Abstract
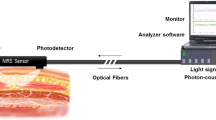
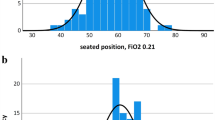
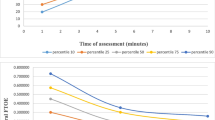
Use of spinal anesthesia (SA) in children may address concerns about potential neurocognitive effects of general anesthesia. We used near-infrared spectroscopy (NIRS) to assess the effects of SA on cerebral and tissue oxygenation in 19 patients aged 7 ± 3 months. Prior to SA placement, NIRS monitors were placed on the forehead (cerebral) and the thigh (tissue). Intraoperative cerebral and tissue saturation were 73 ± 7 and 80 ± 11%, respectively, before SA placement. NIRS measurements were monitored every minute for 30 min after SA placement and modeled using mixed-effects linear regression. Regression estimates showed that cerebral saturation remained stable from 67% [95% confidence interval (CI) 63, 71%] after SA placement to 68% (95% CI 65, 72%) at the conclusion of monitoring. After SA placement, tissue saturation was elevated compared to baseline values; but further change [from 91% (95% CI 89, 93%) to 93% (95% CI 91, 95%) at the end of monitoring] was clinically non-significant. All patients breathed spontaneously on room air without changes in oxygen saturation. Blood pressure and heart rate decreased after SA placement, but no changes in hemodynamic parameters required treatment. These data provide further evidence of the neutral effect of SA on cerebral oxygenation 30 min after block placement.
Similar content being viewed by others
References
Davidson AJ. Anesthesia and neurotoxicity to the developing brain: the clinical relevance. Pediatr Anesth. 2011;21:716–21.
McGowan FX Jr, Davis PJ. Anesthetic-related neurotoxicity in the developing infant: of mice, rats, monkeys and possibly, humans. Anesth Analg. 2008;106:1599–602.
Blaylock M, Engelhardt T, Bissonnette B. Fundamentals of neuronal apoptosis relevant to pediatric anesthesia. Pediatr Anesth. 2010;20:383–95.
Davidson AJ, Morton NS, Arnup SJ, de Graaff JC, Disma N, Withington DE, Frawley G, Hunt RW, Hardy P, Khotcholava M, von Ungern Sternberg BS, Wilton N, Tuo P, SalvoI I, Ormond G, Stargatt R, Locatelli BG, McCann ME, The GAS Consortium. Apnea after awake-regional and general anesthesia in infants: The General Anesthesia compared to Spinal anesthesia (GAS) study: comparing apnea and neurodevelopmental outcomes, a randomized controlled trial. Anesthesiology. 2015;123:38–54.
Dohi S, Naito H, Takahashi T. Age-related changes in blood pressure and duration of motor blockade in spinal anesthesia. Anesthesiology. 1979;50:319–23.
Finkel JC, Boltz MG, Conran AM. Hemodynamic changes during high spinal anesthesia in children undergoing open heart surgery. Paediatr Anaesth. 2003;13:48–52.
Kachko L, Birk E, Simhi E, Tzeitlin E, Freud E, Katz J. Spinal anesthesia for noncardiac surgery in infants with congenital heart diseases. Paediatr Anaesth. 2012;22:647–53.
Payen D, Ecoffey C, Carli P, Dubousset AM. Pulsed Doppler ascending aortic, carotid, brachial, and femoral artery blood flows during caudal anesthesia in infants. Anesthesiology. 1987;67:681–5.
McCann ME, Withington DE, Arnup SJ, Davidson AJ, Disma N, Frawley G, Morton NS, Bell G, Hunt RW, Bellinger DC, Polaner DM, Leo A, Absalom AR, von Ungern-Sternberg BS, Izzo F, Szmuk P, Young V, Soriano SG, de Graaff JC; GAS Consortium. Differences in blood pressure in infants after general anesthesia compared to awake regional anesthesia (GAS study−a prospective randomized trial). Anesth Analg. 2017;125(3):837–45.
Bonnet MP, Larousse E, Asehnoune K, Benhamou D. Spinal anesthesia with bupivacaine decreases cerebral blood flow in former preterm infants. Anesth Analg. 2004;98:1280–3.
Jöbsis FF. Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science. 1977;198:1264–7.
Tobias JD. Cerebral oxygenation monitoring: near infrared spectroscopy. Expert Rev Med Devices. 2006;3:235–43.
Neshat Vahid S, Panisello JM. The state of affairs of neurologic monitoring by near-infrared spectroscopy in pediatric cardiac critical care. Curr Opin Pediatr. 2014;26:299–303.
