Abstract
Key message
Predawn water potential disequilibrium in hybrid aspen coppice growing in humid environment was mostly determined by hydraulic conductance and the ratio of nocturnal to daily water loss was relatively small.
Abstract
Water relations are of crucial importance for biomass accumulation in trees and forest productivity, yet the significance of nocturnal water use is still debated. We investigated which environmental factors influence nocturnal transpiration, development of the equilibrium between soil and plant water potentials and plant hydraulic conductance in hybrid aspen (Populus tremula L. × P. tremuloides Michx.) coppice. Predawn leaf water potential and sap flow were measured simultaneously with relevant environmental factors on 2-year-old coppice shoots during the growing season of 2015. Nocturnal water loss constituted on average 4–5% of daily total transpiration and almost 2/3 of nocturnal sap flow. The nocturnal water loss was mainly driven by atmospheric evaporative demand, while maximum sap flux densities could extend 35% of the daytime values on dry nights. Predawn leaf water potential was constantly more negative than soil water potential, resulting in predawn disequlibrium between leaf and soil water potentials (PDD). Total soil-to-leaf hydraulic conductance was the primary factor explaining the variation in PDD. Our results suggest that in humid conditions plant hydraulic capacity is more substantial determining plant predawn water status than stomatal behaviour with respect to current environmental drivers.
Similar content being viewed by others
Introduction
Nocturnal transpiration and sap flow have received considerable attention from researchers during the last two decades and their occurrence has been reported in a wide range of ecosystems (Benyon 1999; Bucci et al. 2005; Dawson et al. 2007; Sellin and Lubenets 2010; Zeppel et al. 2010). A large meta-analysis shows that nocturnal sap flow constitutes on average 12% of total daily sap flow across diverse woody plant species (Forster 2014). It is highest in species growing in equatorial biomes (29%), and smallest in warm temperate and nemoral ecosystems, averaging 8 and 10.5%, respectively.
Many studies on nocturnal transpiration provide evidence that stomata do not close completely for night allowing substantial diffusion of water vapour from intercellular spaces to the atmosphere (Bucci et al. 2004; Daley and Phillips 2006; Scholz et al. 2007; Howard and Donovan 2010; Resco de Dios et al. 2013). Different mechanisms have been suggested to explain the night-time stomatal openness resulting in transpiration driven by leaf-to-air vapour pressure differences. Nocturnal transpiration may help to acquire more nutrients from poor soil by mass flow of water (Scholz et al. 2007). However, Howard and Donovan (2010) found differences neither in nocturnal transpiration nor stomatal conductance between different nutrient treatments in two Populus species. Nocturnal transpiration may also enhance oxygen delivery to living parenchyma cells in stem (Gansert 2003; Daley and Phillips 2006). It might also facilitate carbon fixation early in the morning (Caird et al. 2006; Dawson et al. 2007; Resco de Dios et al. 2016). This has also implications for regional water balance and global water cycle. Community Land Model using the actual values of nocturnal stomatal conductance instead of broadly used zero values have estimated the runoff and soil moisture to be lower than previously expected, which in turn results in estimates of gross primary production to be smaller, with more pronounced impact in drier regions (Lombardozzi et al. 2017).
Open stomata and concurrent transpiration during the night cause water losses from the plant, which may hinder predawn leaf water potential to equilibrate with that of the soil (Donovan et al. 1999; Sellin 1999; Bucci et al. 2004), leading to a phenomenon called predawn disequilibrium, PDD (Ourcival and Berger 1995; Donovan et al. 1999). As predawn leaf water potential is widely used to estimate soil water status in ecophysiological studies, it is crucial to consider potential occurrence of PDD under any environmental conditions. For example, in areas with prolonged drought periods PDD tends to be greater (Bucci et al. 2005; Rogiers et al. 2012) because the soil water content could decrease to a degree where the contact between root surface and soil solution weakens, which in turn constrains water to enter the roots (Ourcival and Berger 1995; Cochard et al. 1996). At high latitudes, too short summer nights may also prevent leaf and soil water potentials to equilibrate with each other (Sellin 1999). So far, more information on PDD has been reported for species growing in warmer and drier ecosystems, where atmospheric vapour pressure deficit (VPD) and hence transpiration are greater during night (Donovan et al. 2003; Bucci et al. 2004; 2005). However, Sellin (1999) discovered moderate PDD in common boreal species such as Picea abies (L.) Karst. and Vaccinium myrtillus L. growing in habitats with ample water availability.
