Abstract
Purpose
To explore improvement in motor ability, function, health-related quality of life (HRQOL), and symptom severity in patients with sclerotic chronic graft-versus-host disease (ScGVHD) in response to treatment as well as the relationship among changes on such measures.
Methods
This study was a secondary analysis of data from 13 individuals with severe ScGVHD enrolled in a clinical trial evaluating the efficacy of imatinib mesylate (clinicaltrials.gov identifier: NCT00702689). Self-reported, clinician-reported, and performance-based indicators of motor ability, function, HRQOL, and symptom severity were assessed at baseline and 6 months following the administration of imatinib mesylate.
Results
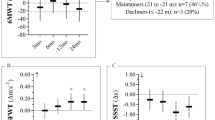
Participants did not show statistically significant improvement on any measures over time. Approximately one-third of patients displayed clinically significant improvement on measures of motor ability (palmar pinch strength, dominant hand, 30.8%), functioning (Manual Ability Measure—36, 41.7%), HRQOL (Short Form 36 [SF-36] Mental Component Summary, 33.3%), and symptom severity (Lee Symptom Scale, 38.5%). Improvement in cGVHD symptom burden was correlated with improvement in function (Assessment of Motor and Process Skills [AMPS] and Disabilities of the Arm, Shoulder, and Hand [DASH] scores) and HRQOL (SF-36 Physical Component Summary scores).
Conclusions
Findings suggest the potential utility of administering patient-reported and performance-based functional measures, such as the DASH and the AMPS, to patients with cGVHD. By understanding the functional consequences of ScGVHD, interdisciplinary teams of health care providers, including rehabilitation professionals, can work to improve long-term outcomes.


Similar content being viewed by others
References
Niederwieser D, Baldomero H, Szer J, Gratwohl M, Aljurf M, Atsuta Y, Bouzas LF, Confer D, Greinix H, Horowitz M, Iida M, Lipton J, Mohty M, Novitzky N, Nunez J, Passweg J, Pasquini MC, Kodera Y, Apperley J, Seber A, (WBMT) AGftWNoBaMT (2016) Hematopoietic stem cell transplantation activity worldwide in 2012 and a SWOT analysis of the Worldwide Network for Blood and Marrow Transplantation Group including the global survey. Bone Marrow Transplant 51:778–785. https://doi.org/10.1038/bmt.2016.18
Bhatia S, Francisco L, Carter A, Sun C-L, Baker KS, Gurney JG, McGlave PB, Nademanee A, O’Donnell M, Ramsay NKC, Robison LL, Snyder D, Stein A, Forman SJ, Weisdorf DJ (2007) Late mortality after allogeneic hematopoietic cell transplantation and functional status of long-term survivors: report from the Bone Marrow Transplant Survivor Study. Blood 110:3784–3792. https://doi.org/10.1182/blood-2007-03-082933
Fraser CJ, Bhatia S, Ness K, Carter A, Francisco L, Arora M, Parker P, Forman S, Weisdorf D, Gurney JG, Baker KS (2006) Impact of chronic graft-versus-host disease on the health status of hematopoietic cell transplantation survivors: a report from the Bone Marrow Transplant Survivor Study. Blood 108:2867–2873. https://doi.org/10.1182/blood-2006-02-003954
Sun C-L, Kersey JH, Francisco L, Armenian SH, Baker KS, Weisdorf DJ, Forman SJ, Bhatia S (2013) Burden of morbidity in 10+ year survivors of hematopoietic cell transplantation: report from the Bone Marrow Transplantation Survivor Study. Biol Blood Marrow Transplant 19:1073–1080. https://doi.org/10.1016/j.bbmt.2013.04.002
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, Palmer J, Weisdorf D, Treister NS, Cheng GS, Kerr H, Stratton P, Duarte RF, McDonald GB, Inamoto Y, Vigorito A, Arai S, Datiles MB, Jacobsohn D, Heller T, Kitko CL, Mitchell SA, Martin PJ, Shulman H, Wu RS, Cutler CS, Vogelsang GB, Lee SJ, Pavletic SZ, Flowers ME (2015) National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. the 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 21:389–401. https://doi.org/10.1016/j.bbmt.2014.12.001
Lee SJ, Onstad L, Chow EJ, Shaw BE, Jim HSL, Syrjala KL, Baker KS, Buckley S, Flowers ME (2018) Patient-reported outcomes and health status associated with chronic graft-versus-host disease. Haematologica 103:1535–1541. https://doi.org/10.3324/haematol.2018.192930