Chakravarti SB, Mittnacht AJ, Katz JC, Nguyen K, Joashi U, Srivastava S. Multisite near-infrared spectroscopy predicts elevated blood lactate level in children after cardiac surgery. J Cardiothorac Vasc Anesth. 2009;23:663–7.
Sood ED, Benzaquen JS, Davies RR, Woodford E, Pizarro C. Predictive value of perioperative near-infrared spectroscopy for neurodevelopmental outcomes after cardiac surgery in infancy. J Thorac Cardiovasc Surg. 2013;145:438–45.
Hoffman GM, Brosig CL, Bear LM, Tweddell JS, Mussatto KA. Effect of intercurrent operation and cerebral oxygenation on developmental trajectory in congenital heart disease. Ann Thorac Surg. 2016;101:708–16.
Kondo Y, Sakatani K, Hirose N, Maeda T, Kato J, Ogawa S, Katayama Y. Effect of spinal anesthesia for elective cesarean section on cerebral blood oxygenation changes: comparison of hyperbaric and isobaric bupivacaine. Adv Exp Med Biol. 2013;765:109–14.
Hirose N, Kondo Y, Maeda T, Suzuki T, Yoshino A, Katayama Y. Oxygen Supplementation is effective in attenuating maternal cerebral blood deoxygenation after spinal anesthesia for cesarean section. Adv Exp Med Biol. 2016;876:471–7.
Sola C, Hertz L, Bringuier S, De La Arena P, Macq C, Deziel-Malouin S, Raux O, Dadure C. Spinal anaesthesia in neonates and infants: what about the cerebral saturation? Br J Anaesth 2017;119:964–71.
Koch HW, Hansen TG. Perioperative use of cerebral and renal near-infrared spectroscopy in neonates: a 24-h observational study. Paediatr Anaesth. 2016;26:190–8.
Olbrecht VA, Skowno J, Marchesini V, Ding L, Jiang Y, Ward CG, Yu G, Liu H, Schurink B, Vutskits L, de Graaff JC, McGowan FX Jr, von Ungern-Sternberg BS, Kurth CD, Davidson A. An international, multicenter, observational study of cerebral oxygenation during infant and neonatal anesthesia. Anesthesiology 2018;128:85–96.
Razlevice I, Rugyte DC, Strumylaite L, Macas A. Assessment of risk factors for cerebral oxygen desaturation during neonatal and infant general anesthesia: an observational, prospective study. BMC Anesthesiol. 2016;16:107.
Conforti A, Giliberti P, Mondi V, Valfré L, Sgro S, Picardo S, Bagolan P, Dotta A. Near infrared spectroscopy: experience on esophageal atresia infants. J Pediatr Surg. 2014;49:1064–8.
Whitaker EE, Wiemann BZ, DaJusta DG, Alpert SA, Ching CB, McLeod DJ, Tobias JD, Jayanthi VR. Spinal anesthesia for pediatric urological surgery: reducing the theoretic neurotoxic effects of general anesthesia. J Pediatr Urol. 2017;13:396–400.
Murkin JM, Arango M. Near-infrared spectroscopy as an index of brain and tissue oxygenation. Br J Anaesth. 2009;103(Suppl 1):i3–13.
Bettesworth J, Bhalla T, Barry N, Tobias JD. Changes in tissue oxygenation following caudal epidural blockade in infants and children. Paediatr Anaesth. 2012;22:1068–71.
Kabon B, Fleischmann E, Treschan T, Taguchi A, Kapral S, Kurz A. Thoracic epidural anesthesia increases tissue oxygenation during major abdominal surgery. Anesth Analg. 2003;97:1812–7.
Tighe PJ, Elliott CE, Lucas SD, Boezaart AP. Noninvasive tissue oxygen saturation determined by near-infrared spectroscopy following peripheral nerve block. Acta Anaesthesiol Scand. 2011;55:1239–46.
Tobias JD, Hoernschemeyer DG. Near-infrared spectroscopy identifies compartment syndrome in an infant. J Pediatr Orthop. 2007;27:311–3.
Funding
The study was funded by departmental resources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics
Institutional Review Board approval at Nationwide Children’s Hospital (Columbus, OH, USA) number IRB16-00465 obtained on June 17, 2016.
Conflict of interest
The authors have no conflict of interest to report.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Froyshteter, A.B., Tumin, D., Whitaker, E.E. et al. Changes in tissue and cerebral oxygenation following spinal anesthesia in infants: a prospective study. J Anesth 32, 288–292 (2018). https://doi.org/10.1007/s00540-017-2446-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-017-2446-8