Leaf water supply and water status are largely determined by plant hydraulic capacity, i.e. how efficiently water can be transferred from root surface to evaporation sites in leaves. Plant hydraulic conductance controls maximum stomatal conductance, which ultimately affects photosynthetic capacity, growth rate and competitive capacity (Brodribb 2009). Therefore, the structure and function of the water transport system govern the productivity and survival of land plants, because the vascular architecture sets physical limits on plant functioning (Ryan et al. 2006). Hydraulic traits mediate the ways in which plants interact with their abiotic and biotic environment. On a landscape to global scale, plant hydraulic properties are important for describing the function of ecological communities and ecosystems (Sack et al. 2016).
Long-distance water transport is not a critical issue at night-time because water losses from the foliage are usually small. However, nocturnal water use may be quite substantial under certain conditions even in northern mesophytes as reported hereinbefore. Plant hydraulic properties are not constant on a daily scale, but exhibit different daily courses depending on tree species and environmental conditions (Yang et al. 2012; Õunapuu and Sellin 2013; Sack et al. 2016). The daily pattern of hydraulic conductance has been recently reported for hybrid aspen (Populus tremula L. × P. tremuloides Michx.) by Meitern et al. (2017). Hydraulic conductance measured just prior to sunrise, when plant water potentials are highest and tissues refilled with water, represents a certain reference point—a basic level of hydraulic efficiency corresponding to particular water availability. After sunrise, primarily two opposite processes govern the daily dynamics of hydraulic traits starting from the basic level: first, hydraulic conductance is upregulated along with rising irradiance and temperature to provide sufficient leaf water supply to meet increasing atmospheric evaporative demand (Sellin et al. 2008; Meitern et al. 2017); second, increasing xylem embolism in roots, stems and leaves is caused by tension in water conducting tissues generated by growing water losses (Pivovaroff et al. 2014; Nardini et al. 2017; Sellin et al. 2017b).
The purpose of the present study was to assess nocturnal water use and predawn physiological traits on a coppice of hybrid aspen, a commercially important tree species in Northern Europe, which is widely used in short-rotation forestry (Tullus et al. 2012b). The specific aims of our experiment were to (1) establish environmental variables governing night-time variation in water relations parameters; (2) estimate how much does nocturnal transpiration contribute to total daily and nocturnal sap flow; (3) test for possible effects of nocturnal transpiration and hydraulic conductance on the development of PDD. Although nocturnal water transport is not a critical point for trees growing at high latitudes where nights are usually cool and humid, we hypothesize that predawn water potential disequilibrium is determined by hydraulic constraints in addition to atmospheric variables.
Materials and methods
Study site and sample trees
The study was conducted in hybrid aspen (Populus tremula × P. tremuloides), an artificial cross of two wide-spread boreal and hemiboreal broadleaved tree species, in an experimental forest plantation at the Free Air Humidity Manipulation (FAHM) facility located at Rõka village (58°14′ N, 27°17′ E), eastern Estonia, in 1st of July until 11th of September of 2015. The hybrid aspen clone grown at the FAHM site is an offspring of aspens from southern boreal regions of Finland and Canada (Tullus et al. 2012a). The study area belongs to the hemiboreal forest zone, a detailed description of the site is presented in Kupper et al. (2011). Summer of 2015 was relatively rainy, which diminished the effect of air humidification, and as a result there was no significant difference in RH between the control and humidified plots across the growing period. Therefore, the data collected from both treatments (elevated RH and control) were treated as a single dataset.
The present experiment was carried out on 2-year-old coppice shoots (root and stump sprouts) with mean height (± SE) 3.25 ± 0.15 m, and mean stem diameter at 0.3 m above the ground 24.1 ± 1.2 mm at the end of the growing season. Sapwood to leaf area ratio estimated by litterfall averaged 2.13 × 10−4 m2 m−2 prior to the beginning of leaf shedding. For litter gathering, two litter traps (0.21 m2) were installed under hybrid aspens at each plot; litter was collected at 2-week intervals from the end of July and weighed after drying to constant weight at 70 °C. Dry litter mass (g m−2) was converted into LAI (m2 m−2) using the conversion factor of 0.0144 m2 g−1 (A. Tullus, unpubl.) based on the specific leaf area of hybrid aspen leaf litter.
