Abstract
Purpose
Purpose of this study was to determine the impact of Oral Mucositis (OM) on health-related quality of life (HRQoL) and quality of life associated symptoms and functions in patients undergoing hematopoietic stem cell transplantation (HSCT).
Methods
Prospective, non-interventional single-center observational study at a German tertiary teaching hospital. Inpatient allogenic and autologous stem cell transplant patients ≥18-year-old with high-dose chemotherapy. OM was assessed with the WHO Oral Toxicity Scale, pain according to the Numeric Rating Scale (NRS) and the performance status using the ECOG Score. QOL was captured with the EORTC QLQ-C30 and the QLQ-OH15 questionnaires.
Results
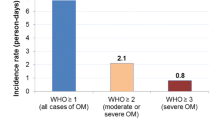
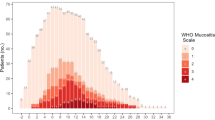
Forty-five stem cell transplant patients (20 autologous, 25 allogenic) were enrolled between August 2016 and February 2017. Twenty-six (58%, 95% CI: 42% - 72%) patients developed OM (10 grade I, 4 grade II, 8 grade III, 4 grade IV). OM affected patients suffered more from pain, sore mouth and sensitive mouth. A lower physical functioning (34.5 vs 7.5, p = 0.003) and a lower oral health-related quality of life (24.3 vs 7.7, p = 0.006) was found in patients with OM development. There was found a positive correlation between the grade of OM and the NRS-value (r = 0.93, 95% CI: 0.89–0.96, p < 0.001).
Conclusion
OM is associated with health-related quality of life and quality of life associated functions and symptoms. More research should be performed to find ways to prevent OM and to stabilize patients’ quality of life during HSCT.


Similar content being viewed by others
References
Bellm LA, Epstein JB, Rose-Ped A, Martin P, Fuchs HJ (2000) Patient reports of complications of bone marrow transplantation. Support Care Cancer : Off J Multinatl Assoc Support Care Cancer 8(1):33–39
Elting LS, Cooksley C, Chambers M, Cantor SB, Manzullo E, Rubenstein EB (2003) The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer 98(7):1531–1539. https://doi.org/10.1002/cncr.11671
Lalla RV, Peterson DE (2005) Oral mucositis. Dent Clin N Am 49 (1):167-184. doi:doi:https://doi.org/10.1016/j.cden.2004.07.009
Woo SB, Sonis ST, Monopoli MM, Sonis AL (1993) A longitudinal study of oral ulcerative mucositis in bone marrow transplant recipients. Cancer 72(5):1612–1617. https://doi.org/10.1002/1097-0142(19930901)72:5<1612::AID-CNCR2820720520>3.0.CO;2-Q
Wardley AM, Jayson GC, Swindell R, Morgenstern GR, Chang J, Bloor R, Fraser CJ, Scarffe JH (2000) Prospective evaluation of oral mucositis in patients receiving myeloablative conditioning regimens and haemopoietic progenitor rescue. Br J Haematol 110(2):292–299. https://doi.org/10.1046/j.1365-2141.2000.02202.x
Jones JA, Qazilbash MH, Shih YC, Cantor SB, Cooksley CD, Elting LS (2008) In-hospital complications of autologous hematopoietic stem cell transplantation for lymphoid malignancies: clinical and economic outcomes from the nationwide inpatient sample. Cancer 112(5):1096–1105. https://doi.org/10.1002/cncr.23281
Martino M, Montanari M, Ferrara F, Ciceri F, Scortechini I, Palmieri S, Marktel S, Cimminiello M, Messina G, Irrera G, Offidani M, Console G, Castagna L, Milone G, Bruno B, Tripepi G, Lemoli RM, Olivieri A Very low rate of readmission after an early discharge outpatient model for autografting in multiple myeloma patients: an Italian multicenter retrospective study. Biol Blood Marrow Transplant 20(7):1026–1032. https://doi.org/10.1016/j.bbmt.2014.03.027