Pidala J, Kurland BF, Chai X, Vogelsang G, Weisdorf DJ, Pavletic S, Cutler C, Majhail N, Lee SJ (2011) Sensitivity of changes in chronic graft-versus-host disease activity to changes in patient-reported quality of life: results from the Chronic Graft-versus-Host Disease Consortium. Haematologica 96:1528–1535. https://doi.org/10.3324/haematol.2011.046367
Detrait MY, Morisset S, Peffault de Latour R, Yakoub-Agha I, Crocchiolo R, Tabrizi R, Bay J-O, Chevalier P, Barraco F, Raus N, Vigouroux S, Magro L, Mohty M, Milpied N, Blaise D, Socié G, Michallet M (2015) Pre-transplantation risk factors to develop sclerotic chronic GvHD after allogeneic HSCT: a multicenter retrospective study from the Société Française de Greffe de Moelle et de Thérapie Cellulaire (SFGM-TC). Bone Marrow Transplant 50:253–258. https://doi.org/10.1038/bmt.2014.244
Skert C, Patriarca F, Sperotto A, Cerno M, Filì C, Zaja F, Stocchi R, Geromin A, Damiani D, Fanin R (2006) Sclerodermatous chronic graft-versus-host disease after allogeneic hematopoietic stem cell transplantation: incidence, predictors and outcome. Haematologica 91:258–261
Uhm J, Hamad N, Shin EM, Michelis FV, Shanavas M, Gupta V, Kuruvilla J, Lipton JH, Messner HA, Seftel M, Kim D (2014) Incidence, risk factors, and long-term outcomes of sclerotic graft-versus-host disease after allogeneic hematopoietic cell transplantation. Biol Blood Marrow Transplant 20:1751–1757. https://doi.org/10.1016/j.bbmt.2014.07.001
Martires KJ, Baird K, Steinberg SM, Grkovic L, Joe GO, Williams KM, Mitchell SA, Datiles M, Hakim FT, Pavletic SZ, Cowen EW (2011) Sclerotic-type chronic GVHD of the skin: clinical risk factors, laboratory markers, and burden of disease. Blood 118:4250–4257. https://doi.org/10.1182/blood-2011-04-350249
Baird K, Steinberg SM, Grkovic L, Pulanic D, Cowen EW, Mitchell SA, Williams KM, Datiles MB, Bishop R, Bassim CW, Mays JW, Edwards D, Cole K, Avila DN, Taylor T, Urban A, Joe GO, Comis LE, Berger A, Stratton P, Zhang D, Shelhamer JH, Gea-Banacloche JC, Sportes C, Fowler DH, Gress RE, Pavletic SZ (2013) National Institutes of Health chronic graft-versus-host disease staging in severely affected patients: organ and global scoring correlate with established indicators of disease severity and prognosis. Biol Blood Marrow Transplant 19:632–639. https://doi.org/10.1016/j.bbmt.2013.01.013
Carpenter PA, Kitko CL, Elad S, Flowers MED, Gea-Banacloche JC, Halter JP, Hoodin F, Johnston L, Lawitschka A, McDonald GB, Opipari AW, Savani BN, Schultz KR, Smith SR, Syrjala KL, Treister N, Vogelsang GB, Williams KM, Pavletic SZ, Martin PJ, Lee SJ, Couriel DR (2015) National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: V. the 2014 Ancillary Therapy and Supportive Care Working Group report. Biol Blood Marrow Transplant 21:1167–1187. https://doi.org/10.1016/j.bbmt.2015.03.024
Lee SJ, Wolff D, Kitko C, Koreth J, Inamoto Y, Jagasia M, Pidala J, Olivieri A, Martin PJ, Przepiorka D, Pusic I, Dignan F, Mitchell SA, Lawitschka A, Jacobsohn D, Hall AM, Flowers MED, Schultz KR, Vogelsang G, Pavletic S (2015) Measuring therapeutic response in chronic graft-versus-host disease. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: IV. The 2014 Response Criteria Working Group report. Biol Blood Marrow Transplant 21:984–999. https://doi.org/10.1016/j.bbmt.2015.02.025
Curtis LM, Grkovic L, Mitchell SA, Steinberg SM, Cowen EW, Datiles MB, Mays J, Bassim C, Joe G, Comis LE, Berger A, Avila D, Taylor T, Pulanic D, Cole K, Baruffaldi J, Fowler DH, Gress RE, Pavletic SZ (2014) NIH response criteria measures are associated with important parameters of disease severity in patients with chronic GVHD. Bone Marrow Transplant 49:1513–1520. https://doi.org/10.1038/bmt.2014.188
Pidala J, Chai X, Martin P, Inamoto Y, Cutler C, Palmer J, Weisdorf D, Pavletic S, Arora M, Jagasia M, Jacobsohn D, Lee SJ (2013) Hand grip strength and 2-minute walk test in chronic graft-versus-host disease assessment: analysis from the Chronic GVHD Consortium. Biol Blood Marrow Transplant 19:967–972. https://doi.org/10.1016/j.bbmt.2013.03.014
Gordon J, Spiera R (2011) Imatinib and the treatment of fibrosis: recent trials and tribulations. Curr Rheumatol Rep 13(1):51–58. https://doi.org/10.1007/s11926-010-0146-6