Environmental variables
Relevant environmental variables were continuously recorded in experimental plots: soil water potential (Ψsoil) with EQ 2 equitensiometers (Delta-T Devices, Burwell, UK) and soil temperature (TS) with ST1 temperature probes (Delta-T Devices) at a depth of 15 and 30 cm; air temperature (TA) and relative humidity (RH) with HMP45A humidity and temperature probes (Vaisala, Helsinki, Finland); wind speed with WindSonic anemometers (Gill Instruments, Lymington, UK). The readings of the sensors were stored as average values every 1-10 min with DL2e data loggers (Delta-T Devices). Atmospheric evaporative demand (AED) was quantified by air vapour pressure deficit (VPD) calculated according to Bolton (1980):
Sap flow measurements
Because of low sap flow rates at night, we applied SFM1 sap flow meters (ICT International, Armidale, Australia) based on heat ratio method thoroughly described by Burgess et al. (2001). Because of the small diameter of the sample trees we installed only one sensor per tree just below the live crown. We sampled twelve trees from four experimental plots simultaneously. To calculate the total water flux in the stem we measured stem diameter: two perpendicular measurements were taken above the sensors and two below the sensors approximately once a week. Bark thickness was subtracted from the average value of the four measurements to calculate the cross-sectional area of xylem. Sap flux density (F) was expressed on an hourly basis per unit leaf area (g m−2 h−1). Zero flow was determined by constant readings at the end of growing season, when trees had shed all the leaves.
Nocturnal sap flow (QN) was separated into nocturnal transpiration (EN) and tissue refilling (RN) using forecasted model (Fisher et al. 2007; Alvarado-Barrientos et al. 2015; Yu et al. 2018). Refilling was interpolated using exponential decay function
where t is time, and a and b are empirical constants. Forecasted curves were constructed using 3 to 4 half hour average sap flow data points from late evening (Fig. 1). The area under the curve during night-time hours denotes RN. Only the evenings with “normal” (no rain event during or prior to the focal period) sap flow patterns were involved in the analysis. EN was calculated as nocturnal sap flow minus RN. The contribution of EN to total daily sap flow was expressed as a percentage (EN/E∙100).
Water potential measurements
Plant water potential was measured with a custom-built Scholander-type pressure chamber with the resolution of 0.01 MPa. Prior to sunrise (3:10–4:15 h) on each measurement day, bulk leaf water potential (Ψleaf) was determined in three detached leaves simultaneously on two trees growing in different plots. Xylem water potential of the branches (Ψbranch) was estimated by applying the bagged leaves method (Brodribb and Holbrook 2003) sampling two leaves per tree, prepared the previous evening. Bagged leaves were enclosed in Minigrip® plastic bags and covered with aluminium foil. Predawn disequilibrium (PDD) of water potentials was expressed as a difference between Ψleaf and Ψsoil recorded before sunrise.
Hydraulic traits
Leaf (KL), soil-to-branch (KS–B) and total (KT) hydraulic conductance were estimated by the evaporative flux method at predawn, assuming that tissue refilling has basically completed by that time:
where Fxa is the sap flux density expressed per xylem cross-sectional area (g cm−2 h−1) and ΔΨ is the driving force (MPa). The driving force for KL, KS–B and KT is represented by water potential drops across the corresponding segments (Ψbranch–Ψleaf, Ψsoil–Ψbranch and Ψsoil–Ψleaf, respectively).
Data analysis
Night was defined as a period between sunset (20:30–22:30 h) and dawn (4:00–6:00 h). In the data analysis, we used arithmetic means of water potentials recorded on three uncovered and two non-transpiring covered leaves. For sap flow analysis only the data recorded before 50% of the foliage has been shed (i.e. until the 12th of September) was used. Nocturnal sap flow values were averaged across the entire night. In the daytime data analysis, we used only the data recorded on Sundays when the air humidification was turned off (to avoid direct effects of the humidification); additionally we excluded rainy days. Analysis regarding nocturnal sap flow included all nights, as humidification was not applied during nights.
Statistical analysis was carried out using Statistica 7 (StatSoft Inc., Tulsa, OK). The differences in water potentials of bare and covered leaves, and soil were analysed with a Tukey’s test. The effects of environmental parameters on physiological traits were assessed by applying multiple linear regression. To assess the proportion of variance that was explained by all the relevant environmental factors, Type III sums of squares was used. Normality of data and homogeneity of variances were checked using the Kolmogorov–Smirnov D-statistic and the Levene test, respectively. When necessary, logarithmic or complex transformations were applied to the data. Relationships between the investigated characteristics and independent variables were analysed using simple linear, forward stepwise or non-linear least squares regressions.
