Summary
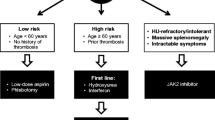
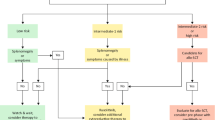
Polycythemia vera (PV) is a clonal disease arising from hematopoietic stem cells. Erythrocytosis is the hallmark of the disease but leukocytosis, thrombocytosis and splenomegaly may also be present. Thromboembolic complications occur in about 20% of patients. Circulatory disturbances as well as pruritus represent frequent symptoms of the disease. Mutations in the JAK2 gene are present in 95% of patients in exon 14 (V617F) and in 3% in exon 12. The main goal of the treatment for patients with PV is the prevention of thromboembolic events, transformation to myelofibrosis and acute myeloid leukemia. Interferon alpha and hydroxyurea are used as first-line treatment for high risk patients. For patients unresponsive to first-line therapy ruxolitinib is available.

Similar content being viewed by others
References
Tefferi A, Rumi E, Finazzi G, Gisslinger H, Vannucchi AM, Rodeghiero F, et al. Survival and prognosis among 1545 patients with contemporary polycythemia vera: an international study. Leukemia. 2013;27:1874–81.
Ania BJ, Suman VJ, Sobell JL, Codd MB, Silverstein MN, Melton LJ 3rd. Trends in the incidence of polycythemia vera among Olmsted County, Minnesota residents, 1935–1989. Am J Hematol. 1994;47:89–93.
Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR, et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med. 2005;352:1779–90.
Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJ, et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocytemia, and myeloid metaplasia with myelofibrosis. Cancer Cell. 2005;7:387–97.
James C, Ugo V, Le Couédic JP, Staerk J, Delhommeau F, Lacout C, et al. A unique clonal JAK2 mutation leading to constitutive signaling causes polycythemia vera. Nature. 2005;434:1144–8.
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S, et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet. 2005;365:1054–61.
Scott LM, Tong W, Levine RL, Scott MA, Beer PA, Stratton MR, et al. JAK2 eon 12 mutations in polycythemia vera and idiopathic erythrocytosis. N Engl J Med. 2007;356:459–68.
Mullally A, Lane SW, Ball B, Megerdichian C, Okabe R, Al-Shahrour F, et al. Physiological Jak2V617F expression causes a lethal myeloproliferative neoplasm with differential effects on hematopoietic stem and progenitor cells. Cancer Cell. 2010;17:584–96.
Hasan S, Lacout C, Marty C, Cuingnet M, Solary E, Vainchenker W, et al. JAK2V617F expression in mice amplifies early hematopoietic cells and gives them a competitive advantage that is hampered by IFN alpha. Blood. 2013;122:1464–77.
Tiedt R, Hao-Shen H, Sobas MA, Looser R, Dirnhofer S, Schwaller J, et al. Ratio of mutant JAK2-V617F to wild-type Jak2 determines the MPD phenotypes in transgenic mice. Blood. 2008;111:3931–40.
Grisouard J, Li S, Kubovcakaova L, Rao TN, Meyer SC, Lundberg P, et al. JAK2 exon 12 mutant mice display isolated erythrocytosis and changes in iron metabolism favoring increased erythropoiesis. Blood. 2016;128:839–51.
Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A, et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114:937–51.
Tefferi A, Lasho TL, Gugioelmelli P, Finke CM, Totunno G, Elala Y, et al. Targeted deep sequencing in polycythemia vera and essential thrombocythemia. Blood Adv. 2016;1:21–30.
Siegel FP, Tauscher J, Petrides PE. Aquagenic pruritus in polycythemia vera: characteristics and influence on quality of life in 441 patients. Am J Hematol. 2013;88:665–9.
Michiels JJ. Erythromelalgia and vascular complications in polycythemia vera. Semin Thromb Hemost. 1997;23:441–54.
Torgano G, Mandelli C, Massaro P, Abbiati C, Ponzetto A, Bertinieri G, et al. Gastroduodenal lesions in polycythemia vera: frequency and role of Helicobacter pylori. Br J Haematol. 2002;117:198–202.
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391–405.
Barbui T, Thiele J, Gisslinger H, Finazzi G, Carobbio A, Rumi E, et al. Masked polycythemia vera (mPV): results of an international study. Am J Hematol. 2014;89:52–4.
