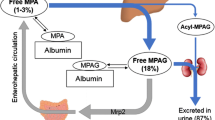
Abstract
Mycophenolate mofetil (MMF) is given to children in fixed doses based either on body weight or body surface area. There are data indicating mycophenolic acid (MPA) blood levels should be monitored in the early period of transplantation. However, there is little information regarding MPA pharmacokinetics (PK) in stable pediatric recipients. We evaluated MPA-PK in 20 stable renal transplant children (11.7±1.9 years) under long-term (46±31 months) MMF (26.1±7 mg/kg per day or 785±183 mg/m2 per day) therapy plus prednisone and cyclosporin A (n=16), tacrolimus (n=3), or MMF/prednisone (n=1). Total MPA levels were measured using the EMIT-MPA assay at 0, 1, 2, 3, 4, 6, and 8 h after an oral dose of MMF. The level at 12 h was considered equal to the trough level for AUC0–12 calculation. Mean C0, Cmax, AUC 0–12, and Tmax were 3.46±1.32, 13.5±0.58 μg/ml, 63.2±24.4 μg.h/ml, and 1.3±0.6 h, respectively. Six (30%) children were considered to have an adequate exposure (36–54 μg.h/ml) to MPA, 11 (55%) showed an AUC0–12 >54 μg.h/ml, and 3 (15%) showed an AUC0–12 <36 μg.h/ml. A Cmax ≥10 μg/ml was seen in 13 (65%) children. MMF dose did not correlate with AUC0–12 or Cmax. The combination of variables C0, C1, and C4 provided an equation to predict exposure (r 2=0.75) where AUC0–12=12.62+(7.78xC0)+(0.90xC1)+(1.30xC2) (P<0.001). The use of MMF without monitoring MPA blood levels may cause unnecessary overexposure to the drug in stable pediatric recipients.



Similar content being viewed by others
References
Hariharan S, Johnson CP, Bresnahan BA, Taranto SE, McIntosh MJ, Stablein D (2000) Improved graft survival after renal transplantation in the United States, 1988 to 1996. N Engl J Med 342:605–612
European Mycophenolate Mofetil Cooperative Study Group (1999) Mycophenolate mofetil in renal transplantation: 3-year results from the placebo-controlled trial. Transplantation 68:391–396
Ojo AO, Meier-Kriesche HU, Hanson JA, Leichtman AB, Cibrik D, Magee JC, Wolfe RA, Agodoa LY, Kaplan B (2000) Mycophenolate mofetil reduces late renal allograft loss independent of acute rejection. Transplantation 69:2405–2409
Weir MR, Anderson L, Fink JC, Gabregiorgish K, Schweitzer EJ, Hoehn-Saric E, Klassen DK, Cangro CB, Johnson LB, Kuo PC, Lim JY, Bartlett ST (1997) A novel approach to the treatment of chronic allograft nephropathy. Transplantation 64:1706–1710
David-Neto E, Araujo LM, Lemos FC, David DS, Mazzucchi E, Nahas WC, Arap S, Ianhez LE (2001) Introduction of mycophenolate mofetil and cyclosporin reduction in children with chronic transplant nephropathy. Pediatr Transplant 5:302–309
Lemos FBC, Ianhez LE, Sabbaga E, David-Neto E (1999) Is the improvement of renal function in chronic allograft nephropathy, after MMF introduction, related to cyclosporin A reduction (abstract). Transplantation 67:S86
Ferraris JR, Tambutti ML, Redal MA, Bustos D, Ramirez JA, Prigoshin N (2000) Conversion from azathioprine to mycophenolate mofetil in pediatric renal transplant recipients with chronic rejection. Transplantation 70:297-301
Weber LT, Shipkova M, Armstrong VW, Wagner N, Schutz E, Mehls O, Zimmerhackl LB, Oellerich M, Tonshoff B (2002) The pharmacokinetic-pharmacodynamic relationship for total and free mycophenolic acid in pediatric renal transplant recipients: a report of the German study group on mycophenolate mofetil therapy. J Am Soc Nephrol 13:759–768
Hale MD, Nicholls AJ, Bullingham RE, Hene R, Hoitsma A, Squifflet JP, Weimar W, Vanrenterghem Y, Van de Woude FJ, Verpooten GA (1998) The pharmacokinetic-pharmacodynamic relationship for mycophenolate mofetil in renal transplantation. Clin Pharmacol Ther 64:672–683
Weber LT, Shipkova M, Armstrong VW, Wagner N, Schutz E, Mehls O, Zimmerhackl LB, Oellerich M, Tonshoff B (2002) Comparison of the Emit immunoassay with HPLC for therapeutic drug monitoring of mycophenolic acid in pediatric renal-transplant recipients on mycophenolate mofetil therapy. Clin Chem 48:517–525
Oellerich M, Shipkova M, Schutz E, Wieland E, Weber L, Toshoff B, Armstrong VW (2000) Pharmacokinetic and metabolic investigations of mycophenolic acid in pediatric patients after renal transplantation: implications for therapeutic drug monitoring. German Study Group on Mycophenolate Mofetil Therapy in Pediatric Renal Transplant Recipients. Ther Drug Monit 22:20–26
Mourad M, Malaise J, Chaib Eddour D, De Meyer M, Konig J, Schepers R, Squifflet JP, Wallemacq P (2001) Pharmacokinetic basis for the efficient and safe use of low-dose mycophenolate mofetil in combination with tacrolimus in kidney transplantation. Clin Chem 47:1241–1248
Jacqz-Aigrain E, Khan Shaghaghi E, Baudouin V, Popon M, Zhang D, Maisin A, Loirat C (2000) Pharmacokinetics and tolerance of mycophenolate mofetil in renal transplant children. Pediatr Nephrol 14:95-99