Results
Environmental variables
Soil was relatively wet throughout the entire growing season, soil water potential (Ψsoil) dropped down to − 0.05 to − 0.06 MPa at the beginning of July and at the end of August in the wettest layer (Fig. 2). On the days when predawn leaf water potential (Ψleaf) was measured, Ψsoil was even closer to zero compared to the mean values calculated across the entire experiment period (Table 1). Nocturnal RH was usually above 90%, but during dry nights dropped to 80% (Fig. 2). During nights when Ψleaf was measured, VPD extended maximally up to 0.2 kPa.
Predawn disequilibrium of water potentials
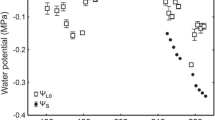
Mean Ψleaf was − 0.20 MPa and it was significantly more negative (P < 0.001) than Ψsoil according to the Tukey’s test (Fig. 3), resulting in mean predawn disequilibrium (PDD) of 0.19 MPa (Table 2), whereas the highest PDD values exceeded 0.5 MPa. Multiple regression model combining VPD and total hydraulic conductance (KT) described less than half of the variance (R2 = 0.43, P < 0.001) in PDD, whereas approximately one-third of it was attributable to KT (Table 3). KT and PDD demonstrated an inverse relationship: the lower the hydraulic conductance, the higher the PDD. Ψleaf was significantly more negative (P < 0.001) than branch water potential (Ψbranch; Fig. 3). Also Ψbranch was significantly (P < 0.001) lower than Ψsoil. Our analysis revealed no relationship between PDD and nocturnal sap flow.
Nocturnal sap flow
Average sap flux density per unit leaf area (FN) over the entire measuring period was 5.9 g m−2 h−1 (Table 2). The highest nightly mean FN values were recorded in the nights with higher vapour pressure deficit (VPD > 0.25 kPa). Single values of FN reached 36 g m−2 h−1 that constituted 35% of the average daily maximum sap flux densities in dry days over the entire measuring period. Multiple regression model combining VPD and Ψsoil described majority of the variance (R2 = 0.85, P < 0.001) in FN, whereas VPD explained most of the total variance (Table 3). When analysed separately from other factors, VPD described 82% of the variance in FN (P < 0.001). However, when separating FN data into two groups based on Ψsoil, then the slope of FN increase in response to VPD was less steep (P < 0.001) for the soil near water saturation (Ψsoil ≥ − 0.01 MPa) compared to drier conditions (Ψsoil < − 0.01 MPa; Fig. 4).
Nocturnal transpiration (EN) and tissue refilling (RN) varied within wide ranges (Fig. 1): EN constituted 17–95% and RN 5–83% of the nocturnal sap flow (QN). The average contribution of EN to nocturnal sap flow was 62.1%, leaving 37.9% to refilling. Nocturnal refilling was positively related to previous day VPD and inversely to Ψsoil (R2 = 0.16, P < 0.001; Table 3). QN constituted on average 7.0% of diurnal total sap flow (E), and EN, estimated with forecast approach, constituted 4.5% of E. Based on Type III sums of squares, wind speed had no significant effect on any of the physiological parameters.
Hydraulics
Both predawn total hydraulic conductance (KT) and soil-to-branch conductance (KS-B) were best described by a multiple regression model combining Ψsoil and VPD averaged across 4 h prior to sunrise (Table 3). Dividing KT and KS-B into two groups based on Ψsoil, revealed significantly (P = 0.020) smaller KT and KS–B—0.029 and 0.073 kg m−2 s−1 MPa−1, respectively—for the soil near water saturation (Ψsoil ≥ − 0.01 MPa). The respective values in drier soil averaged 0.046 and 0.108 kg m−2 s−1 MPa−1. None of the environmental characteristics measured in our experiment was associated with predawn leaf hydraulic conductance (KL), while KL was positively correlated with Ψleaf (R2 = 0.32, P < 0.001). KT was also positively correlated with Ψleaf (R2 = 0.23, P < 0.001), but no correlation was found between KS–B and Ψleaf. Leaf relative resistance constituted on average 58 ± 2% of the total liquid-phase resistance.