Lussana F, Carobbio A, Randi ML, Rumi EC, Finazzi G, et al. A lower intensity of treatment may underlie the increased risk of thrombosis in young patients with masked polycythemia vera. Br J Haematol. 2014;167:541–6.
Barbui T, Thiele J, Passamonti F, Rumi E, Boveri E, Randi ML, et al. Initial bone marrow reticulin fibrosis in polycythemia vera exerts an impact on clinical outcome. Blood. 2012;119:2239–41.
Berlin NI. Diagnosis and classification of the polycythemias. Semin Hematol. 1975;12:339–51.
Barbui T, Barosi G, Birgegard G, Cervantes F, Finazzi G, Griesshammer M, et al. Philadelphia-negative classical myeloproliferative neoplasms: critical concepts and management recommendations from European LeukemiaNet. J Clin Oncol. 2011;29:761–70.
Passamonti F, Rumi E, Pietra D, Elena C, Boveri E, Arcaini L, et al. A prospective study of 338 patients with polycythemia vera: the impact of JAK2 (V617F) allele burden and leukocytosis on fibrotic or leukemic disease transformation and vascular complications. Leukemia. 2010;24:1574–9.
Marchioli R, Finazzi G, Specchia G, Cacciola R, Cavazzina R, Cilloni D, et al. Cardiovascular events and intensity of treatment of polycythemia vera. N Engl J Med. 2013;368:22–33.
Berk PD, Goldberg JD, Silverstein MN, Weinfeld A, Donovan PB, Ellis JT, et al. Increased incidence of acute leukemia in polycythemia vera associated with chlorambucil treatment. N Engl J Med. 1981;304:441–7.
Landolfi R, Marchioli R, Kutti J, Gisslinger H, Tognoni G, Patrono C, et al. Efficacy and safety of low-dose aspirin in polycythemia vera. N Engl J Med. 2004;350:114–24.
Michiels JJ, Berneman Z, Schroyens W, Koudstaal PJ, Lindemans J, Nemann HA, et al. Platelet-mediated erythromelalgia, cerebral, ocular and coronary microvascular ischemic and thrombotic manifestations in patients with essential thrombocythemia and polycythemia vera: Platelets. 2006;17:528–44. a distinct aspirin-responsive and coumadin-resistant arterial thrombophilia.
Passamonti F, Rumi E, Randi ML, Morra E, Cazzola M. Aspirin in pregnant patients with essential thrombocythemia: a retrospective analysis of 129 pregnancies. J Thromb Haemost. 2010;8:411–3.
Kiladjian JJ, Mesa RA, Hoffman R. The renaissance of interferon therapy for the treatment of myeloid malignancies. Blood. 2011;117:4706–15.
Kiladjian JJ, Cassinat B, Chevret S, Turlurle P, Cambier N, Roussel M, et al. Pegylated interferon-alfa-2a induces complete hematologic and molecular responses with low toxicity in polycythemia vera. Blood. 2008;112:3065–72.
Quintas-Cardama A, Kantarjian H, Manshuouri T, Luthra R, Estrov Z, Pierce S, et al. Pegylated interferon alfa-2a yields high rates of hematologic and molecular response in patients with advanced essential thrombocythemia and polycythemia vera. J Clin Oncol. 2009;27:5418–24.
Gisslinger H, Zagrijtschuk O, Buxhofer-Ausch V, Thaler J, Schloegl E, Gastl GA, et al. Ropeginterferon alfa-2b, a novel IFN alfa-2b, induces high response rates with low toxicity in patients with polycythemia vera. Blood. 2015;126:1762–9.
Gisslinger H, Klade C, Georgiev P, Krochmalczyk D, Gercheva L, Egyed M, et al. Ropeginterferon alfa-2b induces high rates of clinical, hematological and molecular responses in polycythemia vera: two-year results from the first prospective randomized controlled trial. Blood. 2017;130:320.
Najean Y, Rain J‑D. Treatment of polycythemia vera: the use of hydroxyurea and pipobroman in 292 patients under the age of 65 years. Blood. 1997;90:3370–7.
Kiladjian JJ, Chevret S, Dosquet C, Chomienne C, Rain J‑D. Treatment of polycythemia vera with Hydroxyurea and Pipibroman: final results of a randomized trial initiated in 1980. J Clin Oncol. 2011;29:3907–13.
