Abstract
Background
Hyperphosphatemic familial tumoral calcinosis (HFTC) is a rare autosomal recessive disease caused by mutations in genes encoding FGF23 or its regulators, and leading to functional deficiency or resistance to fibroblast growth factor 23 (FGF23). Subsequent biochemical features include hyperphosphatemia due to increased renal phosphate reabsorption, and increased or inappropriately normal 1,25-dihydroxyvitamin D (1,25-D) levels.
Case-diagnosis/treatment
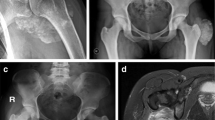
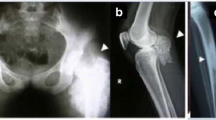
A 15-year-old girl was referred for a 1.2-kg-calcified mass of the thigh, with hyperphosphatemia (2.8 mmol/L); vascular impairment and soft tissue calcifications were already present. DNA sequencing identified compound heterozygous mutations in the FGF23 gene. Management with phosphate dietary restriction, phosphate binders (sevelamer, aluminum, nicotinamide), and acetazolamide moderately decreased serum phosphate levels; oral ketoconazole was secondary administered, leading to significantly decreased 1,25-D levels albeit only moderate additionally decreased phosphate levels. However, therapeutic compliance was questionable. Serum phosphate levels always remained far above the upper normal limit for age. The patient presented with two relapses of the thigh mass, requiring further surgery.
Conclusions
We suggest that control of phosphate metabolism is crucial to prevent recurrences and vascular complications in HFTC; however, the medical management remains challenging.

Similar content being viewed by others
References
Fathi I, Sakr M (2014) Review of tumoral calcinosis: a rare clinico-pathological entity. World J Clin Cases 2:409–414
Bacchetta J, Salusky IB (2012) Evaluation of hypophosphatemia: lessons from patients with genetic disorders. Am J Kidney Dis 59:152–159
Francis F, Henning S, Korn B, Reinhardt R, de Jong P, Poustka A, Lehrach H, Rowe PSN, Goulding JN, Summerfield T, Mountford R, Read AP, Popowska E, Pronicka E, Davies KE, O’Riordan JLH, Econs MJ, Nesbitt T, Drezner MK, Oudet C, Pannetier S, Hanauer A, Strom TM, Meindl A, Lorenz B, Cagnoli B, Mohnike KL, Murken J, Meitinger T (1995) A gene (PEX) with homologies to endopeptidases is mutated in patients with X-linked hypophosphatemic rickets. The HYP Consortium. Nat Genet 11:130–136
ADHR Consortium (2000) Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet 26:345–348
Bastepe M, Jüppner H (2008) Inherited hypophosphatemic disorders in children and the evolving mechanisms of phosphate regulation. Rev Endocr Metab Disord 9:171–180
Benet-Pagès A, Orlik P, Strom TM, Lorenz-Depiereux B (2005) An FGF23 missense mutation causes familial tumoral calcinosis with hyperphosphatemia. Hum Mol Genet 14:385–390
Farrow EG, Imel EA, White KE (2011) Miscellaneous non-inflammatory musculoskeletal conditions. Hyperphosphatemic familial tumoral calcinosis (FGF23, GALNT3 and αKlotho). Best Pract Res Clin Rheumatol 25:735–747
Mozaffarian G, Lafferty FW, Pearson OH (1972) Treatment of tumoral calcinosis with phosphorus deprivation. Ann Intern Med 77:741–745
Shroff R, Long DA, Shanahan C (2013) Mechanistic insights into vascular calcification in CKD. J Am Soc Nephrol 24:179–189
Yamaguchi T, Sugimoto T, Imai Y, Fukase M, Fujita T, Chihara K (1995) Successful treatment of hyperphosphatemic tumoral calcinosis with long-term acetazolamide. Bone 16(4suppl):247S–250S
Ramnitz MS, Gourh P, Goldbach-Mansky R, Wodajo F, Ichikawa S, Econs MJ, White KE, Molinolo A, Chen MY, Heller T, Del Rivero J, Seo-Mayer P, Arabshahi B, Jackson MB, Hatab S, McCarthy E, Guthrie LC, Brillante BA, Gafni RI, Collins MT (2016) Phenotypic and genotypic characterization and treatment of a cohort with familial tumoral calcinosis/hyperostosis-hyperphosphatemia syndrome. J Bone Miner Res 31:1845–1854
Reilly AM, Gray AK, Moe SM, Ichikawa S (2014) Nicotinamide treatment in a murine model of familial tumoral calcinosis reduces serum Fgf23 and raises heart calcium. Bone 67:139–144
Ichikawa S, Gray AK, Padgett LR, Reilly AM, Unsicker TR (2014) High dietary phosphate intake induces development of ectopic calcifications in a murine model of familial tumoral calcinosis. J Bone Miner Res 29:2017–2023
Jost J, Bahans C, Courbebaisse M, Tran T-A, Linglart A, Benistan K, Lienhardt A, Mutar H, Pfender E, Ratsimbazafy V, Guigonis V (2016) Topical sodium thiosulfate: a treatment for calcifications in hyperphosphatemic familial tumoral calcinosis? J Clin Endocrinol Metab 101:2810–2815
Nguyen M, Boutignon H, Mallet E, Linglart A, Guillozo H, Jehan F, Garabedian M (2010) Infantile hypercalcemia and hypercalciuria: new insights into a vitamin D-dependent mechanism and response to ketoconazole treatment. J Pediatr 157:296–302
Sayers J, Hynes AM, Srivastava S, Dowen F, Quinton R, Datta HK, Sayer JA (2015) Successful treatment of hypercalcaemia associated with a CYP24A1 mutation with fluconazole. Clin Kidney J 8:453–455
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The local ethical committee approved this retrospective case description (Comité de Protection Lyon Sud Est II, IRB approval 00009118, session 20/04/2016).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Claramunt-Taberner, D., Bertholet-Thomas, A., Carlier, MC. et al. Hyperphosphatemic tumoral calcinosis caused by FGF23 compound heterozygous mutations: what are the therapeutic options for a better control of phosphatemia?. Pediatr Nephrol 33, 1263–1267 (2018). https://doi.org/10.1007/s00467-018-3945-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-018-3945-z