Discussion
Predawn water potential disequilibrium
Comparison of leaf and soil water potentials measured before sunrise revealed the occurrence of predawn water potential disequilibrium in 2-year-old hybrid aspen coppice. Our results confirm the hypothesis—total soil-to-leaf hydraulic conductance was the main factor contributing to PDD (Table 3). Under low atmospheric evaporative demand at night the impact of atmospheric drivers on leaf water status is weak and consequently the speed of nocturnal rehydration of leaves and the magnitude of PDD are more influenced by plant hydraulic efficiency. The higher the plant hydraulic efficiency, the smaller the PDD, i.e. high KT ensured more efficient water transfer to the leaves resulting in low disequilibrium between soil and leaf water potentials, and vice versa. Factors affecting plant hydraulics are discussed hereinafter.
Atmospheric variables had smaller effect on PDD than we expected, probably also because of moderately rainy and cool weather prevailing in summer of 2015. However, the data analysis demonstrated that some of the variation in PDD is still attributable to VPD, which confirms that stomata were at least partially open during night. This conclusion is also supported by the difference in water potential values between bare and covered leaves (Fig. 3) and the strong relationship observed between VPD and FN (Fig. 4). Contrary to other studies (Donovan et al. 2003; Bucci et al. 2005) VPD was not the major factor explaining PDD in the present case. That was evidently due to high RH prevailing during most of the nights and in predawn hours. Thus, stomatal behaviour was likely a secondary reason for the development of PDD in aspen coppice.
Reduced hydraulic efficiency of the water conducting system is not a crucial issue for trees in the morning when AED is low during the night and soil water availability is not limiting factor. However, when decreased hydraulic conductance is combined with high AED and stomatal openness during the night, then trees might experience more negative water potential in the first half of the day and thereafter. That may lead to early stomatal closure or partial closure in the daytime, which in turn reduces assimilation of CO2 and productivity (Chaves 1991).
Nocturnal sap flow and hydraulic conductance
Sap flow data measured continuously in our field experiment confirms that on average 6–8% of the sap flow in hybrid aspen coppice takes place during night. That is somewhat smaller than the average value reported for nemoral deciduous tree species in the meta-analysis by Forster (2014). However, in our study the nocturnal sap flow was separated into refilling (RN) and transpiration (EN), and the latter constituted 4–5% of the total daily transpiration. Nocturnal sap flow rates strongly correlate with VPD in our study. However, the ecophysiological significance of nocturnal stomatal openness still remains unclear. Kupper et al. (2012) suggest that fast-growing species such as hybrid aspen keep their stomata open at night to enhance nutrient uptake by mass flow of water in fertile sites and support high growth rates. A similar finding has been reported for Ulmus laevis Pall. by Eller et al. (2017), although the same paper reported the opposite result for Fraxinus excelsior L., i.e. nocturnal stomatal conductance was higher in trees growing under nitrogen deficiency.
The proportion of EN in nocturnal sap flow found in our study (roughly 60–65%) is much higher than that estimated with similar method by Fisher et al. (2007); Yu et al. (2018), where proportion of EN averaged 15 and 20%, respectively. These low values seem to result from relatively low soil water content in those studies (volumetric soil water content 15–20%). Substantially higher proportion of EN (75–95%) was reported in a study performed in tropical montane cloud forest where soil water content remained high throughout the study period (Alvarado-Barrientos et al. 2015), as was the case in our experiment (Fig. 2). The positive relationship between proportion of RN and VPD of previous day is an expected result, because high VPD during the daytime caused trees’ water storage to deplete to a greater extent and, therefore, higher proportion of nocturnal sap flow contributed to the refilling of dehydrated tissues.
Some of the variation in FN and plant hydraulic conductance was explained by soil water status (Table 3). Trees experiencing less negative Ψsoil, i.e. wetter soil, exhibited lower FN (Fig. 4) and also lower KT and KS–B. That result may seem counterintuitive, but when soil is quite wet already, then further increase in soil water content causes deficient aeration which may lead to disturbed water uptake and/or to smaller transpiration. The former happens because of decreased root permeability, and the latter due to the accumulation of abscisic acid in leaves, which calls upon stomatal closure (Kozlowski 1997). Both low root hydraulic conductivity and stomatal sensitivity to VPD in response to excess of water have documented in P. abies (Sellin 2001). Decreased root permeability would also explain the negative relationship between KT and PDD: lower hydraulic efficiency prevents Ψleaf to equilibrate with Ψsoil. Moreover, excessive water in the soil disturbs also other physiological processes (diminished nutrient uptake, decline in leaf photosynthetic capacity, disturbed nitrogen metabolism, accumulation of starch, changes in secondary metabolite contents, shifts in resource allocation, etc.) in trees, which may lead to growth decline as shown for Betula pendula (Sellin et al. 2017a). However, we are aware that during this experiment the soil was wet or very wet, and one could suspect that under drier soil conditions FN and plant hydraulic conductance would probably be positively correlated to Ψsoil. Several studies have reported that decreased nocturnal sap flow is associated with lower soil moisture (Zeppel et al. 2010).
