Abstract
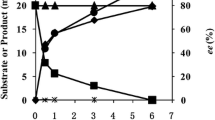
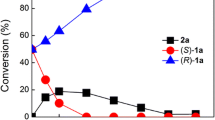
The stereoselective three-enzyme cascade for the one-pot synthesis of (1S,2S)-1-phenylpropane-1,2-diol ((1S,2S)-1-PPD) from inexpensive starting substrates, benzaldehyde and acetaldehyde, was explored. By coupling stereoselective carboligation catalyzed by benzoylformate decarboxylase (BFD), L-selective reduction of a carbonyl group with alcohol dehydrogenase from Lactobacillus brevis (ADHLb) as well as the coenzyme regeneration by formate dehydrogenase (FDH), enantiomerically pure diastereoselective 1,2-diol was produced. Two different multi-enzyme system approaches were applied: the sequential two-step one-pot and the simultaneous one-pot cascade. All enzymes were kinetically characterized. The impact of acetaldehyde on the BFD and ADHLb stability was investigated. To overcome the kinetic limitation of acetaldehyde in the carboligation reaction and to reduce its influence on the enzyme stability, experiments were performed in two different excesses of acetaldehyde (100 and 300%). Due to the ADHLb deactivation by acetaldehyde, the simultaneous one-pot cascade proved not to be the first choice for the investigated three-enzyme system. In the sequential cascade with 300% acetaldehyde excess a 100% yield of vic 1,2-diol was reached.







Similar content being viewed by others
References
Ricca E, Brucher B, Schrittwieser JH (2011) Multi-enzymatic cascade reactions: overview and perspectives. Adv Synth Catal 353:2239–2262
Enoki J, Meisborn J, Müller AC, Kourist R (2016) A multi-enzymatic cascade reaction for the stereoselective production of γ-oxyfunctionalyzed amino acids. Front Microbiol 7:425
Monti D, Ferrandi EE, Zanellato I, Hua L, Polentini F, Carrea G, Riva S (2009) One-pot multienzymatic synthesis of 12-ketoursodeoxycholic acid: subtle cofactor specificities rule the reaction equilibria of five biocatalysts working in a row. Adv Synth Catal 351:1303–1311
Burton SG, le Roes-Hill M (2008) In: Garcia-Junceda E (ed) Multi-step enzyme catalysis—biotransformations and chemoenzymatic synthesis. Wiley, Weinheim
Fischer T, Pietruszka J (2010) Key building blocks via enzyme-mediated synthesis. Top Curr Chem 297:1–43
Hailes HC, Dalby PA, Woodley JM (2007) Integration of biocatalytic conversions into chemical syntheses. J Chem Technol Biotechnol 82:1063–1066
Xue R, Woodley JM (2012) Process technology for multi-enzymatic reaction systems. Bioresour Technol 115:183–195
Rios-Solisa L, Morrisa P, Granta C, Odeleyea AO, Hailesb HC, Warda JM, Dalbya PA, Baganza F, Lyea GJ (2015) Modelling and optimisation of the one-pot, multi-enzymatic synthesis of chiral amino-alcohols based on microscale kinetic parameter determination. Chem Eng Sci 122:360–372
Hall M, Bommarius AS (2011) Enantioenriched compounds via enzyme-catalyzed redox reactions. Chem Rev 111:4088–4110
Lavandera I, Kern A, Ferreira-Silva B, Glieder A, de Wildeman S, Kroutil W (2008) Stereoselective bioreduction of bulky-bulky ketones by a novel ADH from Ralstonia sp. J Org Chem 73:6003–6005
Patel RN (2008) Synthesis of chiral pharmaceutical intermediates by biocatalysis. Coord Chem Rev 252:659–701
Choudary BM, Chowdari NS, Madhi S, Kantam ML (2003) A trifunctional catalyst for one-pot synthesis of chiral diols via heck coupling—n-oxidation—asymmetric dihydroxylation: application for the synthesis of diltiazem and taxol side chain. J Org Chem 68:1736–1746
Goldberg K, Schroer K, Lütz S (2007) Biocatalytic ketone reduction–a powerful tool for the production of chiral alcohols-part II: whole-cell reductions. Appl Microbiol Biotechnol 76:237–255
Kulig J, Simon R, Rose C, Husain S, Häckh M, Lüdke S, Zeitler K, Kroutil W, Pohl M, Rother D (2012) Stereoselective synthesis of bulky 1,2-diols with alcohol dehydrogenases. Catal Sci Technol 2:1580–1589