Spielberger R, Stiff P, Bensinger W, Gentile T, Weisdorf D, Kewalramani T, Shea T, Yanovich S, Hansen K, Noga S, McCarty J, LeMaistre CF, Sung EC, Blazar BR, Elhardt D, Chen MG, Emmanouilides C (2004) Palifermin for oral mucositis after intensive therapy for hematologic cancers. N Engl J Med 351(25):2590–2598. https://doi.org/10.1056/NEJMoa040125
Sakellari I, Angelopoulou M, Tsopra O, Dervenoulas I, Tsirigotis P, Spyridonidis A, Liga M, Tsionos K, Anargyrou K, Pouli A, Anagnostopoulos A (2015) A prospective study of incidence, clinical and quality of life consequences of oral mucositis post palifermin prophylaxis in patients undergoing high-dose chemotherapy and autologous hematopoietic cell transplantation. Ann Hematol 94(10):1733–1740. https://doi.org/10.1007/s00277-015-2437-5
Kim KI, Kim JW, Lee HJ, Kim BS, Bang SM, Kim I, JM O, Yoon SS, Lee JS, Park S, Kim BK (2013) Recombinant human epidermal growth factor on oral mucositis induced by intensive chemotherapy with stem cell transplantation. Am J Hematol 88(2):107–112. https://doi.org/10.1002/ajh.23359
Martinez JM, Pereira D, Chacim S, Mesquita E, Sousa I, Martins A, Azevedo T, Mariz JM (2014) Mucositis care in acute leukemia and non-Hodgkin lymphoma patients undergoing high-dose chemotherapy. Support Care Cancer : Off J Multinatl Assoc Support Care in Cancer 22(9):2563–2569. https://doi.org/10.1007/s00520-014-2199-y
DRST (2016) Jahresbericht 2015. Deutsches Register für Stammzelltransplantation e.V.,
Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P, Duarte RF, Dufour C, Kuball J, Farge-Bancel D, Gennery A, Kroger N, Lanza F, Nagler A, Sureda A, Mohty M (2016) Hematopoietic stem cell transplantation in Europe 2014: more than 40 000 transplants annually. Bone Marrow Transplant 51(6):786–792. https://doi.org/10.1038/bmt.2016.20
World Health Organization (1979) Handbook for reporting results of cancer treatment. World Health Organization, Geneva
Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, Carbone PP (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5(6):649–655. https://doi.org/10.1097/00000421-198212000-00014
McCaffery M, Beebe, A. (1993) Pain: clinical manual for nursing practice. Baltimore: VV Mosby Company 9 (11):55. doi:https://doi.org/10.7748/ns.9.11.55.s69
Aaronson NKAS, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner HFS, de Haes JCJM, Kaasa S, Klee MC, Osoba D, Razavi D, Rofe PBSS, Sneeuw KCA, Sullivan M, Takeda F (1993) The European Organisation for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85(5):365–376. https://doi.org/10.1093/jnci/85.5.365
Shih CL, Chen CH, Sheu CF, Lang HC, Hsieh CL (2013) Validating and improving the reliability of the EORTC qlq-c30 using a multidimensional Rasch model. Value Health : J Int Soc Pharmacoecon Outcomes Res 16(5):848–854. https://doi.org/10.1016/j.jval.2013.05.004
Hjermstad MJ, Bergenmar M, Bjordal K, Fisher SE, Hofmeister D, Montel S, Nicolatou-Galitis O, Pinto M, Raber-Durlacher J, Singer S, Tomaszewska IM, Tomaszewski KA, Verdonck-de Leeuw I, Yarom N, Winstanley JB, Herlofson BB (2016) International field testing of the psychometric properties of an EORTC quality of life module for oral health: the EORTC QLQ-OH15. Support Care Cancer : Off J Multinatl Assoc Support Care Cancer 24(9):3915–3924. https://doi.org/10.1007/s00520-016-3216-0