Both KS–B and KT of hybrid aspen were positively related to VPD, which indicates that hydraulic conductance is upregulated with increasing AED. This stimulus is most likely sensed by leaves and mediated via hydraulic signalling pathway starting with changes in leaf water potential. On a daily scale, the hydraulic parameters of hybrid aspen are most affected by air temperature, but the temperature variation at predawn is very small in the hemiboreal zone (Meitern et al. 2017). Leaf hydraulic resistance constituted more than half of the tree’s total liquid-phase resistance at predawn. Such a high contribution can be explained by two matters: first, the conductance of extravascular compartment is downregulated in the dark (Nardini et al. 2005); second, the small size (3.1–3.4 m) of the experimental trees means a relatively short transport path to pass within roots, stems and branches. In tall forest trees, leaves including petioles commonly contribute 30–45% of the total hydraulic resistance (Sack and Holbrook 2006; Õunapuu and Sellin 2013).
Conclusions
Hybrid aspen coppice exhibited substantial predawn disequilibrium between leaf and soil water potentials driven by hydraulic efficiency of the water transport pathway and nocturnal transpiration. AED is the main determinant of night-time transpiration, which constituted on average 4–5% of daily total transpiration. Growing in humid environment weakens environmental pressure for the development of an efficient water transport system in trees, creating bigger constraints to the water transfer from roots to leaves that lead to considerable predawn disequilibrium between soil and leaf water potentials. Our results suggest that plant hydraulic capacity is more substantial determining plant predawn water status than stomatal behaviour in humid conditions, but it might not be the case in drier environment.
Author contribution statement
OK was responsible for conducting the experiment, analysis of data and preparation of the manuscript. AS was responsible for design of the experiment and preparation of the manuscript. AT was responsible for collecting and analysing leaves shedding data.
References
Alvarado-Barrientos SM, Holwerda F, Geissert DR, Muños-Villers LE, Gotsch SG, Asbjornsen H, Dawson TE (2015) Nighttime transpiration in a seasonally dry tropical montane cloud forest environment. Trees 29:259–274
Benyon R (1999) Nighttime water use in an irrigated Eucalyptus grandis plantation. Tree Physiol 19:853–859
Bolton D (1980) The computation of equivalent potential temperature. Mon Weather Rev 108:1046–1053
Brodribb TJ (2009) Xylem hydraulic physiology: the functional backbone of terrestrial plant productivity. Plant Sci 177:245–251
Brodribb TJ, Holbrook NM (2003) Changes in leaf hydraulic conductance during leaf shedding in seasonally dry tropical forest. New Phytol 158:295–303
Bucci SJ, Scholz FG, Goldstein G, Meinzer FC, Hinojosa JA, Hoffmann WA, Franco AC (2004) Processes preventing nocturnal equilibration between leaf and soil water potential in tropical savanna woody species. Tree Physiol 24:1119–1127
Bucci SJ, Goldstein G, Meinzer FC, Franco AC, Campanello P, Scholz FG (2005) Mechanisms contributing to seasonal homeostasis of minimum leaf water potential and predawn disequilibrium between soil and plant water potential in Neotropical savanna trees. Trees 19:296–304
Burgess SSO, Adams MA, Turner NC, Beverly CR, Ong CK, Khan AAH, Bleby TM (2001) An improved heat pulse method to measure low and reverse rates of sap flow in woody plants. Tree Physiol 21:589–598
Caird MA, Richards JH, Donovan LA (2006) Nighttime stomatal conductance and transpiration in C3 and C4 plants. Plant Physiol 143:4–10
Chaves MM (1991) Effects of water deficits on carbon assimilation. J Exp Bot 42:1–16
Cochard H, Bréda N, Granier A (1996) Whole tree hydraulic conductance and water loss regulation in Quercus during drought: evidence for stomatal control of embolism? Ann Sci Forest 53:197–206
