlaq A, Mao JJ (2004) Mesenchymal stem cells: isolation and therapeutics. Stem Cells Dev 13:436–448. https://doi.org/10.1089/scd.2004.13.436
Bruder SP, Fink DJ, Caplan AI (1994) Mesenchymal stem cells in bone development, bone repair, and skeletal regenaration therapy. J Cell Biochem 56:283–294. https://doi.org/10.1002/jcb.240560303
Cha J, Falanga V (2007) Stem cells in cutaneous wound healing. Clin Dermatol 25:73–78. https://doi.org/10.1016/j.clindermatol.2006.10.002
Gangji V, Toungouz M, Hauzeur J-P (2005) Stem cell therapy for osteonecrosis of the femoral head. Expert Opin Biol Ther. https://doi.org/10.1517/14712598.5.4.437
Centeno CJ (2014) Clinical challenges and opportunities of mesenchymal stem cells in musculoskeletal medicine. PM&R 6:70–77. https://doi.org/10.1016/j.pmrj.2013.08.612
Kim S-W, Han H, Chae G-T et al (2006) Successful stem cell therapy using umbilical cord blood-derived multipotent stem cells for Buerger’s disease and ischemic limb disease animal model. Stem Cells 24:1620–1626. https://doi.org/10.1634/stemcells.2005-0365
Baker CD, Abman SH (2013) Umbilical cord stem cell therapy for bronchopulmonary dysplasia: ready for prime time? Thorax 68:402–404. https://doi.org/10.1136/thoraxjnl-2012-202661
Gu Z, Akiyama K, Ma X et al (2010) Transplantation of umbilical cord mesenchymal stem cells alleviates lupus nephritis in MRL/lpr mice. Lupus 19:1502–1514. https://doi.org/10.1177/0961203310373782
Li X, Duan L, Liang Y et al (2016) Human umbilical cord blood-derived mesenchymal stem cells contribute to chondrogenesis in coculture with chondrocytes. Biomed Res Int 2016:3827057. https://doi.org/10.1155/2016/3827057
Deng H, Liao L, Wu J et al (2017) Clinical efficacy of intravesical electrical stimulation on detrusor underactivity: 8 years of experience from a single center. Med (United States) 96:e8020. https://doi.org/10.1097/MD.0000000000008020
Zheng P, Ju L, Jiang B et al (2013) Chondrogenic differentiation of human umbilical cord blood-derived mesenchymal stem cells by co-culture with rabbit chondrocytes. Mol Med Rep 8:1169–1182. https://doi.org/10.3892/mmr.2013.1637
Mennan C, Wright K, Bhattacharjee A et al (2013) Isolation and characterisation of mesenchymal stem cells from different regions of the human umbilical cord. Biomed Res Int 2013:916136. https://doi.org/10.1155/2013/916136
Maumus M, Jorgensen C, Noël D (2013) Mesenchymal stem cells in regenerative medicine applied to rheumatic diseases: role of secretome and exosomes. Biochimie 95:2229–2234. https://doi.org/10.1016/j.biochi.2013.04.017
Dominici M, Le Blanc K, Mueller I et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The international society for cellular therapy position statement. Cytotherapy 8:315–317. https://doi.org/10.1080/14653240600855905
Ding DC, Shyu WC, Chiang MF et al (2007) Enhancement of neuroplasticity through upregulation of β1-integrin in human umbilical cord-derived stromal cell implanted stroke model. Neurobiol Dis 27:339–353. https://doi.org/10.1016/j.nbd.2007.06.010
Wang D, Chen K, Du WT et al (2010) CD14 + monocytes promote the immunosuppressive effect of human umbilical cord matrix stem cells. Exp Cell Res 316:2414–2423. https://doi.org/10.1016/j.yexcr.2010.04.018
Xu S, Liu H, Xie Y et al (2015) Effect of mesenchymal stromal cells for articular cartilage degeneration treatment: a meta-analysis. Cytotherapy 17:1342–1352. https://doi.org/10.1016/j.jcyt.2015.05.005
Vega A, Martín-Ferrero MA, Del Canto F et al (2015) Treatment of knee osteoarthritis with allogeneic bone marrow mesenchymal stem cells: a randomized controlled trial. Transplantation 17:1342–1352. https://doi.org/10.1097/TP.0000000000000678
Gupta PK, Das AK, Chullikana A, Majumdar AS (2012) Mesenchymal stem cells for cartilage repair in osteoarthritis. Stem Cell Res Ther 3:25. https://doi.org/10.1186/scrt116
van Lent PLEM, van den Berg WB (2013) Mesenchymal stem cell therapy in osteoarthritis: advanced tissue repair or intervention with smouldering synovial activation? Arthritis Res Ther 15:112. https://doi.org/10.1186/ar4190
Zhang B, Wang M, Gong A et al (2015) HucMSc-exosome mediated-Wnt4 signaling is required for cutaneous wound healing. Stem Cells 33:2158–2168. https://doi.org/10.1002/stem.1771
Feng C, Luo X, He N et al (2018) Efficacy and persistence of allogeneic adipose-derived mesenchymal stem cells combined with hyaluronic acid in osteoarthritis after intra-articular injection in a sheep model. Tissue Eng Part A 24:219–233. https://doi.org/10.1089/ten.tea.2017.0039
Matas J, Orrego M, Amenabar D et al (2019) Umbilical cord-derived mesenchymal stromal cells (MSCs) for knee osteoarthritis: repeated MSC dosing is superior to a single MSC dose and to hyaluronic acid in a controlled randomized phase I/II trial. Stem Cells Transl Med 8:215–224. https://doi.org/10.1002/sctm.18-0053
O’Hanlon CE, Newberry SJ, Booth M et al (2016) Hyaluronic acid injection therapy for osteoarthritis of the knee: concordant efficacy and conflicting serious adverse events in two systematic reviews. Syst Rev 5:186. https://doi.org/10.1186/s13643-016-0363-9
Acknowledgements
We would like to thank Boenyamin Setiawan, PhD and National Innovation System Research Incentive Program (INSINAS) for funding our research, as well as Tri Kurniawati, BSc for her assistance throughout the grant administration process.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ismail Hadisoebroto Dilogo, Anissa Feby Canintika, Alberto Lastiko Hanitya, Jeanne Adiwinata Pawitan, Isabella Kurnia Liem and Jacub Pandelaki declare that they have no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dilogo, I.H., Canintika, A.F., Hanitya, A.L. et al. Umbilical cord-derived mesenchymal stem cells for treating osteoarthritis of the knee: a single-arm, open-label study. Eur J Orthop Surg Traumatol 30, 799–807 (2020). https://doi.org/10.1007/s00590-020-02630-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00590-020-02630-5