Alvarez-Larran A, Kerguelen A, Hernandez-Boluda JC, Perez-Encinas M, Ferrer-Marin F, Barez A, et al. Frequency and prognostic value of resistance/intolerance to hydroxycarbamide in 890 patients with polycythemia vera. Br J Haematol. 2016;172:786–93.
Antonioli E, Gugliemelli P, Pieri L, Finazzi M, Rumi E, Martinelli V, et al. Hydroxyurea-related toxicity in 3411 patients with Ph-negative MPN. Am J Hematol. 2012;87:552–4.
Latagliata R, Spadea A, Cedrone M, Ki Giandonenico J, De Muro M, Villivà N, et al. Symptomatic mucocutaneous toxicity of hydroxyurea in Philadelphia chromosome-negative myeloproliferative neoplasms: the mister Hyde face of a sage drug. Cancer. 2012;118:404–9.
Verstovsek S, Passamonti F, Rambaldi A, Barosi G, Rosen PJ, Rumi E, et al. A phase 2 study of Ruxolitinib, an oral JAK1 and JAK2 inhibitor, in patients with advanced polycythemia vera who are refractory or intolerant to hydroxyurea. Cancer. 2014;120:513–20.
Vanucchi AM, Kiladjian JJ, Griesshammer M, Masszi T, Durrant S, Passamonti F, et al. Ruxolitinib versus standard therapy for the treatment of polycythemia vera. N Engl J Med. 2015;372:426–35.
Passamonti F, Griesshammer M, Palandri F, Egyed M, Benevolo G, Devos T, et al. Ruxolitinib for the treatment of inadequately controlled polycythemia vera without splenomegaly (RESPONSE-2): a randomized, open-label, phase 3b study. Lancet Oncol. 2017;18:88–99.
Vanucchi AM, Verstovsek S, Gugliemelli P, Griesshammer M, Burn TC, Naim A, et al. Ruxolitnib reduces JAK2p.V617F allele burden in patients with polycythemia vera enrolled in the RESPONSE study. Ann Hematol. 2017;96:1113–20.
Heine A, Brossart P, Wolf D. Ruxolitnibis a potent immunosuppressive compound: is it time for anti-infective prophylaxis? Blood. 2013;122:3843–4.
Elliott MA, Tefferi A. Thrombosis and haemorrhage in polycythemia vera and essential thrombocythaemia. Br J Haematol. 2005;128:275–90.
Smalberg JH, Arends LR, Valla DC, Kiladjian JJ, Janssen HL, Leebeek FW. Myeloproliferative neoplasms in Budd-Chiari syndrome and portal vein thrombosis: a meta-analysis. Blood. 2012;120:4921–8.
Mancuso A. An update on the management of Budd-Chiari-syndrome: the issues of timing and choice of treatment. Eur J Gastroenterol Hepatol. 2015;27:200–3.
De Stefano V, Qi X, Betti S, Rossi E. Splanchnic vein thrombosis and myeloproliferative neoplasms: molecular-driven diagnosis and long-term treatment. Thromb Haemost. 2016;115:240–9.
Griesshammer M, Struve S, Barbui T. Management of Philadelphia negative chronic myeloproliferative disorders in pregnancy. Blood Rev. 2008;22:235–45.
Acknowledgements
A single meeting for discussion of the final version of these recommendations was sponsored by Novartis Pharma GmbH and AOP Orphan Pharmaceuticals AG.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The following authors declare that they have served as consultants or received speakers fees from industrial companies: S. Burgstaller Novartis, AOP Orphan, K. Geissler Novartis, AOP Orphan, H. Gisslinger Novartis, AOP Orphan, Baxalta, Celgene, M. Krauth Novartis, AOP Orphan, A. Wölfler Novartis, AOP. V. Buxhofer-Ausch, T. Sliwa, C. Beham-Schmid, G. Gastl, T. Melchardt, P. Krippl, A. Petzer and H. Rumpold declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Burgstaller, S., Buxhofer-Ausch, V., Sliwa, T. et al. Austrian recommendations for the management of polycythemia vera. Wien Klin Wochenschr 130, 535–542 (2018). https://doi.org/10.1007/s00508-018-1359-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-018-1359-3