Kihumbu D, Stillger T, Hummel W, Liese A (2002) Enzymatic synthesis of all stereoisomers of 1-phenylpropane-1,2-diol. Tetrahedron: Asymmetry 13:1069–1072
Shanmuganathan S, Natalia D, Greiner L, Dominguez de Maria P (2012) Oxidation-hydroxymethylation-reduction: a one-pot three-step biocatalytic synthesis of optically active a-aryl vicinal diols. Green Chem 14:94–97
Švarc A, Valinger D, Vasić-Rački Đ, Presečki AV (2015) Stereoselective synthesis of phenylpropane 1,2-diols from (S)-2-hydroxypropiophenone by NADH-dependent oxidoreductases. Biochem Eng J 103:250–255
Jakoblinnert A, Rother D (2014) A two-step biocatalytic cascade in micro-aqueous medium: using whole cells to obtain high concentrations of a vicinal diol. Green Chem 16:3472–3482
Bencze LC, Paizs C, Tosa ML, Dan Irimie F, Retey J (2011) Chemoenzymatic one-pot synthesis of both (R)-and (S)-aryl-1,2-ethanediols. ChemCatChem 3:343–346
Kamal A, Sandbhor M, Ahmed K, Adil SF, Ali Shaik A (2013) Chemoenzymatic synthesis of enantiomerically pure terminal 1,2-diols. Tetrahedron: Asymmetry 14:3861–3866
Iding H, Dünnwald T, Greiner L, Liese A, Müller M (2000) Benzoylformate decarboxylase from Pseudomonas putida as stable catalyst for the synthesis of chiral 2-hydroxy ketones. Chem Eur J 6:1483–1495
Peper S, Kara S, Long WS, Liese A, Niemeyer B (2011) Immobilization and characterization of benzoylformate decarboxylase from Pseudomonas putida on spherical silica carrier. Bioprocess Biosyst Eng 34:671–680
Aymard C, Belarbi A (2000) Kinetics of thermal deactivation of enzymes: a simple three parameters phenomenological model can describe the decay of enzyme activity, irrespectively of the mechanism. Enzym Microb Technol 27:612–618
Polakovič M, Bryjak J (2002) Modelling of the kinetics of thermal inactivation of glucoamylase from Aspergillus niger. J Mol Catal B: Enzym 19–20:443–450
Kara S, Long WS, Berheide M, Peper S, Niemeyer B, Liese A (2011) Influence of reaction conditions on the enantioselectivity of biocatalyzed C–C bond formations under high pressure conditions. J Biotechnol 152:87–92
Kuznetsova IM, Turoverov KK, Uversky VN (2014) What macromolecular crowding can do to a protein. Int J Mol Sci 15:23090–23140
Hollmann F, Arends IW, Holtmann D (2011) Enzymatic reductions for the chemist. Green Chem 13:2285–2314
Dominguez de Maria P, Stillger T, Pohl M, Kiesel M, Liese A, Groger H, Trauthwein H (2008) Enantioselective C–C bond ligation using recombinant Escherichia coli-whole-cell biocatalysts. Adv Synth Catal 350:165–173
Jakoblinnert A, Mladenov R, Paul A, Sibilla F, Schwaneberg U, Ansorge-Schumacher MB, Dominguez de Maria P (2011) Asymmetric reduction of ketones with recombinant E. coli whole cells in neat substrates. Chem Commun 47:12230–12232
Acknowledgements
This work was supported by University of Zagreb short-term financial scientific research support under the title “Mathematical modeling of the biocatalytic synthesis of industrially interesting products”. The authors would like to thank Prof. Martina Pohl from the Institute of Bio- and Geosciences, IBG-1: Biotechnology, Research center Jülich, Germany for the gift of alcohol dehydrogenase from Lactobacillus brevis and Davor Valinger from Faculty of Food Technology and Biotechnology, University of Zagreb for the isolation of enzyme benzoylformate decarboxylase.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Presečki, A.V., Pintarić, L., Švarc, A. et al. Different strategies for multi-enzyme cascade reaction for chiral vic-1,2-diol production. Bioprocess Biosyst Eng 41, 793–802 (2018). https://doi.org/10.1007/s00449-018-1912-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-018-1912-5