Fayers PMAN, Bjordal K, Groenvold M, Curran D, Bottomley A (2001) The EORTC QLQ-C30 scoring manual, 3rd edn. European Organisation for Research and Treatment of Cancer, Brussels
Giesinger JM, Kieffer JM, Fayers PM, Groenvold M, Petersen MA, Scott NW, Sprangers MA, Velikova G, Aaronson NK (2016) Replication and validation of higher order models demonstrated that a summary score for the EORTC QLQ-C30 is robust. J Clin Epidemiol 69:79–88. https://doi.org/10.1016/j.jclinepi.2015.08.007
Xing SZ, Zhang Y (2015) Efficacy and safety of transdermal fentanyl for the treatment of oral mucositis pain caused by chemoradiotherapy in patients with esophageal squamous cell carcinoma. Support Care Cancer : Off J Multinatl Assocof Support Care Cancer 23(3):753–759. https://doi.org/10.1007/s00520-014-2419-5
Blijlevens N, de Chateau M, Krivan G, Rabitsch W, Szomor A, Pytlik R, Lissmats A, Johnsen HE, de Witte T, Einsele H, Ruutu T, Niederwieser D (2013) In a high-dose melphalan setting, palifermin compared with placebo had no effect on oral mucositis or related patient’s burden. Bone Marrow Transplant 48(7):966–971. https://doi.org/10.1038/bmt.2012.257
Kim JW, Cha Y, Kim SJ, Han SW, DY O, Lee SH, Kim DW, Im SA, Kim TY, Heo DS, Bang YJ (2012) Association of oral mucositis with quality of life and symptom clusters in patients with solid tumors receiving chemotherapy. Support Care Cancer : Off J Multinatl Assoc Support Care Cancer 20(2):395–403. https://doi.org/10.1007/s00520-011-1126-8
Elting LS, Keefe DM, Sonis ST, Garden AS, Spijkervet FK, Barasch A, Tishler RB, Canty TP, Kudrimoti MK, Vera-Llonch M (2008) Patient-reported measurements of oral mucositis in head and neck cancer patients treated with radiotherapy with or without chemotherapy: demonstration of increased frequency, severity, resistance to palliation, and impact on quality of life. Cancer 113(10):2704–2713. https://doi.org/10.1002/cncr.23898
Cheng KK (2007) Oral mucositis and quality of life of Hong Kong Chinese patients with cancer therapy. Eur J Oncol Nurs : Off J Eur Oncol Nurs Soc 11(1):36–42. https://doi.org/10.1016/j.ejon.2006.03.004
Ohrn KE, Sjoden PO, Wahlin YB, Elf M (2001) Oral health and quality of life among patients with head and neck cancer or haematological malignancies. Support Care Cancer : Off J Multinatl Assoc Support Care Cancer 9(7):528–538. https://doi.org/10.1007/s005200100253
Antunes HS, Herchenhorn D, Small IA, Araujo CM, Viegas CM, Cabral E, Rampini MP, Rodrigues PC, Silva TG, Ferreira EM, Dias FL, Ferreira CG (2013) Phase III trial of low-level laser therapy to prevent oral mucositis in head and neck cancer patients treated with concurrent chemoradiation. Radiother Oncol : J Eur Soc Ther Radiol Oncol 109(2):297–302. https://doi.org/10.1016/j.radonc.2013.08.010
Elting LS, Chang YC, Parelkar P, Boers-Doets CB, Michelet M, Hita G, Rouleau T, Cooksley C, Halm J, Vithala M, Bossi P, Escalante C, Brennan MT (2013) Risk of oral and gastrointestinal mucosal injury among patients receiving selected targeted agents: a meta-analysis. Support Care Cancer : Off J Multinatl Assoc Support Care Cancer 21(11):3243–3254. https://doi.org/10.1007/s00520-013-1821-8
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Staudenmaier, T., Cenzer, I., Crispin, A. et al. Burden of oral mucositis in stem cell transplant patients—the patients’ perspective. Support Care Cancer 26, 1577–1584 (2018). https://doi.org/10.1007/s00520-017-4000-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-017-4000-5