Daley MJ, Phillips NG (2006) Interspecific variation in nighttime transpiration and stomatal conductance in a mixed New England deciduous forest. Tree Physiol 26:411–419
Dawson TE, Burgess SSO, Tu KP, Oliveira RS, Santiago LS, Fisher JB, Simonin KA, Ambrose AR (2007) Nighttime transpiration in woody plants from contrasting ecosystems. Tree Physiol 27:561–575
Donovan LA, Grisé DJ, West JB, Pappert RA, Alder NN, Richards JH (1999) Predawn disequilibrium between plant and soil water potentials in two cold-desert shrubs. Oecologia 120:209–217
Donovan LA, Richards JH, Linton MJ (2003) Magnitude and mechanisms of disequilibrium between predawn plant and soil water potentials. Ecology 84:463–470
Eller F, Jensen K, Reisdorff C (2017) Nighttime stomatal conductance differs with nutrient availability in two temperate floodplain tree species. Tree Physiol 37:428–440
Fisher JB, Baldocchi DD, Misson L, Dawson TE, Goldstein AH (2007) What the towers don’t see at night: nocturnal sap flow in trees and shrubs at two AmeriFlux sites in California. Tree Physiol 27:597–610
Forster MA (2014) How significant is nocturnal sap flow? Tree Physiol 34:757–765
Gansert D (2003) Xylem sap flow as a major pathway for oxygen supply to the sapwood of birch (Betula pubescens Ehr.). Plant, Cell Environ 26:1803–1814
Howard AR, Donovan LA (2010) Soil nitrogen limitation does not impact nighttime water loss in Populus. Tree Physiol 30:23–31
Kozlowski TT (1997) Responses of woody plants to flooding and salinity. Tree Physiol Monogr 1:1–29
Kupper P, Sõber J, Sellin A, Lõhmus K, Tullus A, Räim O, Lubenets K, Tulva I, Uri V, Zobel M, Kull O, Sõber A (2011) An experimental facility for free air humidity manipulation (FAHM) can alter water flux through deciduous tree canopy. Environ Exp Bot 72:432–438
Kupper P, Rohula G, Saksing L, Sellin A, Lõhmus K, Ostonen I, Helmisaari HS, Sõber A (2012) Does soil nutrient availability influence night-time water flux of aspen saplings? Environ Exp Bot 82:37–42
Lombardozzi DL, Zeppel MJB, Fisher RA, Tawfik A (2017) Representing nighttime and minimum conductance in CLM4.5: global hydrology and carbon sensitivity analysis using observational constraints. Geosci Model Dev 10:321–331
Meitern A, Õunapuu-Pikas E, Sellin A (2017) Circadian patterns of xylem sap properties and their covariation with plant hydraulic traits in hybrid aspen. J Plant Physiol 213:148–156
Nardini A, Salleo S, Andri S (2005) Circadian regulation of leaf hydraulic conductance in sunflower (Helianthus annuus L. cv Margot). Plant, Cell Environ 28:750–759
Nardini A, Savi T, Losso A, Petit G, Pacilè S (2017) X-ray microtomography observations of xylem embolism in stems of Laurus nobilis are consistent with hydraulic measurements of percentage loss of conductance. New Phytol 213:1068–1075
Õunapuu E, Sellin A (2013) Daily dynamics of leaf and soil-to-branch hydraulic conductance in silver birch (Betula pendula) measured in situ. Plant Physiol Biochem 68:104–110
Ourcival J-M, Berger A (1995) Equilibrium between soil water potential and predawn water potential of two pre-saharan shrub species of Tunisia. J Arid Environ 30:175–183
Pivovaroff AL, Sack L, Santiago LS (2014) Coordination of stem and leaf hydraulic conductance in southern California shrubs: a test of the hydraulic segmentation hypothesis. New Phytol 203:842–850
Resco de Dios V, Diaz-Sierra R, Goulden ML, Barton CVM, Boer MM, Gessler A, Ferrio JP, Pfautsch S, Tissue DT (2013) Woody clockworks: circadian regulation of night-time water use in Eucalyptus globulus. New Phytol 200:743–752
Resco de Dios V, Loik ME, Smith R, Aspinwall MJ, Tissue DT (2016) Genetic variation in circadian regulation of nocturnal stomatal conductance enhances carbon assimilation and growth. Plant, Cell Environ 39:3–11