Arai S, Pidala J, Pusic I, Chai X, Jaglowski S, Khera N, Palmer J, Chen GL, Jagasia MH, Mayer SA, Wood WA, Green M, Hyun TS, Inamoto Y, Storer BE, Miklos DB, Shulman HM, Martin PJ, Sarantopoulos S, Lee SJ, Flowers MED (2016) A randomized phase II crossover study of imatinib or rituximab for cutaneous sclerosis after hematopoietic cell transplantation. Clin Cancer Res 22:319–327. https://doi.org/10.1158/1078-0432.CCR-15-1443
Baird K, Comis LE, Joe GO, Steinberg SM, Hakim FT, Rose JJ, Mitchell SA, Pavletic SZ, Figg WD, Yao L, Flanders KC, Takebe N, Sarantopoulos S, Booher S, Cowen EW (2015) Imatinib mesylate for the treatment of steroid-refractory sclerotic-type cutaneous chronic graft-versus-host disease. Biol Blood Marrow Transplant 21(6):1083–1090. https://doi.org/10.1016/j.bbmt.2015.03.006
Magro L, Mohty M, Catteau B, Coiteux V, Chevallier P, Terriou L, Jouet J-P, Yakoub-Agha I (2009) Imatinib mesylate as salvage therapy for refractory sclerotic chronic graft-versus-host disease. Blood 114:719–722. https://doi.org/10.1182/blood-2009-02-204750
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, Martin P, Chien J, Przepiorka D, Couriel D, Cowen EW, Dinndorf P, Farrell A, Hartzman R, Henslee-Downey J, Jacobsohn D, McDonald G, Mittleman B, Rizzo JD, Robinson M, Schubert M, Schultz K, Shulman H, Turner M, Vogelsang G, Flowers MED (2005) National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 11:945–956. https://doi.org/10.1016/j.bbmt.2005.09.004
American Academy of Orthopaedic Surgeons (1965) Joint motion: method of measuring and recording. American Academy of Orthopaedic Surgeons, Chicago
Jebsen RH, Taylor N, Trieschmann RB, Trotter MJ, Howard LA (1969) An objective and standardized test of hand function. Arch Phys Med Rehabil 50:311–319
Trites RL (1977) Grooved pegboard test – neuropsychological test manual. Royal Ottawa Hospital, Ottawa
Waters RL, Lunsford BR, Perry J, Byrd R (1988) Energy speed relationship of walking - standard tables. J Orthop Res 6:215–222. https://doi.org/10.1002/jor.1100060208
Mathiowetz V, Weber K, Volland G, Kashman N (1984) Reliability and validity of grip and pinch strength evaluations. J Hand Surg 9:222–226
Hudak PL, Amadio PC, Bombardier C, Upper Extremity Collaborative Group (1996) Development of an upper extremity outcome measure: the DASH (disabilities of the arm, shoulder, and head). Am J Ind Med 29:602–608. https://doi.org/10.1002/(Sici)1097-0274(199606)29:6<602::Aid-Ajim4>3.0.Co;2-L
Daughton DM, Fix AJ, Kass I, Bell CW, Patil KD (1982) Maximum oxygen consumption and the ADAPT quality-of-life scale. Arch Phys Med Rehabil 63:620–622
Herzberg PY, Heussner P, Mumm FHA, Horak M, Hilgendorf I, von Harsdorf S, Hemmati P, Rieger K, Greinix H, Freund M, Lee SJ, Holler E, Wolff D (2010) Validation of the human activity profile questionnaire in patients after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 16:1707–1717. https://doi.org/10.1016/j.bbmt.2010.05.018
Fisher AG, Jones KB (2011) Assessment of motor and process skills: volume 1 - development, standardization, and administration manual. 7th, Revised edn. Three Star Press, Inc, Fort Collins
Chen CC, Bode RK (2010) Psychometric validation of the manual ability Measure-36 (MAM-36) in patients with neurologic and musculoskeletal disorders. Arch Phys Med Rehabil 91:414–420. https://doi.org/10.1016/j.apmr.2009.11.012
Ware JE, Snow KK, Kosinski M, Gandek B, NEMCH (1993) SF-36 health survey: manual and interpretation guide. Health Institute, New England Medical Center, Boston
Lee SJ, Cook EF, Soiffer R, Antin JH (2002) Development and validation of a scale to measure symptoms of chronic graft-versus-host disease. Biol Blood Marrow Transplant 8:444–452. https://doi.org/10.1053/bbmt.2002.v8.pm12234170
Cleeland CS, Mendoza TR, Wang XS, Chou C, Harle MT, Morrissey M, Engstrom MC (2000) Assessing symptom distress in cancer patients: the MD Anderson Symptom Inventory. Cancer 89:1634–1646
Kennedy C, Beaton DE, Soloway S, McConnell S, Bombardier C (2011) The DASH and QuickDASH outcome measure User’s Manual, 3rd edn. Institute for Work & Health, Toronto