Ryffel B, Mihatsch MJ, Fisher GL (1992) Immunosuppression and cancer: the cyclosporin case. Drug Chem Toxicol 15:95–115
Drouet E, Chapuis-Cellier C, Bosshard S, Verniol C, Niveleau A, Touraine JL, Garnier JL (1999) Oligo-monoclonal immunoglobulins frequently develop during concurrent cytomegalovirus (CMV) and Epstein-Barr virus (EBV) infections in patients after renal transplantation. Clin Exp Immunol 118:465–472
Shipkova M, Armstrong VW, Weber L, Niedmann PD, Wieland E, Haley J, Tonshoff B, Oellerich M (2002) Pharmacokinetics and protein adduct formation of the pharmacologically active acyl glucuronide metabolite of mycophenolic acid in pediatric renal transplant recipients. Ther Drug Monit 24:390–399
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet I:307–310
Bland JM, Altman DG (1995) Comparing methods of measurement: why plotting difference against standard method is misleading. Lancet 346:1085–1087
Schwartz GJ, Haycock GB, Edelmann CM Jr, Spitzer A (1976) A simple estimate of glomerular filtration rate in children derived from body length and plasma creatinine. Pediatrics 58:259–263
David-Neto E, Araujo LMP, Alves CF, Sumita NN, Nahas WC, Yanhez LE (2002) A strategy to calculate cyclosporin A area under the time concentration curve in pediatric renal transplantation. Pediatr Transplant 6:313–318
Filler G (2002) Cyclosporin A monitoring and AUC determination—where do we go? Pediatr Transplant 6:176–179
Filler G, Feber J, Lepage J, Weiler G, Mai I (2002) Universal approach to pharmacokinetic monitoring of immunosuppressive agents in children. Pediatr Transplant 6:1-9
Bunchman T, Navarro M, Broyer M, Sherbotie J, Chavers B, Tonshoff B, Birk P, Lerner G, Lirenman D, Greenbaum L, Walker R, Zimmerhackl LB, Blowey D, Clark G, Ettenger R, Arterburn S, Klamerus K, Fong A, Tang H, Thomas S, Ramos E (2001) The use of mycophenolate mofetil suspension in pediatric renal allograft recipients. Pediatr Nephrol 16:978–984
David-Neto E, Lemos FB, Furusawa EA, Schwartzman BS, Cavalcante JS, Yagyu EM, Romano P, Ianhez LE (2000) Impact of cyclosporin A pharmacokinetics on the presence of side effects in pediatric renal transplantation. J Am Soc Nephrol 11:343–349
Armstrong VW, Oellerich M (2001) New developments in the immunosuppressive drug monitoring of cyclosporine, tacrolimus, and azathioprine. Clin Biochem 34:9–16
Aspeslet LJ, Yatscoff RW (2000) Requirements for therapeutic drug monitoring of sirolimus, an immunosuppressive agent used in renal transplantation. Clin Ther 22:B86–B92
Belitsky P, Dunn S, Johnston A, Levy G (2000) Impact of absorption profiling on efficacy and safety of cyclosporin therapy in transplant recipients. Clin Pharmacokinet 39:117–125
Bergan S, Rugstad HE, Bentdal O, Sodal G, Hartmann A, Leivestad T, Stokke O (1998) Monitored high-dose azathioprine treatment reduces acute rejection episodes after renal transplantation. Transplantation 66:334–339
Hubner GI, Eismann R, Sziegoleit W (1999) Drug interaction between mycophenolate mofetil and tacrolimus detectable within therapeutic mycophenolic acid monitoring in renal transplant patients. Ther Drug Monit 21:536–539
Squifflet JP, Backman L, Claesson K, Dietl KH, Ekberg H, Forsythe JL, Kunzendorf U, Heemann U, Land W, Morales JM, Muhlbacher F, Talbot D, Taube D, Tyden G, van Hooff J, Schleibner S, Vanrenterghem Y (2001) Dose optimization of mycophenolate mofetil when administered with a low dose of tacrolimus in cadaveric renal transplant recipients. Transplantation 72:63–69
Filler G, Zimmering M, Mai I (2000) Pharmacokinetics of mycophenolate mofetil are influenced by concomitant immunosuppression. Pediatr Nephrol 14:100–104
Pirsch J, Bekersky I, Vincenti F, Boswell G, Woodle ES, Alak A, Kruelle M, Fass N, Facklam D, Mekki Q (2000) Coadministration of tacrolimus and mycophenolate mofetil in stable kidney transplant patients: pharmacokinetics and tolerability. J Clin Pharmacol 40:527–532
Zucker K, Rosen A, Tsaroucha A, Faria L de, Roth D, Ciancio G, Esquenazi V, Burke G, Tzakis A, Miller J (1997) Unexpected augmentation of mycophenolic acid pharmacokinetics in renal transplant patients receiving tacrolimus and mycophenolate mofetil in combination therapy, and analogous in vitro findings. Transpl Immunol 5:225–232
Acknowledgements
This study was partially supported by Dade-Behring-Brazil and presented at the ATC 2002, Annual Meeting of the AST/ASTS, Washington, and at the World Congress of Transplantation, Miami 2002, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
David-Neto, E., Araujo, L.M.P., Sumita, N.M. et al. Mycophenolic acid pharmacokinetics in stable pediatric renal transplantation. Pediatr Nephrol 18, 266–272 (2003). https://doi.org/10.1007/s00467-002-1057-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-002-1057-1