Rogiers SY, Greer DH, Hatfield JM, Hutton RJ, Clarke SJ, Hutchinson PA, Somers A (2012) Stomatal response of an anisohydric grapevine cultivar to evaporative demand, available soil moisture and abscisic acid. Tree Physiol 32:249–261
Ryan MG, Phillips N, Bond BJ (2006) The hydraulic limitation hypothesis revisited. Plant, Cell Environ 29:367–381
Sack L, Holbrook NM (2006) Leaf hydraulics. Annu Rev Plant Biol 57:361–381
Sack L, Ball MC, Brodersen C, Davis SD, Des Marais DL, Donovan LA, Givnish TJ, Hacke UG, Huxman T, Jansen S, Jacobsen AL, Johnson DM, Koch GW, Maurel C, Mcculloh KA, Mcdowell NG, Mcelrone A, Pellegrini M, Pockman WT, Pratt RB, Sala A, Santiago LS, Savage JA, Scoffoni C, Sevanto S, Sperry J, Tyerman SD, Way D, Holbrook NM (2016) Plant hydraulics as a central hub integrating plant and ecosystem function: meeting report for Emerging Frontiers in Plant Hydraulics (Washington, DC, May 2015). Plant, Cell Environ 39:2087–2094
Scholz FG, Bucci SJ, Goldstein G, Meinzer FC, Franco AC, Miralles-Wilhelm F (2007) Removal of nutrient limitations by long-term fertilization decreases nocturnal water loss in savanna trees. Tree Physiol 27:551–559
Sellin A (1999) Does pre-dawn water potential reflect conditions of equilibrium in plant and soil water status? Acta Oecol 20:51–59
Sellin A (2001) Hydraulic and stomatal adjustment of Norway spruce trees to environmental stress. Tree Physiol 21:879–888
Sellin A, Lubenets K (2010) Variation of transpiration within a canopy of silver birch: effect of canopy position and daily versus nightly water loss. Ecohydrology 3:467–477
Sellin A, Õunapuu E, Kupper P (2008) Effects of light intensity and duration on leaf hydraulic conductance and distribution of resistance in shoots of silver birch (Betula pendula). Physiol Plant 134:412–420
Sellin A, Alber M, Keinänen M, Kupper P, Lihavainen J, Lõhmus K, Oksanen E, Sõber A, Sõber J, Tullus A (2017a) Growth of northern deciduous trees under increasing atmospheric humidity: possible mechanisms behind the growth retardation. Reg Environ Change 17:2135–2148
Sellin A, Alber M, Kupper P (2017b) Increasing air humidity influences hydraulic efficiency but not functional vulnerability of xylem in hybrid aspen. J Plant Physiol 219:28–36
Tullus A, Kupper P, Sellin A, Parts L, Sõber J, Tullus T, Lõhmus K, Sõber A, Tullus H (2012a) Climate change at Northern latitudes: rising atmospheric humidity decreases transpiration, N-uptake and growth rate of hybrid aspen. PLoS One 7:e42648
Tullus A, Rytter L, Tullus T, Weih M, Tullus H (2012b) Short-rotation forestry with hybrid aspen (Populus tremula L. × P. tremuloides Michx.) in Northern Europe. Scand J Forest Res 27:10–29
Yang S-J, Zhang Y-J, Sun M, Goldstein G, Cao K-F (2012) Recovery of diurnal depression of leaf hydraulic conductance in a subtropical woody bamboo species: embolism refilling by nocturnal root pressure. Tree Physiol 32:414–422
Yu T, Feng Q, Si J, Mitchell PJ, Forster MA, Zhang X, Zhao C (2018) Depressed hydraulic redistripution of roots more by stem refilling than by nocturnal transpiration for Populus euphratica Oliv. in situ measurement. Ecol Evol 8:2607–2616
Zeppel MJB, Tissue D, Taylor D, MacInnis-Ng C, Eamus D (2010) Rates of nocturnal transpiration in two evergreen temperate woodland species with differing water-use strategies. Tree Physiol 30:988–1000
Acknowledgements
This study was supported by the Estonian Ministry of Education and Research (institutional research project IUT34-9). Special thanks to Dr Priit Kupper for helpful comments and reviewing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by De Micco.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kangur, O., Tullus, A. & Sellin, A. Night-time transpiration, predawn hydraulic conductance and water potential disequilibrium in hybrid aspen coppice. Trees 34, 133–141 (2020). https://doi.org/10.1007/s00468-019-01903-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-019-01903-9