Abo S, Ritchie D, Denehy L, Panek-Hudson Y, Granger CL (2018) A hospital and home-based exercise program to address functional decline in people following allogeneic stem cell transplantation. Support Care Cancer 26:1727–1736. https://doi.org/10.1007/s00520-017-4016-x
Inamoto Y, Martin PJ, Chai X, Jagasia M, Palmer J, Pidala J, Cutler C, Pavletic SZ, Arora M, Jacobsohn D, Carpenter PA, Flowers MED, Khera N, Vogelsang GB, Weisdorf D, Storer BE, Lee SJ (2012) Clinical benefit of response in chronic graft-versus-host disease. Biol Blood Marrow Transplant 18:1517–1524. https://doi.org/10.1016/j.bbmt.2012.05.016
Inamoto Y, Pidala J, Chai X, Kurland BF, Weisdorf D, Flowers MED, Palmer J, Arai S, Jacobsohn D, Cutler C, Jagasia M, Goldberg JD, Martin PJ, Pavletic SZ, Vogelsang GB, Lee SJ, Carpenter PA (2014) Assessment of joint and fascia manifestations in chronic graft-versus-host disease. Arthritis Rheumatol 66:1044–1052. https://doi.org/10.1002/art.38293
Mitchell SA, Leidy NK, Mooney KH, Dudley WN, Beck SL, LaStayo PC, Cowen EW, Palit P, Comis LE, Krumlauf MC, Avila DN, Atlam N, Fowler DH, Pavletic SZ (2010) Determinants of functional performance in long-term survivors of allogeneic hematopoietic stem cell transplantation with chronic graft-versus-host disease (cGVHD). Bone Marrow Transplant 45:762–769
Bevans MF, Mitchell SA, Barrett JA, Bishop MR, Childs R, Fowler D, Krumlauf M, Prince P, Shelburne N, Wehrlen L, Yang L (2014) Symptom distress predicts long-term health and well-being in allogeneic stem cell transplantation survivors. Biol Blood Marrow Transplant 20:387–395. https://doi.org/10.1016/j.bbmt.2013.12.001
Bevans MF, Mitchell SA, Marden S (2008) The symptom experience in the first 100 days following allogeneic hematopoietic stem cell transplantation (HSCT). Support Care Cancer 16:1243–1254
Pillay B, Lee SJ, Katona L, Burney S, Avery S (2014) Psychosocial factors predicting survival after allogeneic stem cell transplant. Support Care Cancer 22:2547–2555. https://doi.org/10.1007/s00520-014-2239-7
Raven EE, Haverkamp D, Sierevelt IN, Montfoort DO, Pöll RG, Blankevoort L, Tak PP (2008) Construct validity and reliability of the disability of arm, shoulder and hand questionnaire for upper extremity complaints in rheumatoid arthritis. J Rheumatol 35(12):2334–2338. https://doi.org/10.3899/jrheum.080067
Simmonds MJ (2002) Physical function in patients with cancer: psychometric characteristics and clinical usefulness of a physical performance test battery. J Pain Symptom Manag 24(4):404–414
Holm LV, Hansen DG, Johansen C, Vedsted P, Larsen PV, Kragstrup J, Sondergaard J (2012) Participation in cancer rehabilitation and unmet needs: a population-based cohort study. Support Care Cancer 20:2913–2924. https://doi.org/10.1007/s00520-012-1420-0
Acknowledgments
We thank the patients who participated in this study for their contributions to our growing understanding of sclerotic chronic graft-versus-host disease. We also thank the NIH Chronic GVHD Multidisciplinary Team, the research nurses, Christiana Booher, and Bonnie Hodsdon for their respective contributions to this project. We thank the NIH Fellows Editorial Board for their editorial assistance.
Funding
This research was supported by the National Institutes of Health Intramural Research Programs and the National Cancer Institute’s NCT00702689.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The protocol was approved by the National Cancer Institute Institutional Review Board.
Conflict of interest
The authors declare that they have no conflict of interest.
Disclaimer
This article reflects the views of the authors and should not be construed to represent FDA’s views or policies.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 28 kb)
Rights and permissions
About this article
Cite this article
Rosenthal, E.A., Ho, PS., Joe, G.O. et al. Motor ability, function, and health-related quality of life as correlates of symptom burden in patients with sclerotic chronic graft-versus-host disease receiving imatinib mesylate. Support Care Cancer 28, 3679–3689 (2020). https://doi.org/10.1007/s00520-019-05207-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-019-05207-z